When the World Economic Forum published its Global Risks Report this year, it identified the number one greatest risk facing the world as the looming water shortage. While there are both new and old technology solutions for desalinating water that could address this water shortfall, they have remained highly energy intensive, rendering many of them out of reach for most regions outside of the oil-rich Persian Gulf.
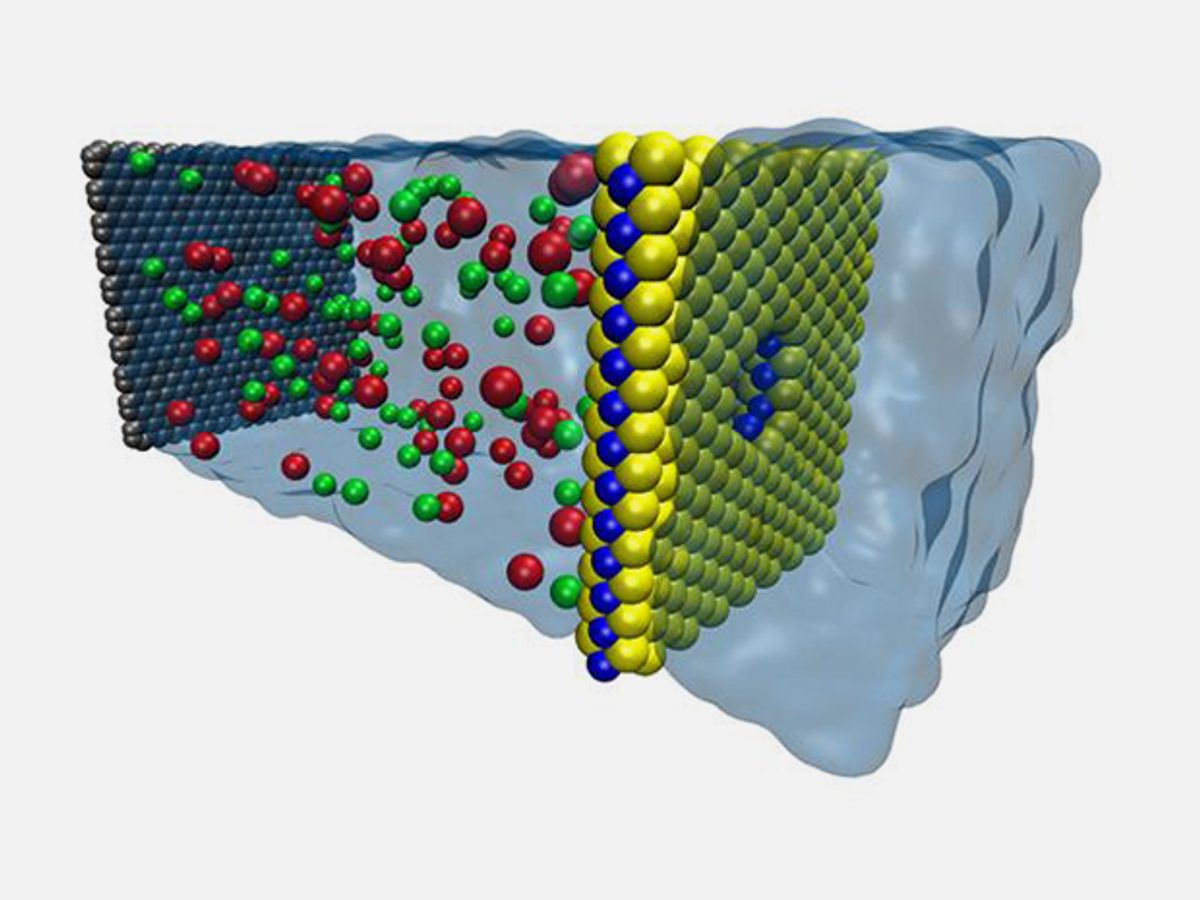
Scientists have been looking to graphene in the search for ways of easing the energy demands of water desalination. Here the material acts a porous membrane that allows water through but blocks the flow of salt ions—a pressure-driven process called reverse osmosis. Researchers at the University of Illinois recently took a look at that material’s two-dimensional cousin molybdenum disulfide (MoS2) in that role and believe that it may remove salt much better.
In research published in the journal Nature Communications, the Illinois scientists modeled various thin-film membrane materials and found that MoS2 was the most efficient, filtering up to 70 percent more water than graphene membranes.
"Finding materials for efficient desalination has been a big issue, and I think this work lays the foundation for next-generation materials,” said Narayana Aluru, a professor at the university and leader of the research, in a press release. “These materials are efficient in terms of energy usage and fouling, which are issues that have plagued desalination technology for a long time."
For any new material to take its place as a reverse osmosis membrane it must have pores of a precisely-controlled size; a resistance to fouling and clogging; and significantly reduce energy costs.
"Reverse osmosis is a very expensive process," Aluru said in the press release. "It's very energy intensive. A lot of power is required to do this process, and it's not very efficient. In addition, the membranes fail because of clogging. So we'd like to make it cheaper and make the membranes more efficient so they don't fail as often. We also don't want to have to use a lot of pressure to get a high flow rate of water."
The Illinois researchers claim that their computer models demonstrate that MoS2 is the best at this because of its particular chemical and physical properties. Because MoS2 has one molybdenum atom sandwiched between two sulfur atoms, a sheet of the material is essentially a sulfur coating on the outside and molybdenum in the middle.
The researchers discovered that when a pore is created in a sheet of MoS2, a ring of molybdenum is forms around the center of the pore, which draws water through the pore due to molybdenum’s ability to attract water.
"MoS2 has inherent advantages in that the molybdenum in the center attracts water, then the sulfur on the other side pushes it away, so we have much higher rate of water going through the pore," said graduate student Mohammad Heiranian, the first author of the study. "It's inherent in the chemistry of MoS2 and the geometry of the pore, so we don't have to functionalize the pore, which is a very complex process with graphene."
While it is typically a big step to go from computer models to producing actual devices of any kind, the researchers seem fairly confident that they are on to something. They have already established collaborations to conduct physical experiments to test MoS2 for water desalination applications.
(For an explanation of the major desalination technologies see “The Saline Solution” IEEE Spectrum June 2010.)
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.