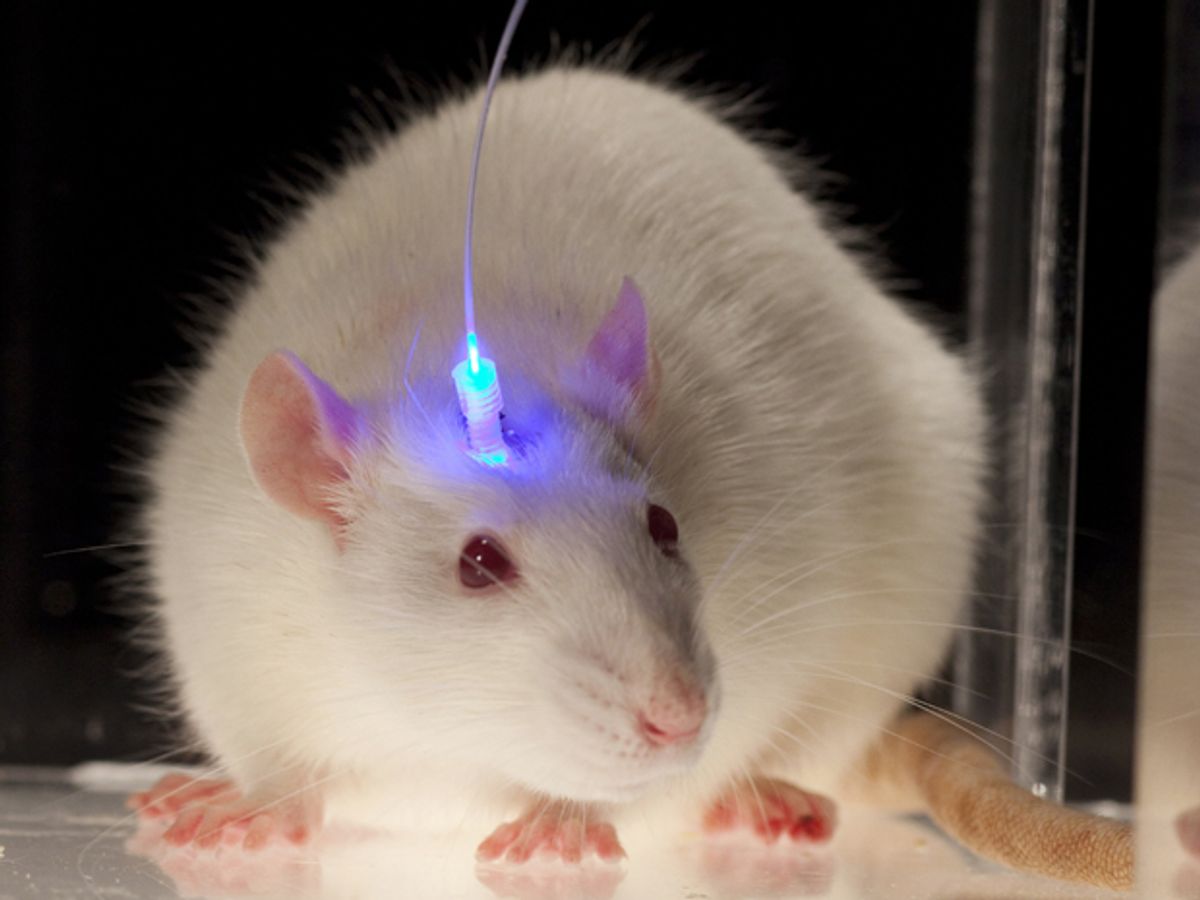
Optogenetic techniques have been used experimentally for more than a decade to control animal behavior and map neuronal networks, and have the potential to offer insight into maladies such as Parkinson's disease, addiction, depression, and spinal cord injuries. The technique involves injecting into the brain a virus that contains genetic constructs that sensitize neurons to light, and then manipulating the neurons using a laser that delivers light via optical fibers.
The MIT study presents a clever application of optogenetics. "This study shows how optogenetics could be used in a new way, to target plasticity and rewire circuits in the brain," said Roger Redondo, a co-author of the report, in an email to IEEE Spectrum. It was an ingenious application of the technology, wrote Tomonori Takeuchi and Richard Morris, neuroscientists at the University of Edinburgh, in the U.K., in an accompanying article in Nature.
The study also sheds light on which parts of the brain's circuitry are responsible for the malleability of emotional memory. It has long been apparent that emotional associations with memories can be changed, but the neuronal mechanisms and brain circuits that enable this were unknown. In fact, doctors have effectively used manipulation of emotional memory in psychotherapy to treat maladaptive behaviors. The MIT researchers wanted to know the scientific basis for how this is possible.
In a set of experiments, male mice were subjected to situations that produced fear (a small electric shock to the foot) or pleasure (interaction with a female mouse) and the memory neurons that were activated during those experiences were labeled. The mice were then put into a rectangular testing arena and the neurons associated with the fear or pleasure experiences were optogenetically reactivated with light whenever the mice entered a target side of the test arena. As expected, mice with fear memories avoided that area of the arena, and mice with pleasure memories lingered.
The researchers than attempted to reverse the positive or negative emotion associated with the memory. To do this, they took the animals to a new place, and allowed the fear-conditioned mice access to a female while optogenetically reactivating the fear neurons. They also subjected the pleasure-conditoned mice to foot shocks while reactivating the pleasure neurons with the lasers. "The assumption here is that associations in the brain are formed between neurons that are active at the same time," said Redondo during a press briefing held yesterday. "If this is right, we should be forcing neurons associated with fear . . . to link up with new neurons expressing the pleasure of spending time with a female."
The experiment worked. The next testing day, the mice were put into the rectangular testing arena again, and the animals that previously had run away from the light from the laser were now spending more time near that side of the arena, while those that had lingered near the target side were now running away. In other words, the result suggests that the mice that had been given foot shocks now "remembered" the experience positively and the mice that had spent time with a female "remembered" it negatively.
In further experiments, the MIT scientists determined that the neural circuits that were malleable were located in the brain's hippocampus in a region called the dentate gyrus. "We think their application of optogenetics was ingenious because they were able to reactivate a specific place memory representation from the training and later change its association from bad to good, or vice versa," said Takeuchi. But there may be limitations to the strategy, he noted in his article in Nature, "notably when sequences of neural firing are the essence of the memory" such as when the memory is of a musical tune or a sequence of actions.
Optogenetics is pretty invasive, with viruses, genetic constructs and hardware implants all going into the brain. And then there's the bioethical considerations. So far the technique has only been tested in animals. Slightly less invasive variations, including wireless optogenetics, have been proposed, but would still require some kind of an implant.
Because of these limitations, the authors were careful not to gush about potential therapeutic applications in humans. "It's a bit far-fetched at the moment because we don't have the proper technology," said co-author Susumu Tonegawa, a Nobel laureate, during the press briefing.
The hope is that, if the technique can prove effective and safe, emotional memory manipulation could help with afflictions like severe depression and post-traumatic stress disorder.
In the press briefing, a reporter brought up a theoretical mad scientist scenario in which bad guys are zapping the memories of people against their will. Tonegawa laughed, and said: "We have no intention . . . to use this kind of technology in order to alter normal, healthy people's minds or cognition. If there is any application of this in humans it is for pathological conditions—to reduce the suffering in people who have these psychiatric disorders." Added Redondo: "In any case, the media is much better than neuroscientists at changing memories of a series of events."
Fair enough.
Emily Waltz is the power and energy editor at Spectrum. Prior to joining the staff in January 2024, Emily spent 18 years as a freelance journalist covering biotechnology, primarily for the Nature research journals and Spectrum. Her work has also appeared in Scientific American, Discover, Outside, and the New York Times. Emily has a master's degree from Columbia University Graduate School of Journalism and an undergraduate degree from Vanderbilt University. With every word she writes, Emily strives to say something true and useful. She posts on Twitter/X @EmWaltz and her portfolio can be found on her website.