Updated 1 November 2018
After what has seemed a bit of a lapse in the timeline of their development, graphene-enabled supercapacitors may be poised to make a significant advance. Researchers at the University of California, Santa Cruz, and Lawrence Livermore Laboratory (LLNL) have developed an electrode for supercapacitors made from a graphene-based aerogel. The new supercapacitor component has the highest areal capacitance (electric charge stored per unit of surface area) ever reported for a supercapacitor.
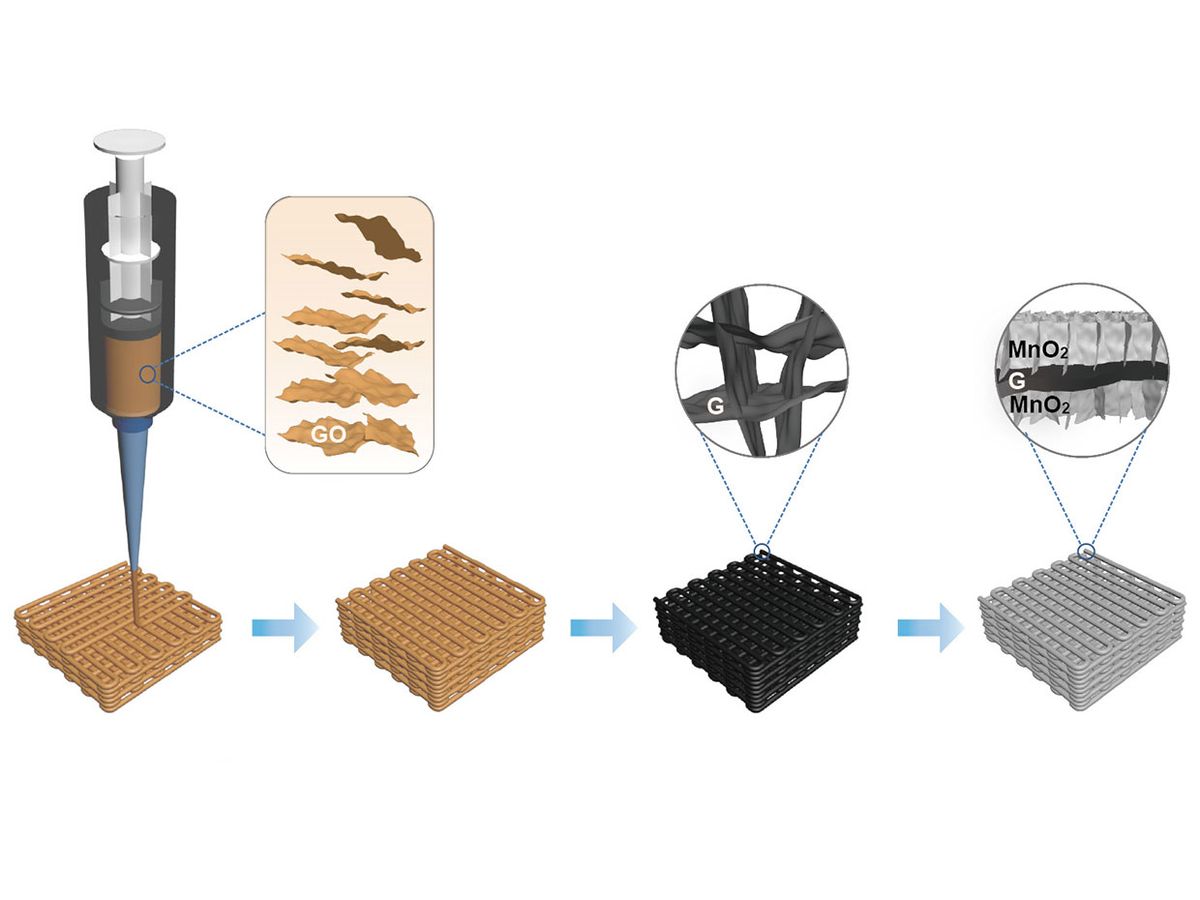
The 3D-printing technique they leveraged to make the graphene electrode may have finally addressed the trade-offs between the gravimetric (weight), areal (surface area), and volumetric (total volume) capacitance of supercapacitor electrodes that were previously thought to be unavoidable.
In previous uses of pure graphene aerogel electrodes with high surface area, volumetric capacitance always suffered. This issue has typically been exacerbated with 3D-printed graphene aerogel electrodes; volumetric capacitance was reduced even further because of the periodic large pores between the printed filaments.
This work demonstrates for the first time that a 3D-printing technique could sidestep that conundrum, says Yat Li, a professor at UCSC, and coauthor of the paper in the journal Joule that describes the team’s research. According to Li, this is the first time that high mass loading of active materials over 180 milligrams per square centimeter has been achieved without sacrificing their electrochemical performance.
“We have successfully addressed low volumetric capacitance in 3D-printed graphene aerogel electrodes by ‘filling’ more nanomaterials into this porous graphene scaffold and increasing their densities,” said Li. “Our 3D-printed graphene aerogel/MnO2 electrodes have achieved high volumetric capacitances, hundreds higher than the pure graphene aerogel electrodes.”
These mass loadings they achieved are around two or three orders of magnitude higher than the previous reports in academia, according to Li. These results may prove critical to the future of energy-storage devices. Heretofore, most materials lost a majority of their energy-storage ability when the mass loadings were increased due to the insufficient ion diffusion within the electrodes. This work also validates the concept of “printing” supercapacitor electrodes, leading to a revolution in the fabrication process of supercapacitors.
This latest research comes after a steady stream of such stories a few years back in which graphene-enabled supercapacitors were reaching ever-higher figures for energy density (the amount of energy stored per unit mass). In 2015, this seemed to reach a climax when South Korean researchers claimed to have achieved an energy density of 131 watt-hours per kilogram for a supercapacitor equipped with graphene electrodes. At the time, state-of-the-art supercapacitors using other materials for their electrodes were averaging 28 Wh/kg. While 131 Wh/kg was more than a quadrupling of the average energy density for supercapacitors, it still fell well short of the 200-Wh/kg average for lithium-ion batteries.
This energy-density race in graphene-based supercapacitors developed in part because some believed that since supercapacitors could be recharged so much more quickly than Li-ion batteries, they might make for a suitable alternative for powering electric vehicles. The reasoning went that if supercapacitors could replace Li-ion batteries, it would make driving an EV much like driving a gasoline-powered car: You go to a charging station and, in the time it would have taken you to fill up your car with gasoline, you could fully recharge your EV. While that aim has not disappeared, many are finding it hard to see how supercapacitors could ever fit the bill.
Initially, the energy density numbers measured for these latest 3D-printed graphene aerogel electrodes may not change skeptics’ outlook on the prospects for supercapacitors in EVs. Li said that the UCSC/LLNL team’s measurements demonstrated that these new electrodes could deliver a maximum energy density of 4.3 Wh/kg at a power density of 0.55 W/kg and a maximum power density of 89.4 W/kg at an energy density of 1.3 Wh/kg.
These numbers might seem significantly lower than some of the numbers that were reported a few years back for graphene-based supercapactors. However, Li notes the measurements of their device were taken at mass loadings of 182.2 mg/cm2. In contrast, the higher energy densities from previous devices were taken under extreme low active material loadings (below 1 mg/cm2), a level at which electron transportation and ion diffusion are much more efficient than those electrodes at higher mass loadings. These factors put these latest supercapactiors squarely in the middle of the pack for energy densities of current state-of-the-art supercapacitor devices, according to Li.
“These supercapacitors with high areal energy density are very promising for those applications with limited available surface area, for example, the wearable energy-storage devices because of the limited total surface area of human body,” said Li. “These supercapacitors also have the potential to be applied to electric vehicles because of their impressive electrochemical performance even at superhigh mass loadings.”
Li notes that the electrodes they have fabricated thus far are symmetric, meaning the electrodes are made from the same material. When asymmetric supercapacitors are fabricated using different materials for the two electrodes, energy densities can be further improved, leading to higher working voltages and energy densities.
While lab results in any battery storage technology can be promising, the engineering of commercial battery technology is complicated. What is promising in the lab could lead to an engineering nightmare. Li concedes that these 3D graphene aerogels will pose their own set of challenges for commercial adoption.
First, mass production of these electrodes would consume a lot of graphene or graphene oxide materials, which currently are not widely available worldwide. Second, the high price of graphene and graphene oxide nanosheets with few layers impede their wide application in industry.
But Li notes that the 3D-printing method offers a new and easier way to fabricate supercapacitor electrodes with designed shapes, which could satisfy special applications with specific requirements.
After having demonstrated symmetric supercapacitors, Li says the next step will be to fabricate asymmetric supercapacitors. At present, the researchers have been able to fabricate only the symmetric supercapacitor devices at these extreme high-mass loadings because they haven’t been able to find an appropriate anode material that can match the ultrahigh areal capacitance of their 3D-printed graphene cathode materials.
Li adds that the team is continuing this research by looking for likely candidates.
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.