Continuously tracking chemicals in the bloodstream can give doctors a critical insight into a person’s health. Take glucose. A recently–FDA-approved artificial pancreas measures blood glucose levels every few minutes and gives an appropriate insulin dose will likely save many a diabetic’s life.
A team led by Stanford University electrical engineer H. Tom Soh has taken that idea even further, developing a universal biosensor system that can not only track, but also control, in real-time, the concentration of drugs in the bloodstreams of live animals. The biosensor can measure a wide variety of molecules, including various drugs and potentially even proteins. “There could be some pretty cool applications,” Soh says.
What the artificial pancreas does for diabetes, the small electronic biosensor could do for several other life-threatening conditions, from thrombosis to cancer. It would be especially useful for administering drugs such as anticancer drugs and antibiotics. For those treatments, there is a narrow range of doses in which there is enough of the drug for it to be effective, but not so much that it causes unwanted side effects, says Soh. By tracking drug or enzyme levels in real-time, the biosensor system could deliver drugs right at the dosage sweet spot at just the right time. In the future, it may also be able to spot proteins that provide early warning of infection, heart attacks, and diseases.
In 2013, Soh and his colleagues reported that they had used the sensor to measure, in rats and humans, micromolar concentrations of a chemotherapy drug and an antibiotic.
Soh’s postdoctoral researcher, Peter Mage, has since made the device much faster, with less measurement lag. Mage has tested a closed-loop system that can continually control drug concentration in live animals much like the artificial pancreas controls blood sugar. In the lab setup, blood drawn continuously from one ear of a rabbit goes into the sensor. The researchers inject the rabbit with the chemotherapy drug doxorubicin, which is widely used but difficult to dose. The sensor measures drug concentration, and the data goes to a computerized feedback system that figures out how much of the drug should be introduced in order to keep blood levels ideal. Then a programmable pump injects that amount of the drug into the animal’s other ear. Soh presented these details at the IEEE EMBS Micro and Nanotechnology in Medicine Conference on 16 December.
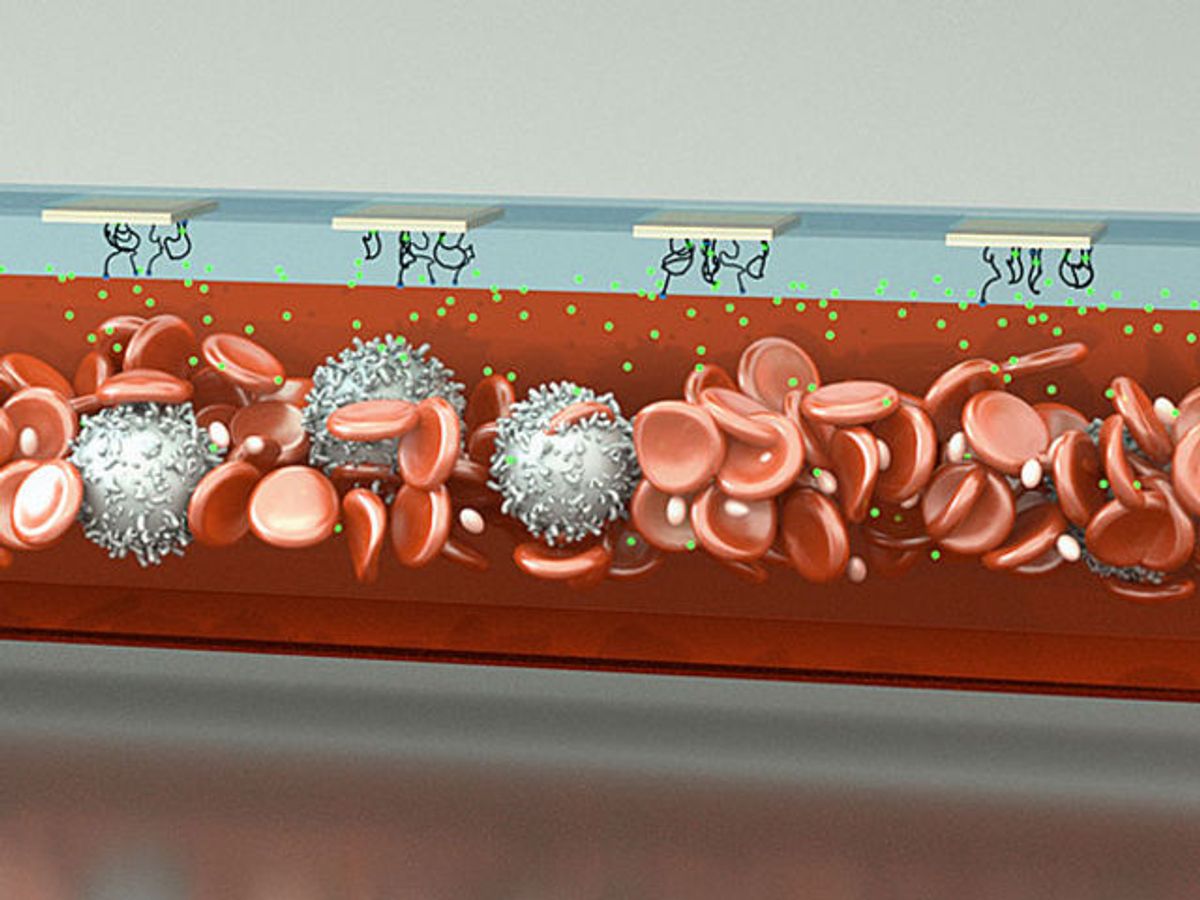
The device features a setup comprising two glass wafers sandwiching a drilled microfluidic channel. The channel is lined with gold electrodes, with artificial DNA strands called aptamers sticking out. When a target molecule comes by, the aptamer wraps around it, bringing its tip down so that electrons from the tip jump to the gold electrode, creating a tiny measurable current spike.
Previous aptamer-based sensors didn’t work well in whole blood because blood cells or large proteins tended to clog them. To get around that, the Stanford researchers inject a thin stream of buffer solution between the blood and the electrode. This acts like a filter, keeping large particles at bay, but letting smaller target molecules through to reach the electrodes.
Switching the aptamers allows the sensor to detect different molecules. The sensor currently works for a few hours, but the researchers aim to make a longer-lasting device with disposable sensor elements that could be swapped out for continuous long-term monitoring. Soh and his team are now working with pediatric cancer specialists to measure the precise therapeutic windows for various drugs in children.
Prachi Patel is a freelance journalist based in Pittsburgh. She writes about energy, biotechnology, materials science, nanotechnology, and computing.