Over two decades ago, researchers at Drexel University identified a new kind of material they dubbed Max phase (the M is for transition metal, the A for "A group" metal, and the X for carbon and/or nitrogen). At the time, the scientists believed the material could serve as a kind of primordial goo from which all things came—it contained all the elements but needed to be organized by scientists.
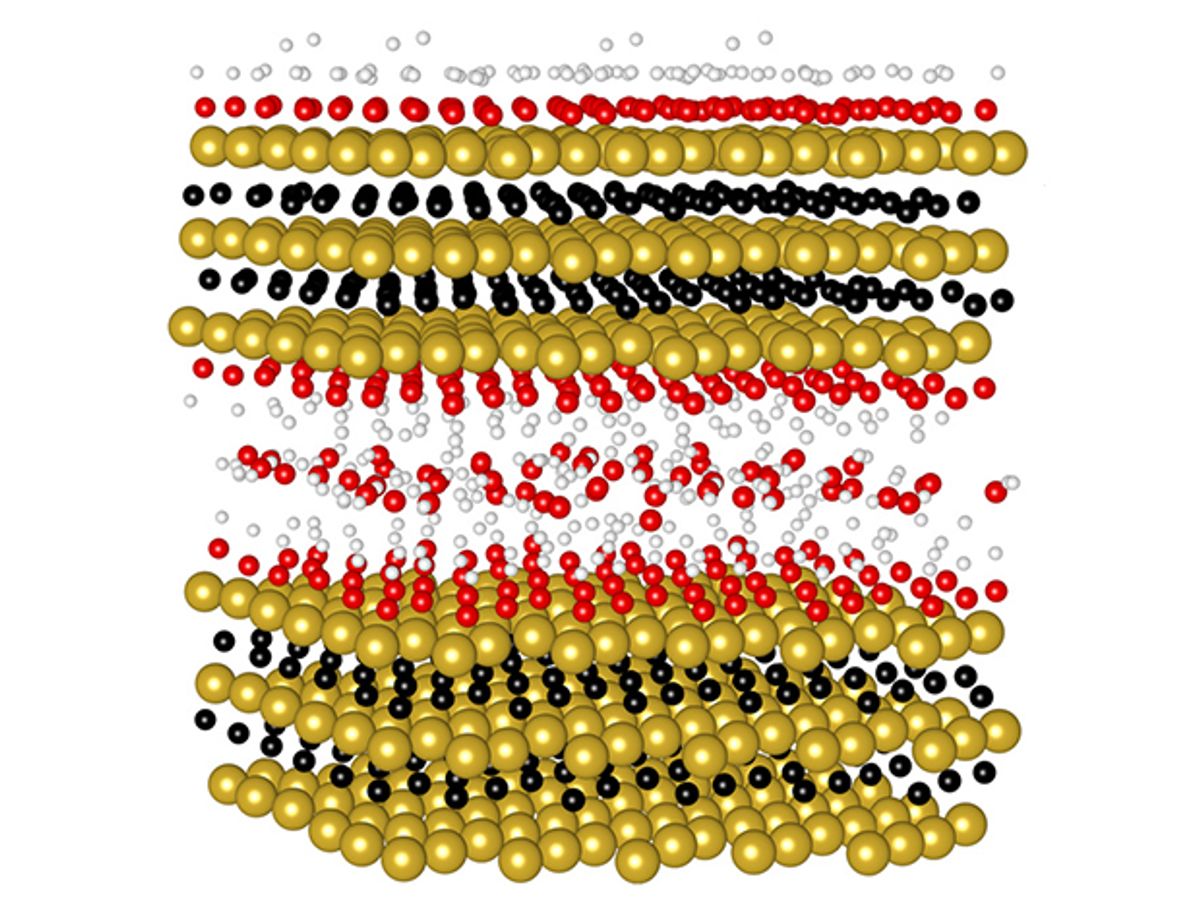
Since 2011, Drexel researchers have been working with an iteration of this Max phase goo called “MXene.” It was essentially a two-dimensional (2D) material that derived its name from the process of etching and exfoliating atomically thin layers of aluminum from MAX phases. In 2013, the Drexel researchers first discovered that one of the dozens of MXene materials they were working with could serve as a material for energy storage.
Now in research described in the journal Nature Energy, the Drexel scientists in collaboration with researchers at the Université Paul Sabatier in Toulouse, France, have used an earlier reported MXene material made from titanium and molybdenum carbide and applied a number of improvements to it to reach new charging rate horizons for pseudocapacitive materials with millisecond charging times.
Unlike in the other varieties of supercapacitor such as electric double layer capacitors, in pseudocapacitors charge is stored via a chemical mechanism, like in batteries. Pseudocapacitors sit somewhere in between batteries and EDLCs, fully charging in anywhere from tens of seconds to several minutes. Their main advantage is that they can store as much charge as some batteries, while operating much faster.
The Drexel researchers have shrunk the charging times for pseudocapacitors from the tens of seconds down to milliseconds.
“The key to the faster charging times was the electrode architecture, which allowed easy access of ions on the surface of MXene sheets,” said Maria Lukatskaya, a PhD student at Drexel and co-author of the research, in an e-mail interview with IEEE Spectrum. “Also, in this work we demonstrate an extended operational voltage window which leads to a higher energy density.”
The researchers haven’t provided the energy density values as of yet since because such metrics are characteristics of a device, not a material. The researchers concede that the energy density of a supercapacitor—even pseudocapacitor using a battery-like charge storage—is going to be lower than that of a Li-ion battery.
However, if you want to have energy storage devices that allow today’s numerous electronic gadgets to be charged quickly—in seconds to minutes, instead of hours—pseudocapacitors could offer this opportunity.
“The key value of this work is in the demonstration that pseudocapacitive charge storage can be achieved at the same high rates as in double-layer capacitors using physical charge storage (but with 5-10 times more charge stored per unit of weight or volume), as long as the electrode material offers sufficient electronic and ionic conductivity,” Yury Gogotsi, whose lab led the research, said in an e-mail interview with IEEE Spectrum. “We believe this is a game-changing development in the energy storage field.”
In order for this work to become a real-world energy storage device, Gogotsi believes that they will initially need to scale-up material synthesis. At present, they are producing up to 100 grams per batch in their labs. After that, they will need to design energy storage devices employing MXenes and other highly conductive pseudocapacitive materials.
Gogotsi added: “We are developing matching anodes to MXene cathodes that will further expand the voltage window—this means doubling the energy density.”
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.