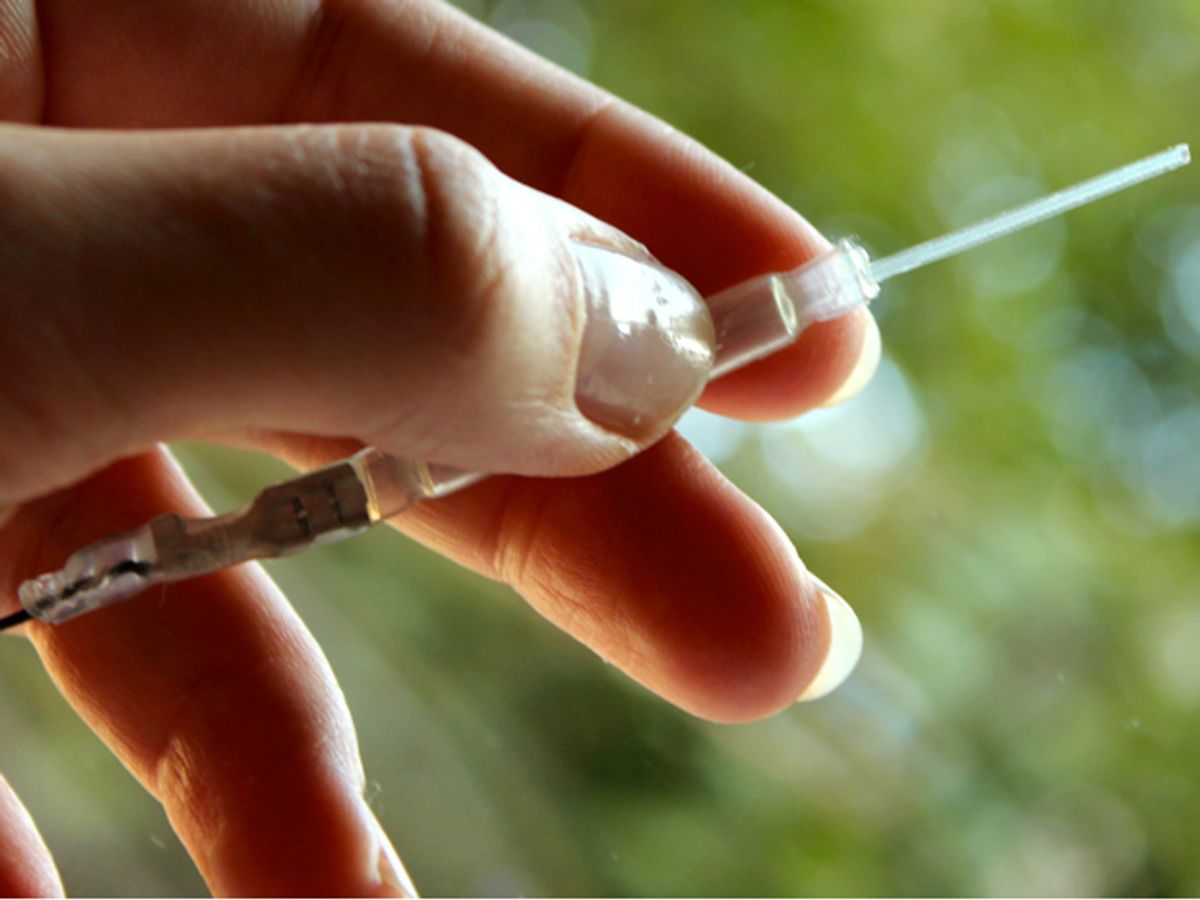
Researchers invented an implanted bioelectronic device that delivers therapeutic drugs to rats’ spinal cords with unprecedented precision, reducing pain without side effects, according to a study published today in Science Advances. The researchers say that by delivering lower doses of pain medication in a more targeted way, side effects that often accompany traditional administration of pain medications can be avoided.
“To the best or our knowledge, this is the first demonstration of drug delivery with a bioelectronic device in such a selective way,” says Daniel T. Simon, an assistant professor at Linköping University in Sweden, and a researcher on the project. “There has been electrophoretic delivery of drugs done. But there hasn’t been this type of precision and local delivery,” he says.
Localized delivery was the key, says Simon. Pain medications and other drugs are often administered systemically—spreading throughout the body. Someone recovering from a knee surgery, for example, might take an oral narcotic to block the pain, but in doing so, other pain and regulatory signals in the nervous system are also blocked, causing lethargy. A drug that can be administered directly to the site of injury in small doses could reduce such side effects.
The new devices, an organic electronic ion pump custom designed for the anatomy of rats, is based on the principle of electrophoresis: the transport of molecules through a fluid or gel under the influence of an electric field. “This is the technique you see when people are doing genetic analysis and separating strands of DNA,” says Simon. “It’s extremely common in the lab.”
What’s uncommon is the use of electrophoresis with a conducting polymer to precisely deliver a therapeutic substance. That substance, in this case, is GABA (gamma-aminobuyric acid), which is the primary inhibitory neurotransmitter in the central nervous system. “What we’re doing here is we’re moving charged neurotransmitters—in this case GABA—through an electric field inside of a selective gel,” says Simon. He calls the gel selective because it is loaded with negative charges, allowing only positively charged substances such as GABA to pass through.
The device consists of a reservoir for the drug, a conducting polymer control electrode, and a channel containing the transport gel. The channel has four drug delivery outlets designed to target specific neural junctions on the spinal cord. When a voltage is applied, electrons withdraw from the polymer electrode, and GABA pumps through the selective transport gel, eventually reaching the four outlets. At those four points on the spinal cord, GABA interferes with the pain signaling.
The dosage delivered is extremely precise. For each electron extracted from the conducting polymer, an ion—a positively charged GABA molecule—takes part in the electrochemical reaction to complete the circuit. The ion pump selects for that positive charge, pushing it from the reservoir and out the door. “It’s a one-to-one ratio, and we have precise control over it,” says Simon. “If we want to deliver 1000 molecules at the tip of the device we specify 1000 electrons. And as soon as we turn off the voltage, we don’t get an diffusion of the drugs out of the device” because the GABA is compensating for the negative charges in the transport gel material, he says.
To test the device, the researchers used rats with nerve injuries. The rats’ sciatic nerves were cut in a way that induced abnormal pain signals to shoot up from the hind paws to the spinal cord, and up to the brain where it was perceived as pain. The induced injury mimics neuropathic pain in a human.
The researches aimed the GABA delivery at four sites on the rats’ spinal cord where the pain signal coming from the hind legs connects. The goal was to stop the pain signal at that point of connection, before it traveled up the spinal cord to the brain. The experiment worked. Rats treated with GABA were not nearly as sensitive to pain as the control group.
“You can take opium and that can have the same effect, but in that case you’re turning off all the pain signals,” which can lead to side effects, says Simon. “The idea with our device is to just turn off this one malfunctioning signal.”
Previous methods of delivering drugs using an electric field—a technique generally called iontophoresis—have not been nearly as effective at selectively delivering only the drug without the accompaniment of other substances, making dosage measurements difficult. The researchers say their demonstration presents a viable alternative to existing pain treatments, and paves the way for future implantable bioelectronic therapeutics.
The study was conducted by a collaboration of engineers and scientists led by Magnus Bergen at the Laboratory of Organic Electronics at Linköping University and the research team of Bengt Linderoth in the department of clinical neuroscience at the Karolinska Institute in Stockholm.
Emily Waltz is a features editor at Spectrum covering power and energy. Prior to joining the staff in January 2024, Emily spent 18 years as a freelance journalist covering biotechnology, primarily for the Nature research journals and Spectrum. Her work has also appeared in Scientific American, Discover, Outside, and the New York Times. Emily has a master's degree from Columbia University Graduate School of Journalism and an undergraduate degree from Vanderbilt University. With every word she writes, Emily strives to say something true and useful. She posts on Twitter/X @EmWaltz and her portfolio can be found on her website.