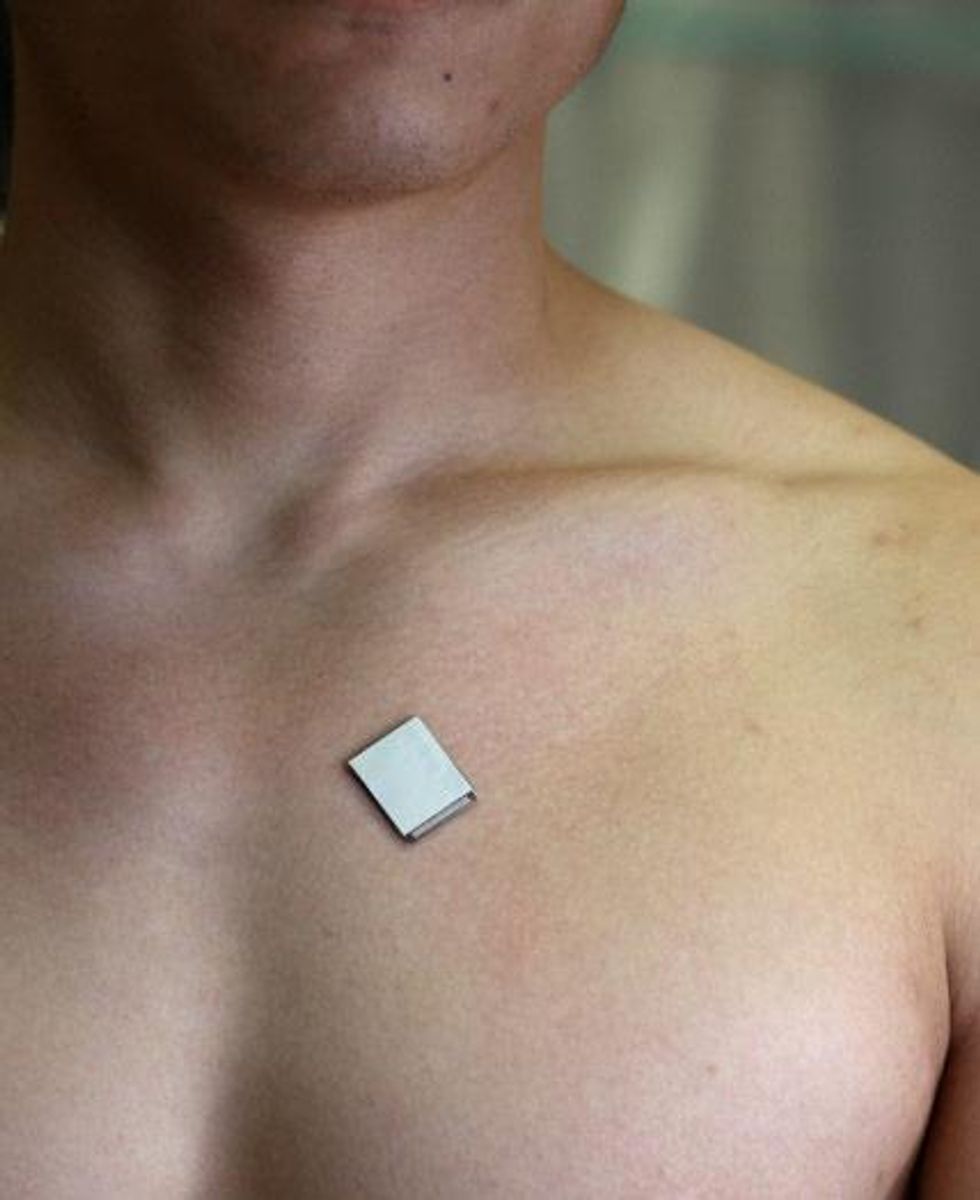
A wearable ultrasound imager for the heart that is roughly the size of a postage stamp, can be worn for up to 24 hours, and works even during exercise may one day help doctors spot cardiac problems that current medical technology might miss, a new study finds.
Heart disease is the leading cause of death among the elderly, and is increasingly becoming a problem among those who are younger as well because of unhealthy diets and other factors. The signs of heart disease are often brief and unpredictable, so long-term cardiac imaging may help spot heart anomalies that might otherwise escape detection.
For instance, patients with heart failure may at times seem fine at rest, “as the heart sacrifices its efficiency to maintain the same cardiac output,” says study colead author Hongjie Hu, a nanoengineer at the University of California, San Diego. “Pushing the heart towards its limits during exercise can make the lack of efficiency become apparent.”
In addition, the heart can quickly recover from problems it may experience during exercise. This means doctors may fail to detect these issues, since cardiac imaging conventionally happens after exercise, not during it, Hu says.
However, cardiac imaging techniques are often limited in terms of when they can analyze the heart, due in large part to the bulkiness of the equipment. For example, traditional cardiac ultrasound imaging “evaluates images of the heart right before and right after intensive exercise, as holding an ultrasound probe over the chest by hand and maintaining a stable position for it is impossible during this process,” Hu says. Previous wearable heart sensors could capture signals only on the skin.
Now scientists have developed a wearableultrasound device that can enable safe, continuous, real-time, long-term, and highly detailed imaging of the heart. They detailed their findings online on 25 January in the journal Nature.
“Potential applications include continuously monitoring the heart in daily life, during exercise, during surgery, and much more,” says study coauthor Ray Wu, a nanoengineer at UC San Diego. “This will open up the possibility to detect previously undetectable symptoms of disease, identify symptoms in their very early stages, and greatly improve patient outcomes.”
The new device is a patch 1.9 centimeters long by 2.2 cm wide and only 0.9 millimeters thick. It uses an array of piezoelectric transducers to send and receive ultrasound waves in order to generate a constant stream of images of the structure and function of the heart. The researchers were able to get such images even during exercise on a stationary bike. No skin irritation or allergy was seen after 24 hours of continuous wear.
“The most exciting result is that our patch performs well when an individual is moving,” Hu says. “Our patch allows us to evaluate heart performance throughout exercise, providing valuable information about the heart when it is under high stress.”
The new patch is about as flexible as human skin. It can also stretch up to 110 percent of its size, which means it can survive far more strain than typically experienced on human skin. These features help it stick onto the body, something not possible with the rigid equipment often used for cardiac imaging.
“The wearable imager removes much of the bulkiness of traditional imagers and adheres to the body on its own,” Wu says. “This enables the heart to be imaged in ways and scenarios that were never possible before while also freeing up the hands of the doctor from having to continuously operate a traditional ultrasound probe.”
In the new study, the researchers focused on imaging the left ventricle, the largest of the heart’s four chambers “and strongly considered to be the most important in terms of cardiovascular health, as it is responsible for pumping oxygenated blood to the entire body,” Wu says. Cardiac imaging generally focuses on the left ventricle, but the new device can image all of the heart’s four chambers simultaneously, “so it may be possible for future research to focus on other or multiple chambers,” he adds. In addition, “the imager can be applied to image various other organs, such as the stomach, kidney, or liver.”
Traditional cardiac ultrasound imaging constantly rotates an ultrasound probe to analyze the heart in multiple dimensions. To eliminate the need for this rotation, the array of ultrasound sensors and emitters in the new device is shaped like a cross so that ultrasonic waves can travel at right angles to each other.
The scientists developed a custom deep-learning AI model that can analyze the data from the patch and automatically and continuously estimate vital details, such as the percentage of blood pumped out of the left ventricle with each beat, and the volume of blood the heart pumps out with each beat and every minute. The root of most heart problems is the heart not pumping enough blood, issues that often manifest only when the body is moving, the researchers note.
The scientists did not begin investigating heart imaging until after they developed a wearable ultrasound device.
“After learning in a casual chat that one of our colleague’s relatives had died of a sudden heart attack, we realized our technology could make real impacts on people’s lives,” says study colead author Hao Huang, a nanoengineer at UC San Diego. “We did not expect the imaging quality of the imager to be very good. However, it turned out we could achieve a comparable quality to commercial probes after careful engineering. We knew we were onto something big when we saw the images full of details after many rounds of improvement, and we thought we should keep pursuing this path to not waste the capabilities of the imager.”
In the beginning, “we were always struggling with focusing of the ultrasound beam,” Huang recalls. “Debugging involved intensive trial and error based on the readings. After many failures, we used an automatic alignment strategy to make the transducer array with a high uniformity and perfect alignment so that it guarantees beam focusing.”
Another set of problems involved noise in the signals. “We did lots of trial and error to identify all the sources of noise and isolate the imager from all of them but were still left with the most major one, the human body,” Huang says. They ultimately developed a stretchable electromagnetic shield made of the same liquid metal used in the device’s electrodes “to remove the noise from the human body while maintaining the flexibility and stretchability of the imager. It turned out that the optimized shielding layer worked very well, and we could finally remove the noise from the images.”
The researchers note that right now, the device is connected through cables to a computer. They have developed a wireless version of the patch, details of which are in a study currently under review, Hu says.
The study’s senior author, Sheng Xu, a professor at UC San Diego, plans to commercialize this technology through Softsonics, a company cofounded with another of the study’s coauthors, Shu Xiang. Further work is needed to develop this device into a fully integrated wearable system with a built-in power source, Hu says.
Charles Q. Choi is a science reporter who contributes regularly to IEEE Spectrum. He has written for Scientific American, The New York Times, Wired, and Science, among others.



