With just one week under our belts in this New Year, we already have some world-record news in relation to lithium-ion (Li-ion) batteries and nanoparticles. Researchers at Stanford University and the SLAC National Accelerator Laboratory have developed a Li-ion battery in which its sulfur cathode was capable of storing five times more energy than is possible with today’s commercially available batteries.
The research—not surprisingly—was led by Stanford’s Yi Cui. What may be somewhat surprising is that Cui has focused his attention in this research on the cathode rather than the anode of the battery. Much of Cui’s most recent work has been on improving the anodes of Li-ion batteries through the use of nanostructured silicon. In this latest research, he has not only shifted his attention to the cathode, but also developed an entirely new material to do it.
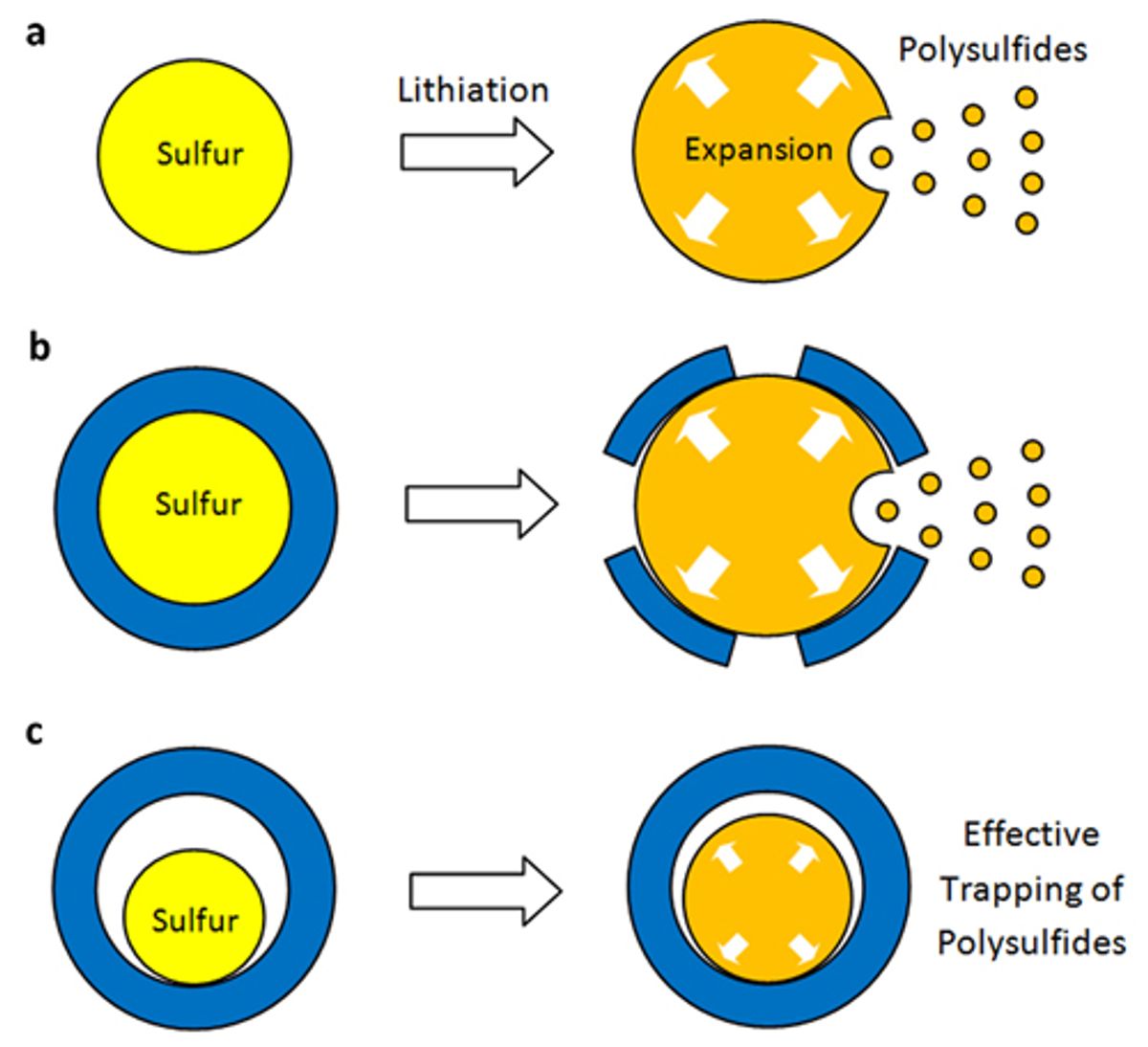
The new material, which is described in the Jan. 8 edition of Nature Communications (“Sulphur–TiO2 yolk–shell nanoarchitecture with internal void space for long-cycle lithium–sulphur batteries”), is a nanoparticle that is made up of an inner core of sulfur surrounded by an outer layer of porous titanium-oxide. The nanoparticles architecture resembles that of the yolk and shell of an egg.
While it has been known that sulfur could store more lithium ions than other cathode materials, the combination of sulfur atoms with lithium ions resulted in a compound—though necessary for the cathode to operate—that kept dissolving and limited the storage capacity of the battery. Also, when the lithium ions went into the cathode, it would expand the size of the cathode by 80 percent. Attempts to employ protective coatings to correct the first problem of the compound dissolving just resulted in them cracking as soon the lithium ions expanded the cathode.
In the new architecture that Cui has developed, there is space between the sulfur and the hard—but porous—titanium oxide. What this means is that the sulfur-lithium intermediate compound does not dissolve because it is protected by hard outer surface of the nanoparticle and the sulfur inside has enough room to expand in the space between the core and the outer shell.
“It basically worked the first time we tried it,” Cui says in a press release. “The sulfur cathode stored up to five times more energy per sulfur weight than today’s commercial materials."
“After 1,000 charge/discharge cycles, our yolk-shell sulfur cathode had retained about 70 percent of its energy-storage capacity. This is the highest performing sulfur cathode in the world, as far as we know,” he said. “Even without optimizing the design, this cathode cycle life is already on par with commercial performance. This is a very important achievement for the future of rechargeable batteries.”
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.