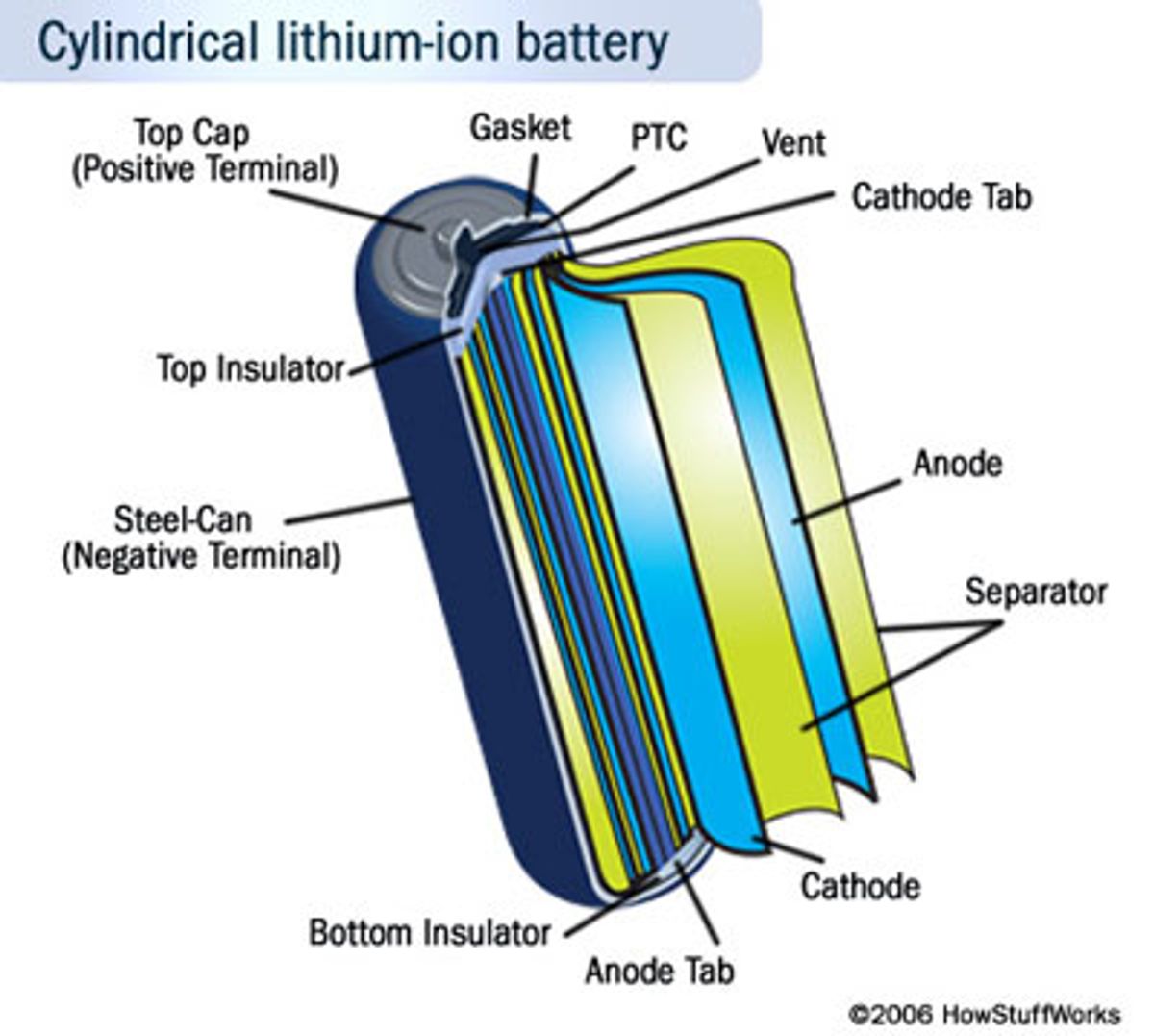
It is my fervent belief that nanotechnology’s ability to push the lowly battery to new heights will be one of the field’s biggest achievements in the not-too-distant future. Sure expanding the water supply and better harvesting the sun’s energy are no doubt big achievements. But from a very personal level, I want my cell phone, MP3 player and laptop to last a lot longer on charge than they currently do.
Researchers Bharat Bhushan, Suresh Babu and Lei Raymond Cao first started with infrared thermal imaging of each electrode and soon progressed to using scanning electron microscopy, atomic force microscope, scanning spreading resistance microscopy, Kelvin probe microscopy, transmission electron microscopy all to get different length scale resolutions. (This various length scale issue is a big one as I have pointed out before. While we are working out making batteries, let’s see if we can’t get an equivalent to Google Earth for microscopy tools.)
What they discovered was that “the finely-structured nanomaterials on these electrodes that allow the battery to rapidly charge and discharge had coarsened in size.”
They also found out with neutron depth profiling that most of the lithium was no longer available for charge transfer. Now the researchers have not yet connected the coarsening of the nanomaterials on the electrodes with this loss of lithium, but their future research may establish this connection. In terms of real-world applications the article cited above points to this research enabling a faster rollout of electric cars. Electric cars? I would like my iPod to hold a charge for longer than 30 minutes after owning it for a couple of years.Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.