The astonishing thing about stem cells is that they can be coaxed, in the laboratory, into becoming nearly any kind of cell—from bone marrow to heart muscle. That remarkable capability has for years kept scientists busy tinkering with stem cells and injecting them into animal models in an attempt to grow and replace damaged tissue.
Such scientists have received a ton of attention in that line of work. But there’s a smaller group of researchers working, to far less fanfare, on a different part of the stem cell challenge: how to deliver those cells to the body’s hard-to-reach places.
Researchers typically deliver stem cells via injection—a needle. But that method can damage healthy tissue, especially when the target is a deep brain structure, or delicate vasculature, or the inner ear.
A group out of Hong Kong announced this week that they had invented a new delivery tool using tiny, magnetically-controlled robots. The cell-carrying machines move noninvasively through the body to a target site and deliver their stem cell cargo.
The researchers, led by Dong Sun, a professor at City University of Hong Kong, demonstrated their device in zebrafish and mice, and reported their success in the journal Science Robotics.
The advance is notable because Sun and his colleagues were able to demonstrate that their robots work in animals. “It’s really uncertain how to make these tiny machines move in living organisms,” says Bradley Nelson, a microroboticist at ETH Zürich, who was not involved in the project.
Several other groups, including Nelson’s, have demonstrated cell-carrying microbot designs in computer simulations and test tubes, but “in vivo is harder,” he says.

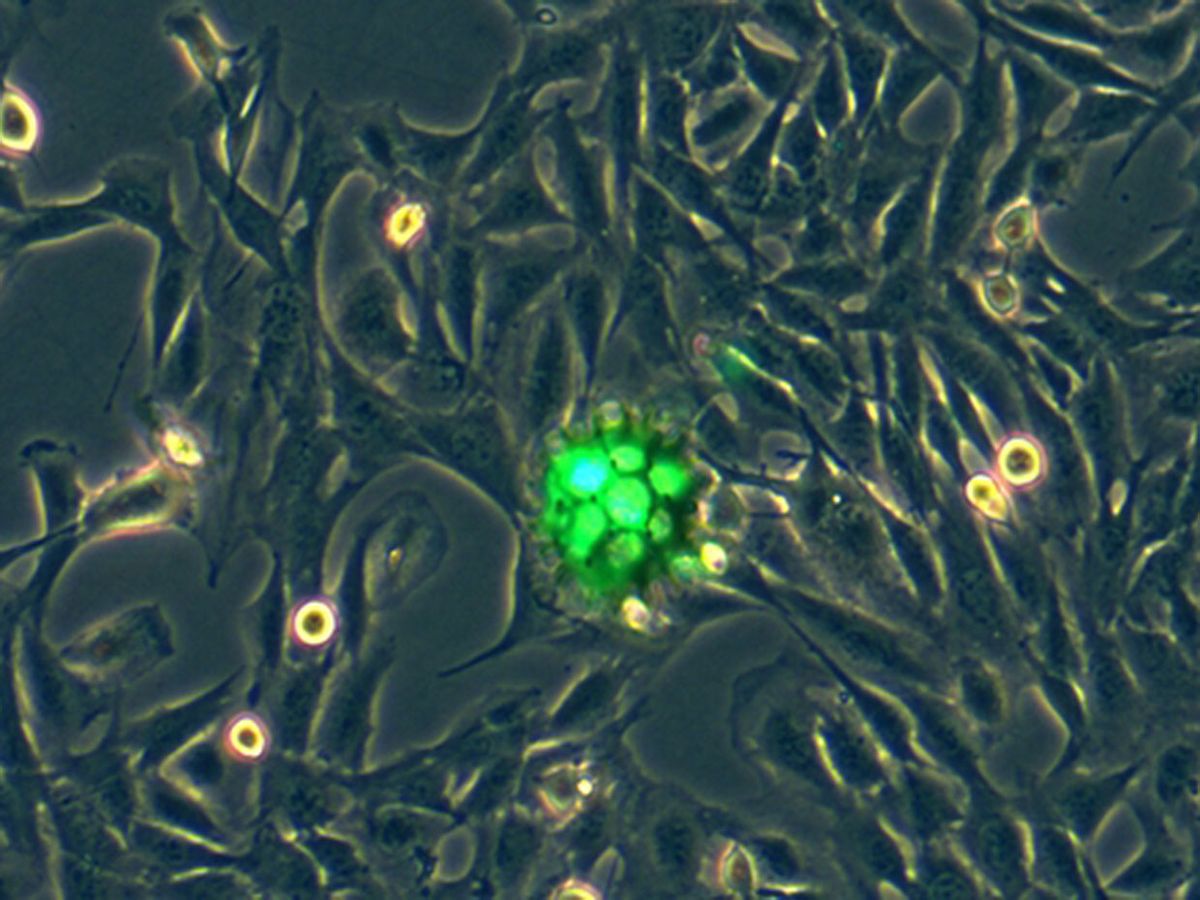
Sun designed the robots to resemble burrs (the prickly spherical seed pods that cling to clothing). He and his colleagues fabricated them using 3D laser lithography, and coated them in nickel for magnetic actuation, and titanium, for biocompatibility.
The porous, burr-shaped bots hold cells between their spikes. Using magnetic fields, researchers can guide the bots to a site of interest in the body. There, they release the cells, which then proliferate and do their job in regenerating damaged tissue.

Sun and his colleagues tested the mobility of the robots by injecting them into the yolks of anesthetized zebrafish embryos. They navigated them around to specified points inside the yolk while observing that the heart of the zebrafish continued to beat.
Then they tested whether the robots could carry and release cells. They loaded them up with a voracious type of cell called HeLa, and injected swarms of them into mice. The robots did their job in releasing the cells, which then proliferated in the mice.
A next step would be to load up the microbots with stem cells to test their therapeutic capabilities. One limitation of the design is that the cells are released spontaneously, rather than with a controlled mechanism.
Nelson cautions that Sun’s burr-bots are only one piece in the stem cell puzzle. Far more work must be done before stem cell therapies can be used to actually heal organs or treat disease.
The cell-carrying bots add to the growing list of medical microrobots designed to perform various jobs in the body, such as delivering drugs or fighting infections.
Emily Waltz is the power and energy editor at Spectrum. Prior to joining the staff in January 2024, Emily spent 18 years as a freelance journalist covering biotechnology, primarily for the Nature research journals and Spectrum. Her work has also appeared in Scientific American, Discover, Outside, and the New York Times. Emily has a master's degree from Columbia University Graduate School of Journalism and an undergraduate degree from Vanderbilt University. With every word she writes, Emily strives to say something true and useful. She posts on Twitter/X @EmWaltz and her portfolio can be found on her website.