Nelson Sepúlveda was sitting in the stands at Spartan Stadium, watching his hometown Michigan State players bash heads with their cross-state football rivals from the University of Michigan, when he had a scientific epiphany.
Perhaps the nanotechnologies he had been working on for years—paper-thin devices known as ferroelectret nanogenerators that convert mechanical energy into electrical energy—could help save these athletes from the ravages of traumatic brain injury.
An electrical engineer at MSU, Sepúlveda had made self-powered loudspeakers, flexible microphones, and e-textiles out of his miniature, energy-harvesting platform. Now he wanted to apply the technology to the problem of concussion monitoring.
The Spartans lost 21–7 to the Wolverines on that October afternoon in 2018, but Sepúlveda left the stadium with a winning idea.
“There is a lot of scope to piezoelectric-type sensors for measuring movement and providing feedback to people.”
—Andrew McIntosh, Monash University
“I think it’s the best application that we’ve found so far for this [technology],” he says.
Existing concussion safety protocols dictate that players involved in on-field collisions must undergo medical evaluation for symptoms of head trauma before returning to action, but this system is easily manipulated—and it overlooks many of the subconcussive blows that can collectively cause long-term effects on the brain.
Sensor-laden helmets and mouthguards exist that gather accelerometry data for a more quantitative measure of impact forces to the head. However, helmets can slide around, and mouthguards often get chewed up. So these embedded sensors do not always offer an accurate picture of what’s happening inside the skull—or even if someone has truly had their bell rung.
Video 1www.youtube.com
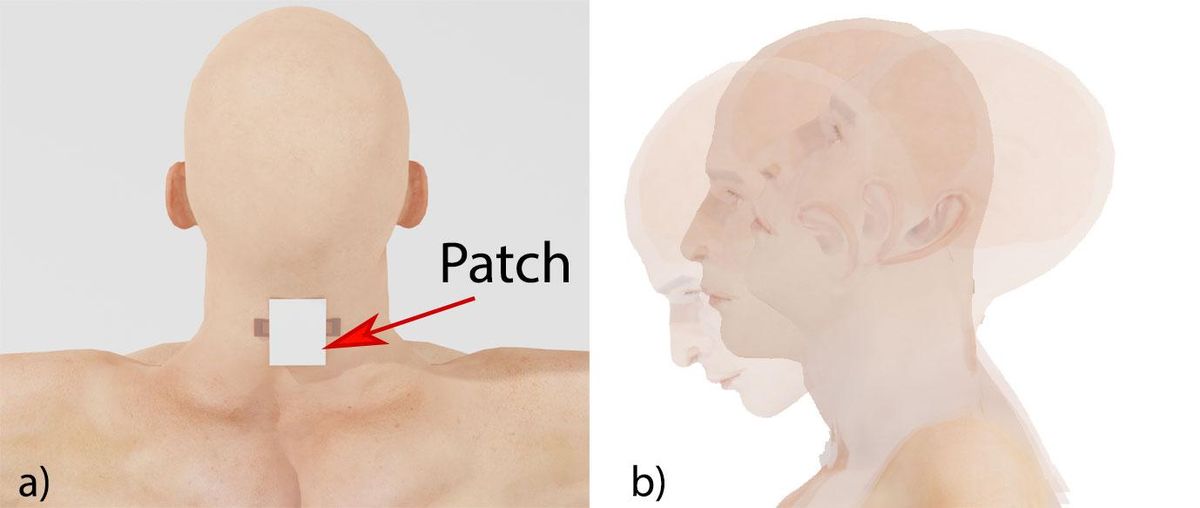
“You end up with a lot of false readings,” says Sepúlveda. A better option, he thought, would be a wearable patch applied directly to the skin around the neck.
Sepúlveda and his students kitted out crash-test dummies with four of their piezoelectric devices—one on the throat, one on the nape, and one on each side of the neck. They performed simulated whiplash experiments on the dummies, and found that the Band-Aid–size patches accurately captured indicators of brain injury.
The sensors transformed tensile strains applied to the neck into voltage pulses. And that electrical output proved highly predictive of rotational forces deep in the head, as measured by accelerometers implanted inside the dummies’ noggins.
Sepúlveda and his colleagues published their findings in the journal Scientific Reports. With an eye to commercial applications, the researchers are now adapting their sensors for wireless data transmission. They are also encapsulating the devices in protective materials that shield the technology against sweat and other grime. Trials in human athletes should follow.
Sepúlveda points to several potential benefits of his neck-sensor system over the kinds of intelligent helmet and mouthguard technologies that the National Football League and other sports associations are beginning to adopt.
“When you separate your measurement from the location of impact, as they are doing, you introduce a lot of potential error.”
—Jake Merrell, XOnano Smartfoam
Because Sepúlveda’s devices are self-powered, there are no batteries to swap out or recharge. Some smart mouthguards, by comparison, require charging after every game. His patches are cheap to manufacture, which should make for a more cost-competitive product. (Sepúlveda envisions a retail price well below the US $99-plus of other sensor-backed wearables.) The patches should also be usable for high-impact sports for which protective head gear and mouthpieces are not routinely worn. The technology might therefore help expand the reach of real-time concussion detection into soccer, cheerleading, and any number of other activities.
Sepúlveda does acknowledge one limitation of his design, however. The patches do not capture the type of head-on-head injuries that directly compress the neck without twisting it—think rugby scrums or collisions between opposing linemen in a football game. “But any other angle of impact,” Sepúlveda says, “we’ll get it.”
Still, not every concussion researcher is convinced that wearable neck sensors are the best path forward.
“I agree with their conclusion that measuring acceleration in the helmet is inherently noisy and prone to error. However, I don’t see their neck sensor doing much better,” says Jake Merrell, president and lead engineer at XOnano Smartfoam, a company that is developing piezoelectric padding for smart helmets. “When you separate your measurement from the location of impact, as they are doing, you introduce a lot of potential error.”
That’s why Barclay Morrison, who studies the biomechanics of brain injuries at Columbia University, continues to prefer high-tech mouthguards for the collection of information on impact speed, direction, force, location, and severity.
“Because the upper mandible is the same bone as the skull,” he says, “the data more accurately represents skull acceleration.”
Sepúlveda and his team put that idea to the test, however, and found it rang hollow. In unpublished experiments, they tried placing their nanogenerators inside the dummies’ mouths, as well as up their noses. According to Sepúlveda, no other patch location proved as accurate as the neck.
A coalition of experts, known as the Consensus Head Acceleration Measurement Practices (CHAMP) group, is currently developing best practices for how the developers of impact sensor systems should gather, report, and analyze head-acceleration data in sports. Andrew McIntosh is an Australian expert in impact injury biomechanics and a member of that group.
In his view, “there is a lot of scope to piezoelectric-type sensors for measuring movement and providing feedback to people.” But he is reserving judgement on the MSU system until data from human subjects become available.
“It’s in a very early phase,” he says.
McIntosh sees the technology’s potential, though—including in the diagnosis and prevention of sports injuries generally, not just those affecting the head. Consider knee ligament injuries, for example. Many athletes experience sprains or tears as a result of repetitive stress to the knee joint, and the MSU patches could help these individuals monitor for early signs of overuse strain or indicators of injury-inducing form.
“So, I wouldn’t think of it only as an alternative way to measure head kinematics,” McIntosh says. “I would be thinking of it as a general sensor for...many practical situations during sporting training and injury prevention.”
- Quanterix's Magnetic Tech for Fighting Football Concussions - IEEE ... ›
- Using VR to Diagnose Concussions - IEEE Spectrum ›
Elie Dolgin is a science writer specializing in biomedical research and drug discovery. After a PhD spent studying the population genetics of nematodes, he swapped worms for words—entering journalism as an editor at The Scientist, Nature Medicine, and STAT. Now a freelancer, Elie is a frequent contributor to New Scientist, Nature, IEEE Spectrum, and more.