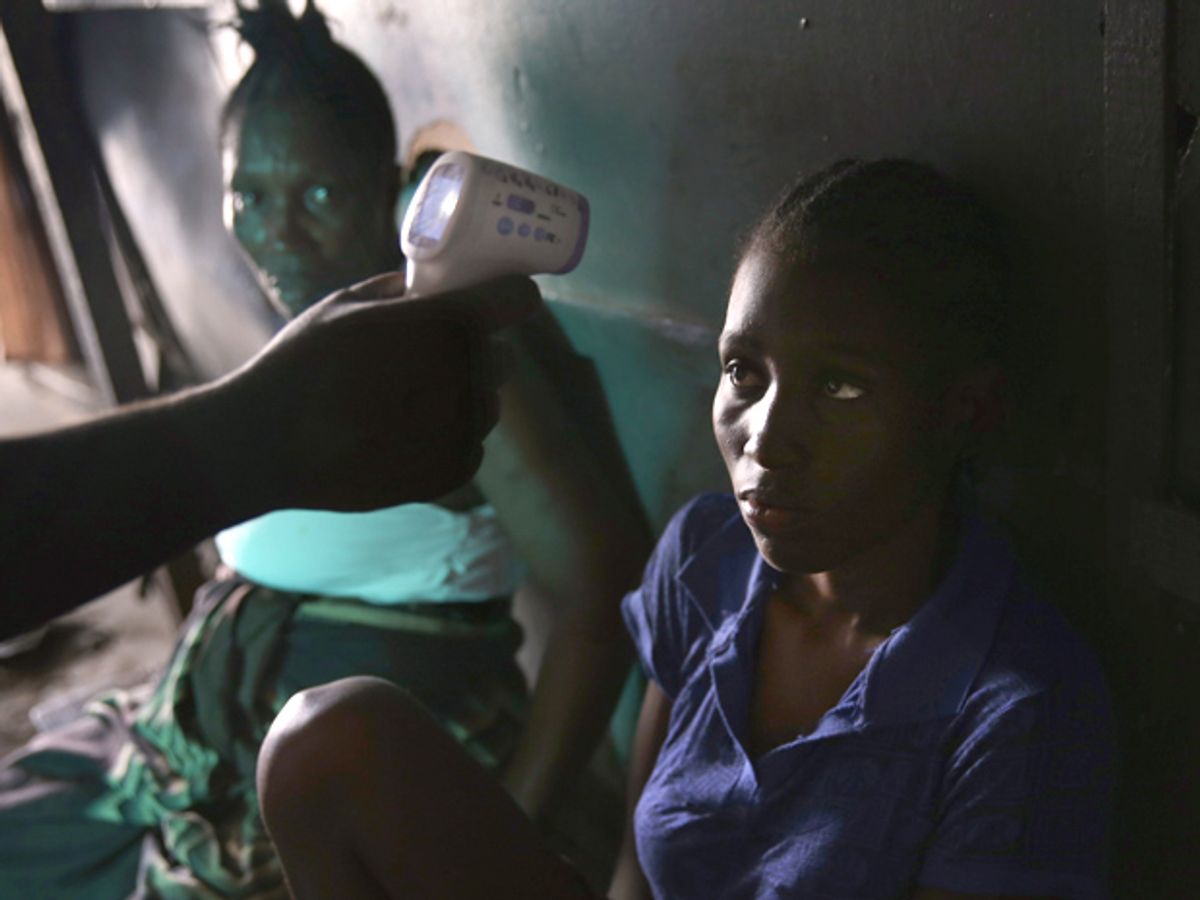Researchers are working to improve public health responses to Ebola outbreaks by enhancing computational models of the disease’s spread. A team led by researchers at Arizona State University received two grants from the National Science Foundation—one for $120,000 awarded in December and one for half a million awarded in 2013—to make it happen.
The research team aims to improve disease models by making them more specific to Ebola epidemics. They will use a combination of analytical tools and algorithms to sift through tens of thousands of simulations of an Ebola epidemic to determine which parameters provide the key patterns of the virus’s transmission. They will combine that with real mobility data—observations of social behviors, interventions, and ground and air traffic patterns collected during the current Ebola epidemic in West Africa.
The researchers say they hope that their improved model will help decision makers make better-informed choices in future outbreaks. “We don’t know if it will be applicable to the current outbreak,” since the epidemic’s trajectory has, fortunately, slowed, says Kasim Selcuk Candan, a team leader and computer scientist specializing in data management and analysis at Arizona State University in Tempe, in an interview with Spectrum. Candan says he hopes to have a model ready by spring.
The current Ebola virus outbreak has infected more than 21,000 people worldwide, and killed more than 8,400, according to the latest numbers from the World Health Organization. Scientists are developing experimental drugs, vaccines and even gold nanostars to combat the virus.
Ebola’s spread is tough to predict because when an outbreak occurs, people’s behavior changes. They go places they normally don’t go. They flee or avoid an affected area or head to a region where they can get treatment. They respond to education campains on how to avoid contracting the virus. Air traffic and local ground traffic changes.
Intervention efforts of local decision makers can also bring about changes both good and bad, depending on how they are implemented. A local official may decide to isolate a group of people, and instead of complying, people flee. An official decides to close a school and that affects local ground traffic and social interaction patterns. A treatment center is set up and it becomes an incubator of the disease, rather than a stopping point. Burial practices change. The list goes on.
Not only do changes in social interaction occur, but those changes tend to be a moving target as a disease progresses. That’s what makes predicting the spread of Ebola so difficult. And without the information from models, decisions made about interventions often aren’t well-informed.
“It’s extremely hard to make a global model that is effective,” says Candan. To make such a model, one has to understand complex mobility patterns across multiple countries with different policies and intervention strategies—all of which may change as the disease progresses, he says. “There are lots of unknowns,” he says.
There are large-scale models for simulating the spread of disease, including The Global Epidemic and Mobility Model (GLEAM) and the Spatiotemporal Epidemiological Modeler (STEM). The simulators work like this: punch in the disease characteristics (infection rate, recovery rate, etc), some social components (assumptions about local and global interactions and mobility patterns), plus intervention steps (such as vaccinations, school closures and quarantine), along with a starting point (metro area versus rural village), and the model will tell you how the disease will evolve over time.
But these simulators were designed for highly communicable diseases such as influenza, rather than Ebola, which is transmitted through close contact. And there’s a bigger problem: If the models are going to be effective, they have to be continuously revised to reflect the evolution of an epidemic, and that’s hard to do. “Doing one simulation is good if you know all the parameters and you want to test one intervention,” says Candan. “But when you want to understand a complex disease like Ebola...you need to run many of these simulations—tens of thousands of them—and each simulation gives a potentially different view of the disease,” he says. “The question is can we sift through all of these simulations to understand what are the critical parameters and critical decisions being made, and which one of these seems to match what you observe on the ground the best?”
Sifting through the thousands of simulations and interpreting them is the heart of Candan’s team’s project. To address that, the team is developing novel algorithms in what it calls an “epidemic data management system” or EpiDMS. They plan to combine that with the use of novel data mining and analysis tools. The team includes Gerardo Chowell-Puente, a mathematical epidemiologist at Georgia State University in Atlanta, and Maria Luisa Sapino, a data analysis expert at the University of Torino in Italy.
The observational data on interventions and behaviors will come from the team’s contacts in West Africa and at Columbia University, who collected the data in West Africa during the epidemic. They will also use epidemiological data of the epidemic’s trajectory that has been made publicly available by the Wold Health Organization. Candan’s team also plans to buy air traffic data that details activity during certain time periods before and during the epidemic to calibrate the model on patterns of international spread.
Spectrum asked Candan: What if geopolitical and social practices are completely different when the next Ebola outbreak hits—will the data and models we generate now be applicable? “The past is not a perfect predictor of the future,” Candan replied. “There might be a civil war” that changes everything. What the improved models can tell us is the reaction to specific intervention choices. Says Candan: “What we can say is: ‘You don’t want to take this action because it results in X.”
Emily Waltz is a features editor at Spectrum covering power and energy. Prior to joining the staff in January 2024, Emily spent 18 years as a freelance journalist covering biotechnology, primarily for the Nature research journals and Spectrum. Her work has also appeared in Scientific American, Discover, Outside, and the New York Times. Emily has a master's degree from Columbia University Graduate School of Journalism and an undergraduate degree from Vanderbilt University. With every word she writes, Emily strives to say something true and useful. She posts on Twitter/X @EmWaltz and her portfolio can be found on her website.