How We’re Shrinking Chemical Labs Onto Optical Fibers
Lab-on-fiber sensors could monitor the environment and hunt for disease inside your body
Think back to the last time you got a blood test. Maybe you had your cholesterol checked or got screened for infections, heart disease, stroke risk, thyroid troubles, or osteoporosis. Easy, right? A nurse simply drew your blood and shipped the vials to a lab. But behind the scenes, the process gets more complex. Today’s laboratory technologies require rooms full of temperature-controlled chemicals, analytical machines worth hundreds of thousands of dollars, and trained technicians to run them. That’s why it probably took days, maybe even a week or two, to get your results. And depending on the tests, a panel of them could easily have set you back several hundred dollars.
Such expenses are a major reason that health-care costs are rising around the world. In rural communities or developing countries, the situation may be much worse than inconvenient. Patients may travel hours or days just to reach a clinic and often don’t make it back to collect the results. Many of them can’t afford tests for such life-threatening diseases as malaria and tuberculosis.
To help solve these problems, we need better laboratory instruments. They should be cheap and portable so that they can be distributed to remote clinics, village doctors, battlefields, disaster zones, and patients’ homes. They should be easy to use so that nurses, hospice workers, soldiers, and even patients themselves can administer tests with minimal training. Finally, these tools should provide results quickly and reliably so that patients don’t have to wait or worry.
Imagine an entire laboratory that fits inside a case the size of a tablet computer. The lab would include an instrument for reading out results and an array of attachable microsize probes for detecting molecules in a fluid sample, such as blood or saliva. Each probe could be used to diagnose one of many different diseases and health conditions and could be replaced for just a few cents.
This scenario is by no means a pipe dream. The key to achieving it will be optical glass fibers—more or less the same as the ones that already span the globe, ferrying voluminous streams of data and voice traffic at unmatchable speeds. Their tiny diameter, dirt-cheap cost, and huge information-carrying capacity make these fibers ideal platforms for inexpensive, high-quality chemical sensors.
We call this technology a lab on fiber. Beyond being an affordable alternative to a traditional laboratory, it could take on tasks not possible now. For instance, it could be snaked inside industrial machines to ensure product quality and test for leaks. It could monitor waterways and waste systems, survey the oceans, or warn against chemical warfare. One day, maybe as soon as a decade from now, it could be injected into humans to look for disease or study the metabolism of drugs inside the body.
Diagnosis by Light
A lab on fiber uses near-infrared frequencies to detect precise concentrations of chemicals or biological molecules in a solution. Here’s how it works. Hover over the numbers for more information.
Researchers first began imagining ways to build low-cost mobile laboratories in the late 1960s. Around that time, engineers had figured out how to pack thousands—and eventually billions—of transistors onto a single chip the size of a thumbnail, paving the way for powerful microprocessors and dense, fast memory. And as microelectromechanical systems advanced, biomedical investigators began using these technologies to build compact arrays of chemical sensors on a single silicon chip.
Such a system, known as a lab on a chip, varies widely in design and complexity. A typical setup includes miniature pumps and valves that guide a small liquid sample—a drop of blood, say—through microchannels to various detection stations. At these sites, target molecules in the blood—such as glucose or viral antibodies—react with other chemicals on the chip, changing the voltage across electrodes or varying the current flowing through a conductor. The chip amplifies these signals, digitizes and analyzes them, and then sends the results through wires or a radio channel to a handheld display. The whole process takes no more than 20 minutes.
Lab-on-a-chip sensors are ideal for use in rural clinics or at a patient’s bedside. But their widespread use for other tasks has long been stalled by seemingly insurmountable obstacles. For example, in wet environments—such as inside the body or outdoors—a chip’s metal conductors easily corrode or short, making the sensor unreliable. Many chips also contain materials such as arsenic that are toxic to humans. Their biggest drawback, though, is size. Today’s power sources, processors, and transmitters take up at least a few square centimeters—too big to squeeze through blood vessels.
To overcome many of these problems, some researchers are seeking to replace a chip’s electronic circuits with optical ones. By using light rather than current to read chemical reactions, a photonic chip works reliably in aqueous solutions, is immune to electromagnetic radiation, tolerates a wide range of temperatures, and poses fewer risks to biological tissues.
And light has another important advantage. Electronics relay information using only one measurable parameter, the magnitude of a current or voltage. Optical devices similarly encode data by varying the intensity, or amplitude, of a light wave. But light can also be divided into many wavelengths, or colors, providing multiple delivery channels. This ability to multiplex data vastly increases a circuit’s information capacity, which could make photonic lab-on-a-chip sensors orders of magnitude more sensitive than electronic ones.
Yet despite its many desirable qualities, a photonic lab on a chip still presents some significant challenges. Like electronic chips, optical systems are far too large to fit in the body. And in at least one respect, the problem is even worse. Light waves can’t be squeezed into spaces much narrower than their wavelengths. So photonic chips, which use near-infrared wavelengths of around 1 micrometer, are much larger than their electronic cousins, whose ICs can have features smaller than 30 nanometers.
Photonic lab-on-a-chip sensors are also expensive to make, because they require complex arrangements of lenses and mirrors to shape light beams and direct the waves to chemical-detection stations. Photonic ICs built on silicon substrates, for example, which are used in telecommunications equipment today, cost several hundred dollars apiece. Meanwhile, you can buy an electronic chip for pennies.
So how do we take advantage of light’s wonderful properties without the drawbacks? We build our lab not on a silicon photonic chip but on a glass fiber.
Ever since optical fibers became cheap and abundant in the 1980s, researchers have experimented with all sorts of ways to build lab-on-fiber sensors. Now, several groups around the world are homing in on the best materials and assembly methods for manufacturing robust systems cheaply and on a large scale.
For instance, my group at Carleton University, in Ottawa, Canada, and our collaborators, including engineers at the Université de Mons, in Belgium, and Jinan University, in China, are developing a lab on fiber that is simple to make and yet can provide extremely precise measurements. We begin the fabrication process with a standard telecom fiber, which costs next to nothing.
This hair-thin glass fiber consists of an inner core and outer cladding encased in a protective polymer jacket. While the cladding is made of pure silica, the core is laced with tiny amounts of germanium oxide, which raises its refractive index, a measure of the speed of light in a material. If light traveling through the core strikes the cladding within a certain critical angle, the slight difference in refractive indices between the two layers causes the beam to bend back into the core—a property called total internal reflection. In this way, light can ping-pong down a fiber’s core for up to several kilometers. The ability to carry light over long distances with very little loss could allow fiber labs to be inserted into water supply networks, lowered into the ocean, or strung throughout hospitals.
Where to Put The Probe?
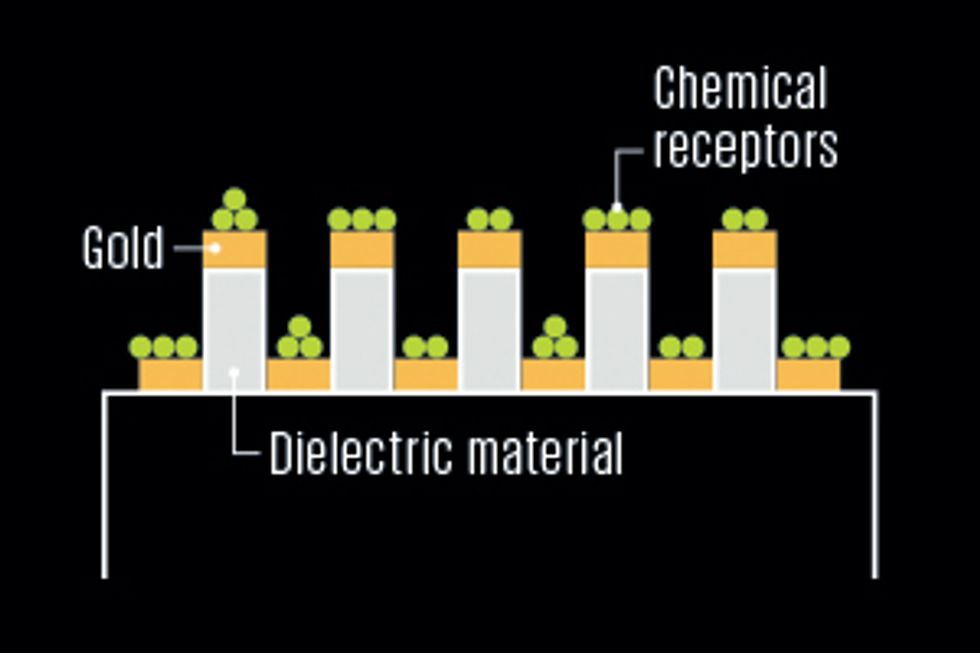
One of the best ways to make a lab on fiber is to build the chemical detector, or probe, on the outer surface of an optical fiber [see illustration, "Diagnosis by Light"]. But that’s not the only approach. Here are two leading alternatives.
On the Tip
Inside the Fiber
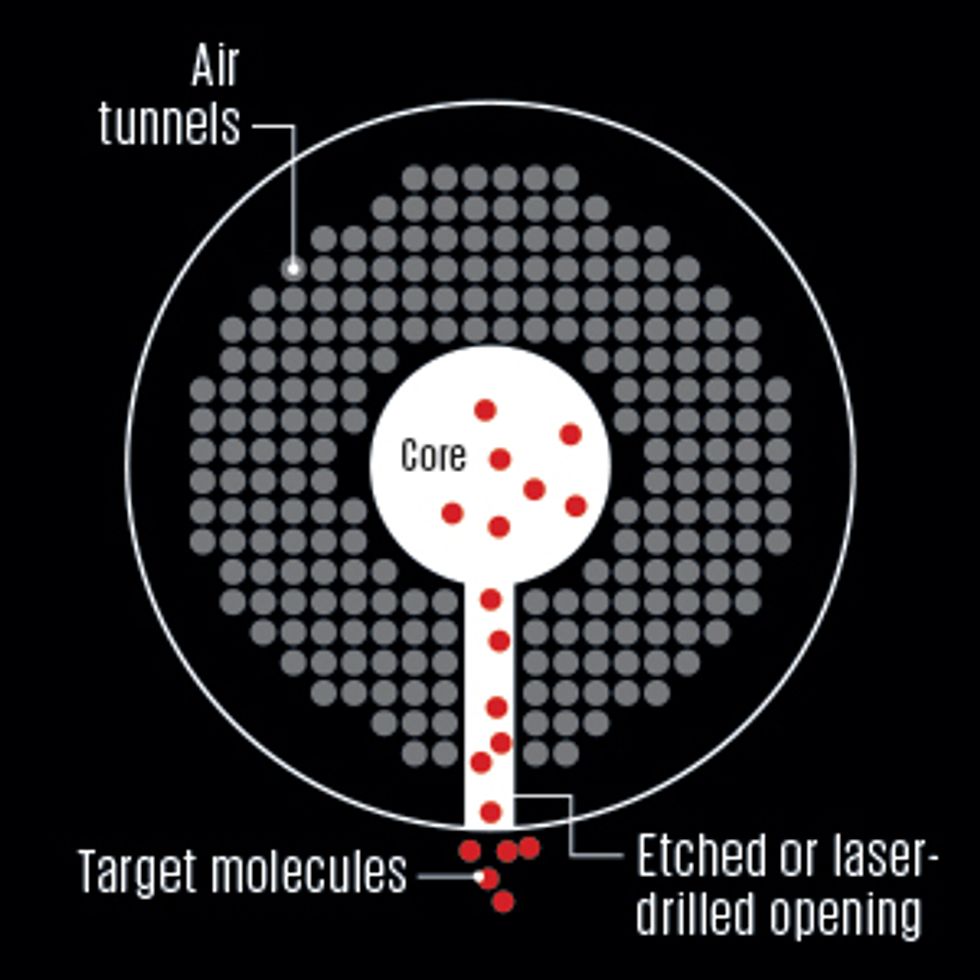
To transform our inert fiber into a chemical sensor, we must first choose where to put the detection site. Some groups are investigating designs that place this probe on a fiber’s tip or inside tiny air tunnels in the cladding of an experimental type of fiber called a microstructured optical fiber [see sidebar, "Where to Put the Probe?"]. We have found, though, that the simplest—and therefore cheapest—approach is to use a short segment of a fiber’s outer surface, around 1 to 10 millimeters long.
We coat this probe with a chemical compound, called a reagent, that will interact with whatever target molecules we want to measure, such as blood enzymes or food additives. There are many different compounds that can serve as reagents and many ways to attach them to a fiber. One of the most promising processes that we are developing takes two steps. First we cover the surface of the probe with a thin layer of metal using standard deposition techniques such as sputtering, evaporation, or electroless plating. Then we dip the probe into a salt bath filled with synthetic molecules called aptamers, which bind to the surface coating, forming the reagent.
Chemical supply companies can make a huge variety of aptamers that each bind to a different target molecule, including various proteins, toxins, and even whole bacteria. So it’s possible to make fiber probes that can test for just about any chemical or biological substance. We can repurpose a used probe by stripping the original aptamers in a chemical wash and attaching new ones.
Now that our fiber lab has the ability to capture molecules, we need a way to count them. To perform this task, we use light. We connect a micro light source, such as a laser diode, to one end of the fiber, which naturally guides the light through the core to our chemical probe. We cap the opposite end of the fiber with a gold mirror so that we can analyze the returning waves with a commercial spectrometer.
But this uniform reflection won’t tell us anything about changes occurring at the probe, such as molecules attaching to its aptamers. In order for us to observe these events, the light must be able to interact with the probe’s outer surface, where these aptamers reside. So we must somehow force the light to leave the core.
Many early solutions involved removing part of a fiber’s cladding, through polishing or chemical etching, to expose the light to the outside world. But such alterations weaken the fiber and are difficult to do precisely, which increases the cost of the sensor.
In our lab, we instead build what’s known as a tilted grating inside the core of our probe. It is essentially a permanent hologram, which we create by exposing the fiber to intense ultraviolet laser light for just a couple of minutes before adding the metal coating and reagent. First we dissolve the plastic buffer around the probe, making it transparent to UV light. We shine the UV light through a grooved glass plate, called a phase mask, which splits it into two beams. Then we place the fiber behind the mask, where these beams interfere with one another, creating a periodic radiation pattern: intervals of high exposure separated by intervals of no exposure. The radiation breaks some of the electronic bonds in the fiber core, increasing its refractive index in the same pattern as the interfering light, like an image being formed on a photographic film. Each rise in the core’s refractive index is called a fringe, and together they make up the grating.
The disklike fringes act like tiny imperfect mirrors: Each reflects a small amount of light traveling through the probe. If we build the grating so that its fringes align straight up and down inside the core, they will reflect this light back toward its source. But remember, we want the grating to redirect the light through the cladding to the fiber’s surface. So we simply tilt the fiber as it’s being irradiated with the UV light, forming a tilted grating.
It’s important to note that a grating doesn’t reflect all light out of the core. Rather, it targets only certain wavelengths known as resonances, which depend on the distance between fringes and the refractive properties of the fiber. As these resonances leave the core, they bounce off the fiber’s surface and propagate backward through the cladding. They travel only a couple of centimeters at most before being fully absorbed by the fiber’s plastic covering. So by analyzing the entire spectrum of light that reflects off the gold mirror and returns to the spectrometer through the core, we can identify these escaped resonances and how much of this light was lost.
Now we’re ready to put our probe to work. When we dip it in a test solution, such as human blood, any target molecules that are present—traces of pharmaceutical drugs, for example—will bind to the aptamers on the probe’s surface, altering the way it reflects and absorbs light. This small physical transformation tweaks the color or intensity of the resonances leaving the core. Then by analyzing the characteristics and magnitude of these changes, we can determine the concentration of molecules reacting with our probe.
So how sensitive is our little fiber laboratory? That depends on the material we use to attach the molecule-catching aptamers to a probe’s surface. You may recall that we first coat the fiber with metal before attaching the aptamers. Remarkably, we have found that this metal layer can increase the sensitivity of our lab by as many as four orders of magnitude.
The explanation involves some rather bizarre physics. The thickness of our coating is typically between 10 and 100 nm, as little as 1/100th the length of the resonant light waves that reach it. If the coating were made of almost any other material, a lot of this light would pass by without being affected by changes at the coating’s surface, such as from target molecules attaching to the aptamers. That’s because these wavelengths are so much larger than the width of the coating. We might say that the light fails to notice the chemical changes.

But our metal coating has unique properties that force more light to interact with it. Because metal is conductive, its surface electrons oscillate in the presence of an electromagnetic field, such as that produced by a light wave. Under the right conditions, having to do with the wavelength and angle of the light and the material properties of the coating, the oscillations of electrons belonging to nanoparticles in the metal will resonate with the oscillations of the electromagnetic field. These electron resonances are called localized surface plasmons. Like sound vibrations amplified by the air inside a guitar, they absorb energy from the light wave inducing the field, creating hot spots of electromagnetic energy that can extend as far as several hundred nanometers beyond the metal surface. Because plasmons take up so much more space than the nanoparticles they surround, a light wave is far more likely to “see” the molecular events that affect them.
In one experiment, for example, we demonstrated that an uncoated fiber probe could detect 20 micrograms of a common protein, called biotin, per liter of test solution. A gold-coated probe, however, could detect concentrations as small as 2 nanograms per liter, which translates to roughly a pinch of table salt in a 25-meter swimming pool. (We use gold because it’s chemically inert, so it’s considered safe to use inside the body and won’t degrade over time.)
Due to its small size and extraordinary sensitivity, a lab on fiber could take on many diverse roles. As reported in a 2012 paper in the journal Small, for instance, researchers led by Jeroen Lammertyn at Katholieke Universiteit Leuven, in Belgium, used a gold-coated probe to detect the smallest possible variations in snippets of DNA. The results suggest that this inexpensive tool could one day provide quick and accurate genetic screening for complex conditions such as cystic fibrosis, cancers, and certain infections.

More recently, our group demonstrated that a lab on fiber can also be used to monitor living cells. For this experiment, we submerged a probe in a culture of skin cells, which attached themselves to it. When we fed the cells nutrients, they grew, increasing the density of the fiber’s surface and thereby altering the patterns of light arriving at our spectrometer. Conversely, when we exposed the cells to toxins, they died and detached from the probe, decreasing its surface density and resulting in differences in the spectrogram. Scientists may find lab-on-fiber sensors particularly favorable for studying living tissues because they are too small to affect the cells’ behavior.
Ultimately, we aim to develop a lab on fiber that can be inserted directly into humans to monitor biological changes happening in real time. We are currently planning experiments—first in test tubes and eventually in animals—to see if a fiber probe can detect metastasized cancer cells in the bloodstream. We hope to shed light (literally) on the process by which these cells invade other organs. We also hope that this work will help develop new cancer-screening technologies that are less invasive than current tools such as biopsies. For instance, a doctor could insert a fiber probe into a blood vessel using a hypodermic needle. The procedure would be no more painful than a flu shot.
It will probably be at least five years before lab-on-fiber instruments are ready for commercial use. For example, a remaining major challenge is figuring out how to toughen the surface coating on the probes so that they can be stored for several months without becoming unstable and thereby losing their ability to bind with target molecules.
Nevertheless, lab-on-fiber technology is tantalizingly close to being able to compete in cost and performance with today’s diagnostic tools for many applications. One of the first might very well be a blood test: Imagine turning on your home lab kit, pricking your finger, and blotting the blood on an array of fiber probes. In just a few minutes, the machine would automatically e-mail the results to your doctor, who could get back to you within hours if there was a problem. Meanwhile, you could get on with the rest of your day.
This article originally appeared in print as “A Lab On Fiber.”
About the Author
Jacques Albert heads the Advanced Photonic Components group at Carleton University in Ottawa, Canada. He is captivated by the elegant way that physical models can find “simplicity in things that are apparently complex.” As an engineer of fiber optic devices, he strives for simplicity in his designs. “I look at those complex devices and tell myself, ‘There has to be a better way.’ ”