Grooves in the kind of hydrogen fuel cells most likely to power vehicles may boost the performance of the devices by up to 50 percent, a new study finds.
A fuel cell converts the chemical energy stored in fuels such as hydrogen into electricity. It works by reacting the fuel with oxygen or another oxidizing agent that can strip electrons from the fuel. Fuel cells have two electrodes—an anode, where the fuel is oxidized, or loses electrons, and the cathode, where the oxidizing agent is reduced, or gains electrons.
Fuel cells that run off hydrogen, the most common element in the universe, hold great promise as clean, efficient sources of energy. When hydrogen reacts with oxygen in fuel cells to generate electricity, instead of yielding pollutants as fossil fuels do, the result is simply water.
“It’s not just the materials you put in the electrode that matter—it’s also how you structure the materials at the microscale.”
—Jacob Spendelow, Los Alamos National Laboratory
The best candidate fuel cells for powering automobiles are polymer electrolyte membrane (PEM) fuel cells, also called proton exchange membrane fuel cells, according to the U.S. Department of Energy. These use a proton-conducting polymer membrane as the electrolyte interposed between the fuel and oxidizer to help shuttle electric charge within the fuel cell.
Heavy-duty trucking provides especially promising opportunities for PEM fuel cells, researchers say in the new study. Using batteries to power these vehicles is relatively difficult, since they require long range, fast refueling, low weight, and small size.
Now scientists have found that adding grooves to PEM fuel cells can improve the performance of these devices by up to 50 percent compared with state-of-the-art conventional electrodes under standard operating conditions.
“I find it surprising that the structure and the arrangement of the electrode can have such a strong effect, enabling high performance and durability with a conventional commercially available catalyst,” says study senior author Jacob Spendelow, a chemical engineer at Los Alamos National Laboratory, in New Mexico. “It shows that it’s not just the materials you put in the electrode that matter—it’s also how you structure the materials at the microscale.”
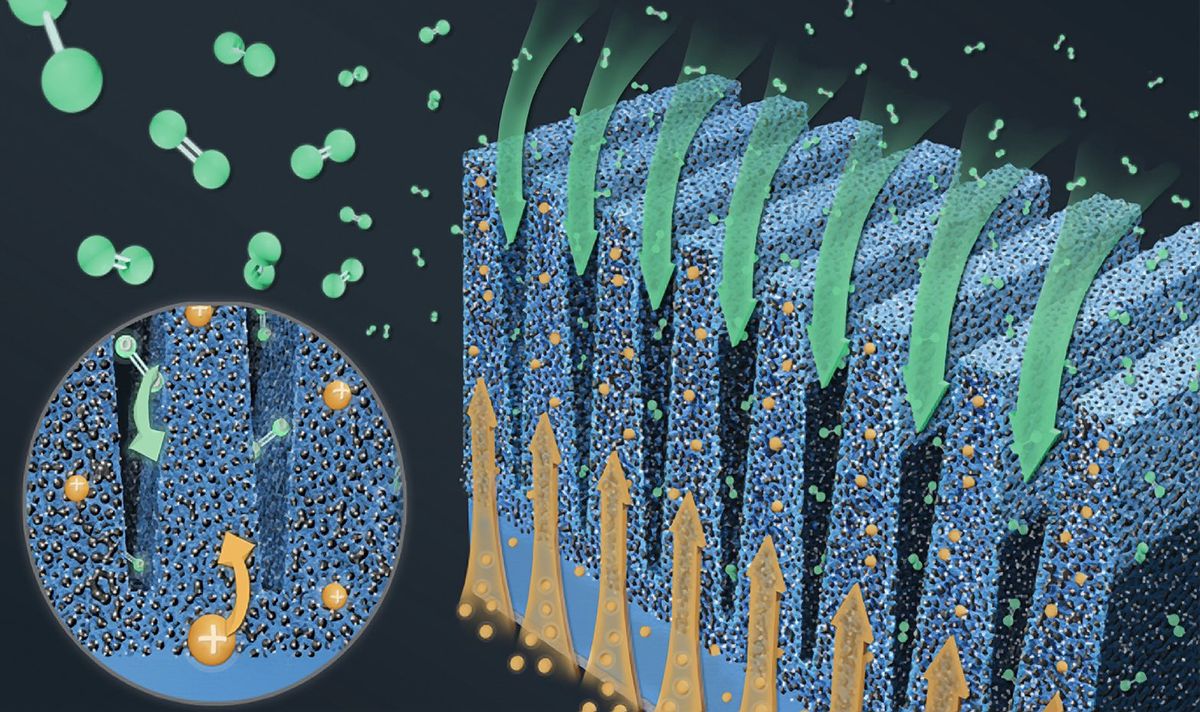
Conventional PEM fuel cell electrodes are composed of a carbon-supported platinum catalyst and ion-conducting polymers known as ionomers, which are mixed in an ink slurry and deposited on a membrane or other structure as a porous electrode. This creates a random electrode structure with a complex, mazelike network of narrow pores that limits the flow within the fuel cell. This structure has largely stayed the same for more than 30 years, the researchers note.
By separating proton and oxygen flow along grooves and ridges, the new electrodes help improve the transport of both.
In contrast, the new electrodes feature catalyst ridges loaded with ionomers separated by empty grooves. The ridges improve proton transport, while the grooves simultaneously help oxygen flow.
In conventional PEM fuel cell electrodes, a high ionomer content may enhance proton transport, but it typically also limits oxygen flow. By separating proton and oxygen flow along grooves and ridges, the new electrodes help improve the transport of both. This also helps keep reaction rates uniform in the fuel cell, boosting catalyst performance.
The scientists fabricated the new electrodes by depositing a mixture of carbon-supported platinum catalyst and ionomer in a patterned silicon template, followed by a transfer to a membrane made of the common polymer electrolyte known as Nafion. They experimented with electrodes with grooves 1 to 2 micrometers wide, and had grooves repeat across the electrodes every 3 to 6 µm. The narrower the grooves and the shorter the distances between them, the better the performance.
“We came up with the basic concept of grooved electrode design about four years ago, but figuring out how to make the electrodes, and make them work, involved a lot of effort,” Spendelow says. He notes the microfabrication skills of study lead author ChungHyuk Lee at Toronto Metropolitan University, in Canada, “were the key factor that that enabled us to transform our ideas into reality.”
The best-performing grooved electrodes were also significantly more durable than regular electrodes, displaying 170 percent higher current density after 500 cycles of activity. The researchers note that pores in conventional electrodes collapse over time due to corrosion, hampering their performance. By contrast, the grooved electrode structures remained relatively intact.
In addition, machine-learning analysis suggested there was a lot of potential to further optimize grooved electrodes. For instance, the scientists found making the grooves wider and deeper could further boost performance by 36 percent compared with the best grooved electrodes they tested, although better fabrication methods are needed to confirm these findings.
“The critical next step is to perform manufacturing R&D to develop scalable production of grooved electrodes,” Spendelow says. “From an academic perspective, there are many aspects of the grooved electrode design that we’d like to continue exploring, but from a practical perspective, demonstrating manufacturability is key.”
The scientists detailed their findings 25 May in the journal Nature Energy.
- Filling Up a Fuel-Cell Car With Hydrogen Gets One Step Closer ... ›
- 'Hydrogen-On-Tap' Device Turns Trucks Into Fuel-Efficient Vehicles ... ›
- Perovskites Key to New Type of Hydrogen Fuel Cell ›
- Tiny Superlens Refocuses the Solar-to-Hydrogen Game - IEEE Spectrum ›
- Daimler Truck Fuels Big Rigs With Hydrogen - IEEE Spectrum ›
Charles Q. Choi is a science reporter who contributes regularly to IEEE Spectrum. He has written for Scientific American, The New York Times, Wired, and Science, among others.