A scientist wanting to hack into an animal’s brain used to have three different tools to choose from: electric current, drugs, and light. Now there’s a fourth: magnetic fields. In a paper published last week in the open-access journal eLife, scientists at the University at Buffalo used magneto-thermal genetics to manipulate brain cells in mice, enabling the researchers to control the animal’s behavior.
Magneto-thermal genetics has been previously shown to activate neurons in anesthetized rodent brains, but this is the first time anyone has reported using the tool to manipulate animal behavior, says Arnd Pralle, the University at Buffalo biophysicist who led the research.
Brain hacking tools help scientists better understand the wiring of the brain—the arrangement of neural circuits and which ones control different movements and behaviors. These tools could someday lead to the development of artificial human eyes and ears, or treatments for paralysis, traumatic brain injury, and diseases such as Parkinson’s and depression.
Over the past few years, major funding agencies have encouraged scientists and bioengineers to focus their work on the body’s internal wiring. The U.S. National Institutes of Health (NIH) and DARPA have been doling out grants for work on both the peripheral and central nervous systems.
Engineers play a key role in the research. The body’s nervous systems communicate, after all, in a language of electrical signals. Researchers must not only map those signals, but also figure out how to interface with them, and override them when they malfunction.
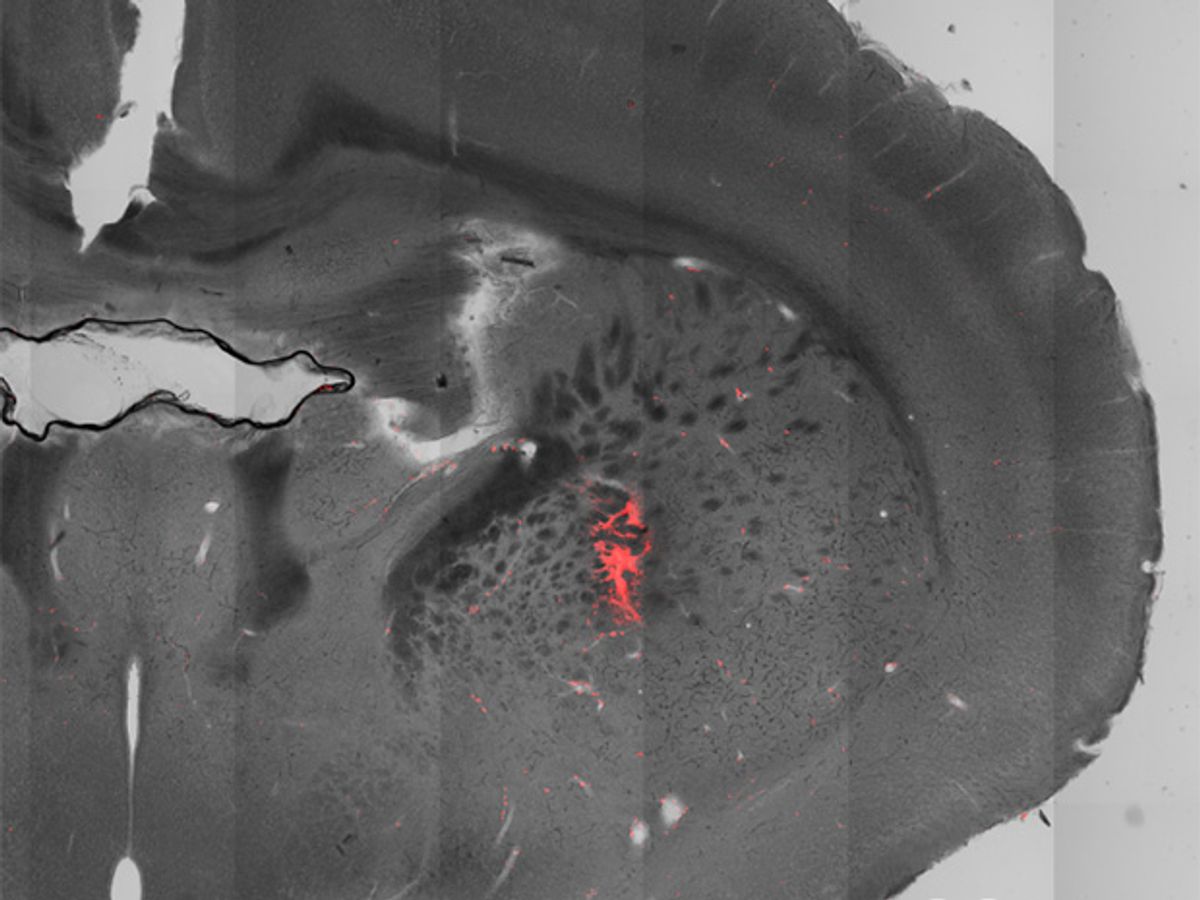
Magnetic fields can do the job (following some complicated, multi-step bioengineering). In Pralle’s experiments, he and his team injected a virus containing a gene and some helper genetic elements into the brains of mice. This genetic material gets incorporated into the DNA of the mouse’s brain cells, or neurons. The foreign gene makes the neurons heat sensitive. Next, they injected magnetic nanoparticles into a specific region of the mouse brain that latch onto the neurons in that region. They then applied alternating magnetic fields, which cause the nanoparticles to heat up a couple of degrees. The rise in temperature triggers the heat-sensitive neurons to open ion channels. Positively-charged ions flow into the neuron, causing it to fire.
Pralle demonstrated proof of the concept in 2010, and others, such as Polina Anikeeva, a professor of materials science and engineering at MIT, have since improved upon it. Those studies confirmed that the technique could indeed activate neurons in the rodent brain.
In the new study, Pralle and his team show how magneto-thermal genetics can manipulate behavior in mice that are awake and freely moving. In their experiments, they activated regions of the brain that made the mice run faster around the perimeter of their cages, spin in circles, and, eerily, freeze the motion of all four paws.
Those same behaviors have been induced in rodents by activating neurons using other brain hacking tools, including optogenetics (in which neurons are genetically sensitized to respond to light), and chemogenetics (in which neurons are genetically sensitized to respond to designer drugs).
Those three tools—magneto-thermal genetics, optogenetics, and chemogenetics—are new and purely experimental. A fourth tool—electrical stimulation—has been around for decades, with some success in treating Parkinson’s, depression, memory loss, paralysis, and epilepsy in humans.
None of the tools has made a dent, relatively speaking, in the range of functions that the brain, spinal cord and peripheral nerves control. It’s like owning four different musical instruments and knowing how to play only a few rudimentary toddler songs on each of them. That untapped potential has inspired scientists to continue to test and develop the tools.
That means overcoming the shortcomings of each tool. Electrical stimulation of deep brain regions requires, at least for now, an invasive surgical procedure to implant electrodes. That limits the number of patients willing to undergo the surgery. The method is also limited in how specifically it can target small brain regions or cell types.
Optogenetic techniques can target specific neurons, but animals in these experiments usually have to be tethered to an optical fiber or other kind of implant that delivers the light, which can affect their behavior. Study animals undergoing chemogenetic modulation can run free, but their response to the drugs is much slower than to light or electrical stimulation.
Magneto-thermal genetic tools are non-invasive, tetherless, and induce a response within seconds of turning on the magnetic fields. But there’s controversy over how the tool works.
Pralle’s team has shown that the magnetic nanoparticles injected into the mouse brains latch onto the membranes of the neurons, thus restricting the heating to those membranes rather than diffusing out to the surrounding liquid. “This makes little sense from a physics point of view, and contradicts basic principles of heat transfer,” says Markus Meister, a bioengineer at the California Institute of Technology in Pasadena.
Meister has also argued that previous experiments in magnetogenetics—a sister tool to magneto-thermal genetics that involves a different mechanism—contradict the laws of physics. He laid out his back-of-the-envelope calculations last year in a paper in eLife, which garnered a lot of attention in the field of neuromodulation.
However, Pralle’s main claim, that he successfully used magnetic heating to control animal behavior, “looks well supported,” Meister says. “Bottom line, the reported effects on behavior look real, but just what the mechanism is behind them remains to be understood.”
Pralle says his work clearly demonstrates and measures local heating at the cell membrane, showing that it does indeed occur. Why that’s happening, however, is unclear, he says. “We cannot completely explain why the increase in heat stays within a few tenths or hundredths of nanometers” of the neuronal membrane, Pralle says. The heat “should diffuse more quickly into the [surrounding] water solution, so it shouldn’t have much of a local heating effect.”
Several theorists and experimentalists, including Anikeeva, have formulated and are testing models to explain the phenomenon. “Similar effects have been seen, measured and correctly predicted for laser heating of gold nanoparticles in water,” Pralle says.
Anikeeva says she sees no controversy in Pralle’s latest work. Meister’s argument is based on a model that is “not applicable to nanoscale heat transport,” she says.
Next, Pralle plans to develop, in collaboration with Anikeeva, a magneto-thermal genetics tool that can modulate multiple areas of the brain simultaneously, allowing the researchers to more fully control behavior, or multiple behaviors at one time. “If we dream about it we can overcome the technical hurdles,” Pralle says.
Emily Waltz is a features editor at Spectrum covering power and energy. Prior to joining the staff in January 2024, Emily spent 18 years as a freelance journalist covering biotechnology, primarily for the Nature research journals and Spectrum. Her work has also appeared in Scientific American, Discover, Outside, and the New York Times. Emily has a master's degree from Columbia University Graduate School of Journalism and an undergraduate degree from Vanderbilt University. With every word she writes, Emily strives to say something true and useful. She posts on Twitter/X @EmWaltz and her portfolio can be found on her website.