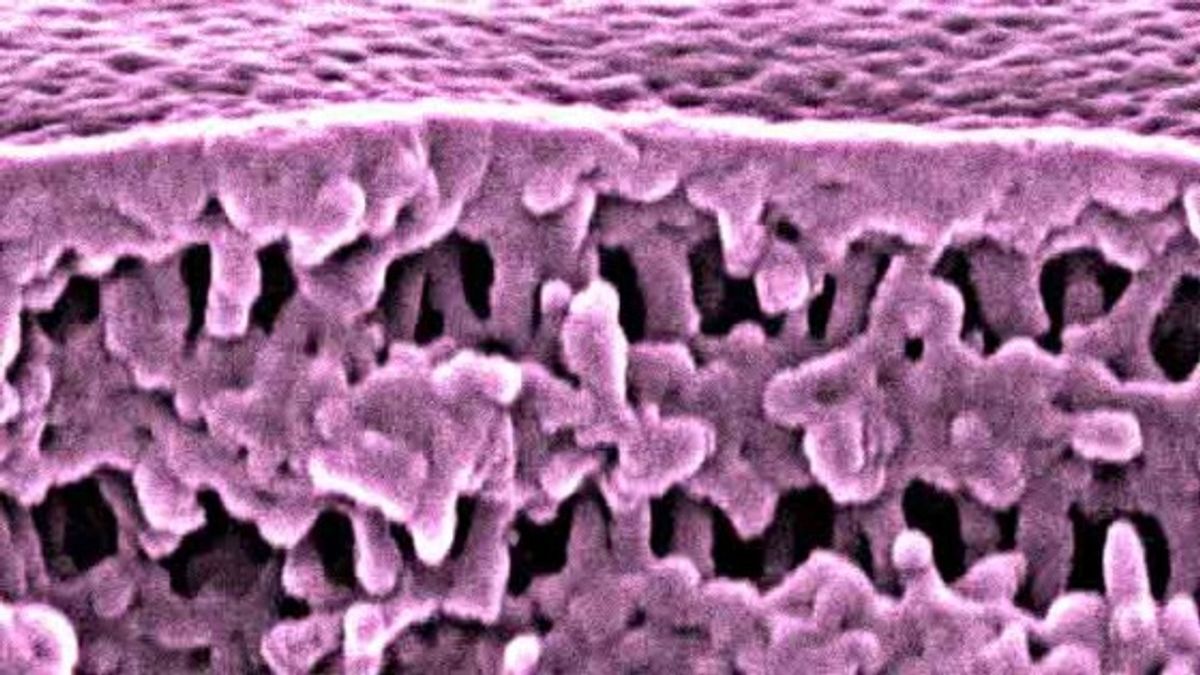
This method provides a far more flexible method than what had been employed up till now. In the now old method when the minor component was removed to leave behind the pores it was necessary that the component had to be connected both throughout the material and to the outside of the material.
The research, which was led by Dr Easan Sivaniah and published in the journal Nature Materials, showed that a process called collective osmotic shock (COS) meant that even when the minor component is entirely encapsulated that material is still porous.
"The experiment is rather similar to the classroom demonstration using a balloon containing salty water,” explains Sivaniah in the Cambridge University press release covering the research. “How does one release the salt from the balloon? The answer is to put the balloon in a bath of fresh water. The salt can't leave the balloon but the water can enter, and it does so to reduce the saltiness in the balloon. As more water enters, the balloon swells, and eventually bursts, releasing the salt completely.
"In our experiments, we essentially show this works in materials with these trapped minor components, leading to a series of bursts that connect together and to the outside, releasing the trapped components and leaving an open porous material."
Initial applications appear to be in water filtration systems. While this may not be a groundbreaking application for nanoporous materials it is an area in which there is still an acute need for new and better systems to create clean drinking water.
"It is currently an efficient filter system that could be used in countries with poor access to fresh potable water, or to remove heavy metals and industrial waste products from ground water sources,” says Sivaniah. “Though, with development, we hope it can also be used in making sea-water drinkable using low-tech and low-power routes."
Intriguingly the researchers have been collaborating with other labs in using the material in photonic and optoelectronic applications.
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.