In Marvel comic books, the Living Laser is a mad scientist, demolished and re-aligned by lost love, who defends his evil intentions by shooting lasers out of his wrists.
That was in 1966, right around the time lasers were invented.
Now, forty-five years later, Malte Gather and Seok Hyun Yun from the Wellman Center for Photomedicine at Massachusetts General Hospital in Boston have constructed a real living laser. It's not a man, but a single cell.
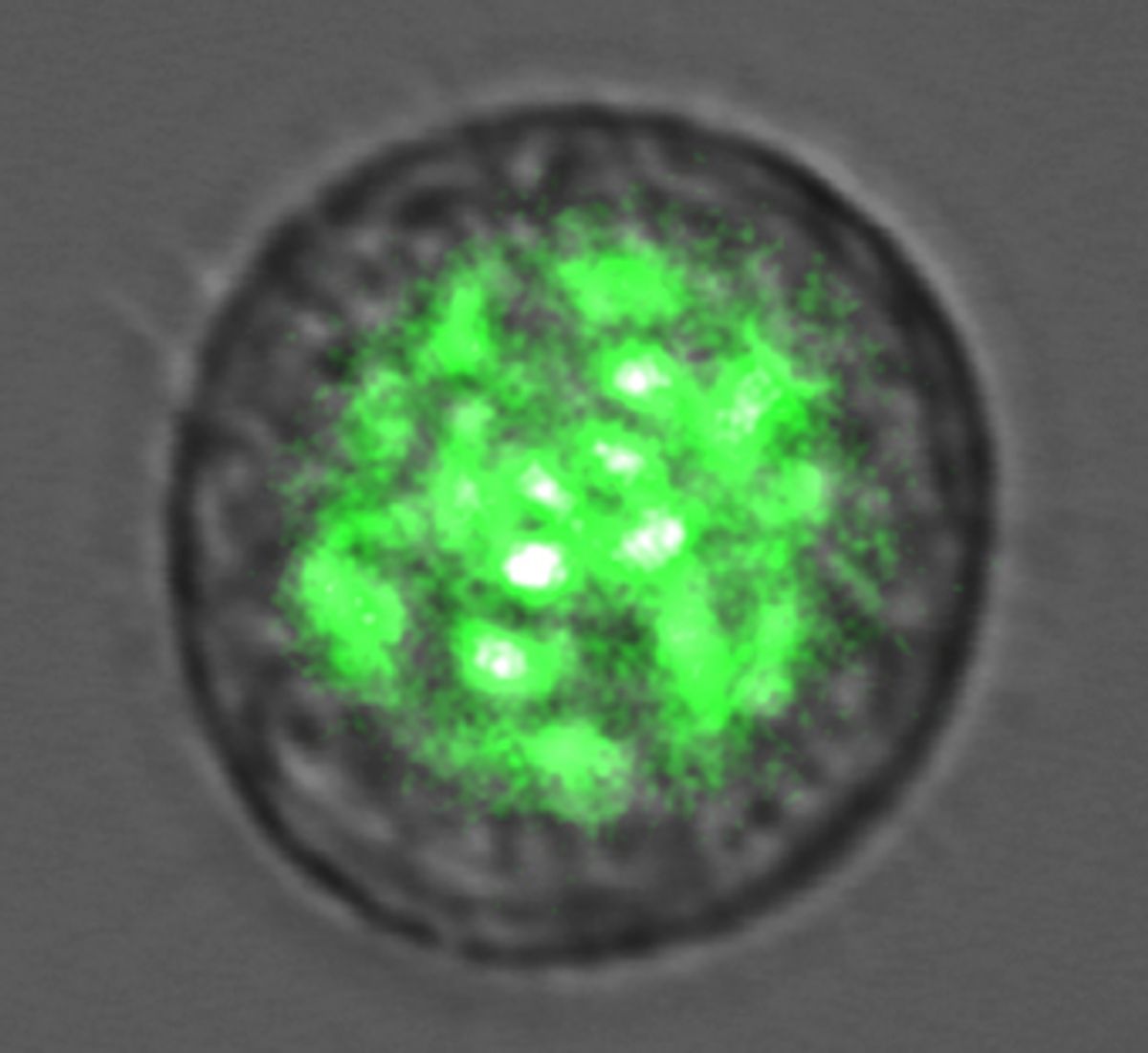
The type of cell they use is a stock character in every microbiology lab. In the early 90s, researchers made a series of breakthroughs in cellular imaging technology when they sequenced a protein in jellyfish called GFP (short for green fluorescing protein). Once the protein was sequenced, biologists were able to genetically incorporate it into any cell-type they wanted, linking it to the specific proteins they were interested in. To this day, one of the best ways to study a protein is to tag it with GFP and track it under a microscope. The cell can be alive or frozen in time with a fixative. All you have to do is shine a blue light on it and look for green fluorescence.
Malte and Yun found a way to convert these cellular emissions into a laser beam.
A laser has three basic components: a light source, a gain medium, and an optical resonator. When energy from the light source enters the gain medium, it is amplified in a process called stimulated emission. Reflectors in the optic resonator bounce the light back and forth through the medium, repeating the amplification, then direct the output into a coherent beam, what we call a laser.
People have used all kinds of materials as gain mediums—gas, crystals, glass, semiconductors—and this is the part of the assembly that is alive in Malte and Yun's design.
In a study described this month in the journal Nature Photonics, they placed a single, GFP-transfected kidney cell into a reflective chamber 20 microns wide and pumped it with short pulses of blue (465nm) light. Normally, the cells would simply fluoresce. But under these circumstances they lased.
Malte and Yun had to pump at least 1 nanojoule of energy into the cell to cause stimulated emission and activate the laser. The cells survived the experiment.
It's hard to say what their bio-laser will be useful for. The researchers have suggested that GFP expressing cells could be used to deliver laser treatment into the body. But there is still some major hardware, like the energy source, that will have to tag along with the biological components.
So many people are finding ways to re-engineer inorganic machines with living parts these days that it seems reasonable to think there's an application for this right under our noses. Researchers at Southern Methodist University, for example, are designing robotic prostheses that will link right into the body with optical fibers that can both listen and talk back to motor nerves that were severed during amputation. When it comes time for lasers to assemble at this fault line between man and machine, the living laser could be a handy tool.
The paper in Nature Photonics about Malte and Yun's work is available here.