Here’s a futuristic problem that may not have occurred to you: If self-driving cars really catch on and the number of traffic fatalities plunges, so will the number of organs available for transplant. Currently, about 20 percent of donated organs come from people who die in car accidents.
Luckily, there’s a futuristic solution: 3D-printed organs. This technology is far from ready for the clinic, as researchers are still trying to figure out how to print out complex tissue structures with blood vessels and nerves. But for one early indicator of progress in this field, look to the eye.
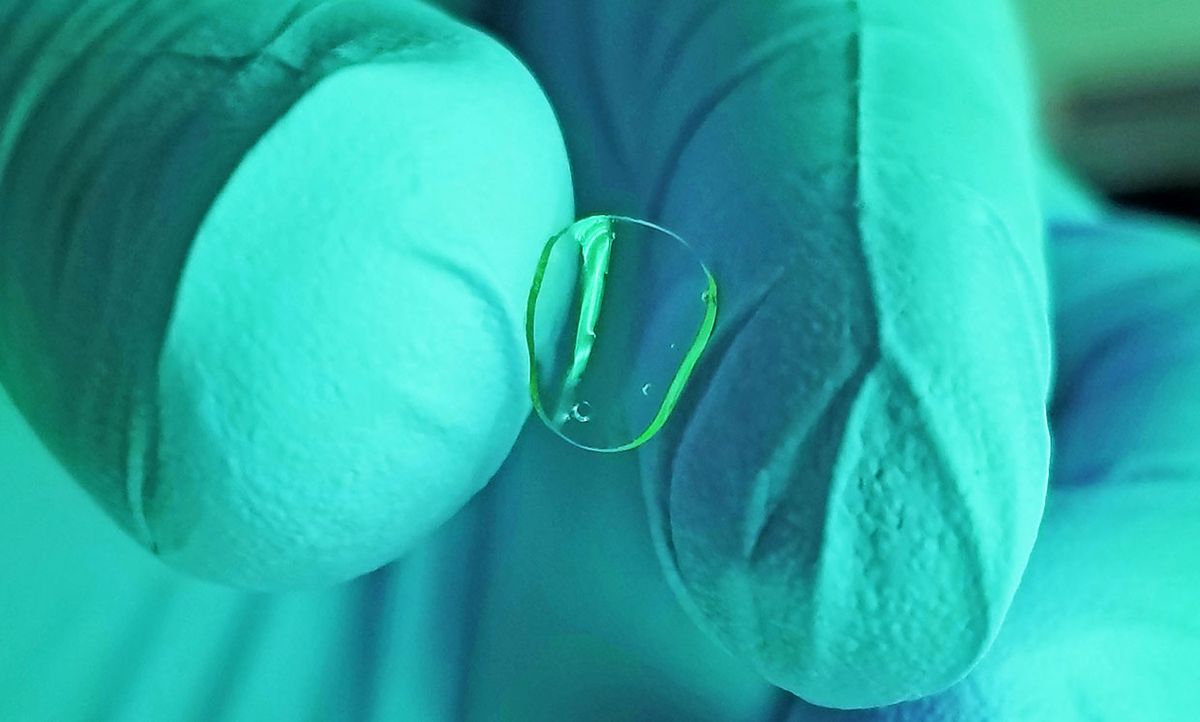
Precise Bio, a North Carolina-based startup founded by several professors at the renowned Wake Forest Institute for Regenerative Medicine, is working on bioprinting tissues for a variety of medical applications. The company just announced that its first products will be for the eye—starting with a human cornea suitable for transplantation. “We plan to put our printers in eye banks,” says Precise Bio CEO Aryeh Batt.
Bioprinters are a specialized type of 3D printers. Instead of putting down layers of plastic or metal to gradually build a structure, they put down layers of cells and biocompatible materials to build tissue.

In an exclusive interview with IEEE Spectrum, Batt says that his company is aiming high, and wants to eventually overhaul the whole organ transplant system. “Physicians can use tissues that are printed, not taken from donors,” he says. The company’s bioprinters could fabricate tissues with consistent specs, or they could print custom tissues to meet a patient’s needs. “It’s a manufacturing process,” says Batt. Either way, it would be a departure from today’s morbid luck-of-the-draw system.
Bioprinting is a hot area of research, with great promise in applications such as printing skin for burn victims and printing bone and cartilage for orthopedic repairs. But these research projects haven’t yet turned into real treatments for human patients.
Corneas could be the first mainstream application of bioprinting, Batt says, in part because they have a layered structure that’s a good match for the technology. Each layer consists of different types of cells and fibers, which the printer could lay down in sequence, and these layers don’t contain blood vessels or nerves. What’s more, putting a new kind of transplant in the eye is inherently safer than implanting one deep in the body, since physicians could easily check for signs of trouble and could remove the tissue if anything seemed wrong.
There’s certainly a need for more corneas in the world, says Kevin Corcoran, president and CEO of the Eye Bank Association of America. In 2017, his members supplied nearly 51,000 transplantable corneas to patients in the United States, and also sent more than 26,000 abroad. Internationally, “there is a tremendous amount of unmet demand,” he says. “It’s estimated that 10 million people suffer from corneal blindness globally, primarily because they lack access to effective and affordable treatment.”
Part of Precise Bio’s proprietary approach is its printer, which uses a technique called laser-induced forward transfer to propel droplets of bioinks onto a surface. Just as a desktop printer has cartridges containing different colors of inks, the Precise Bio printer has cartridges containing different biological materials such as epithelial cells and collagen.
The role of the laser is to focus energy on material inside the printer head, causing tiny bubbles to form and then pop, thus propelling tiny jets of bioink onto the substrate. Batt says the laser energy isn’t applied directly to cells and therefore doesn’t damage them. Competing bioprinting methods do more cell damage, he argues: Micro-extrusion techniques damage cells by putting pressure on them to force them through an aperture, and inkjet techniques can damage cells by heating them up.
While bioprinting is usually considered a type of 3D printing, Precise Bio likes to refer to their process as 4D printing, emphasizing the role of the fourth dimension: time. After the printer fabricates a piece of tissue, that tissue goes into a bioreactor for maturation. For bioprinted corneas, that means spending 10 to 14 days in a warm medium where the cells and fibers knit themselves together. “The cells connect to each other and form the same morphology that you have in human tissue,” Batt says. “Essentially, the biology does the work, but you have to put them in the correct environment to make it happen.”
Precise Bio has already done its first safety studies in animals, and is working toward a first-in-human trial for its printed corneas. The company will have to answer a lot of questions about safety, however, before getting to that stage.
For example, it will have to prove that the bioprinted corneas will behave like normal corneas once implanted in a human eye. In normal corneas, the endothelial cells don’t divide and multiply. But in the Precise Bio maturation process, the printed endothelial cells are coaxed into proliferation. “Before we place it in the eye, we stop this process,” Batt says. “But it is one thing we’ll have to show in our clinical studies.”
Eliza Strickland is a senior editor at IEEE Spectrum, where she covers AI, biomedical engineering, and other topics. She holds a master’s degree in journalism from Columbia University.