Artificial intelligence in healthcare is often a story of percentages. One 2017 study predicted AI could broadly improve patient outcomes by 30 to 40 percent. Which makes a manifold improvement in results particularly noteworthy.
In this case, according to one Israeli machine learning startup, AI has the potential to boost the success rate of in vitro fertilization (IVF) by as much as 3x compared to traditional methods. In other words, at least according to these results, couples struggling to conceive that use the right AI system could be multiple times more likely to get pregnant.
The Centers for Disease Control and Prevention defines assisted reproductive technology (ART) as the process of removing eggs from a woman’s ovaries, fertilizing it with sperm and then implanting it back in the body.
The overall success rate of traditional ART is less than 30%, according to a recent study in the journal Acta Informatica Medica.
But, says Daniella Gilboa, CEO of Tel Aviv, Israel-based AiVF—which provides an automated framework for fertility and IVF treatment—help may be on the way. (However, she also cautions against simply multiplying 3x with the 30% traditional ART success rate quoted above. “Since pregnancy is very much dependent on age and other factors, simple multiplication is not the way to compare the two methods,” Gilboa says.)
In the U.S. alone, 7.3 million women are battling infertility, according to a 2020 report from the American Society for Reproductive Medicine. In the U.S., 2.7 million IVF cycles are performed each year.
AiVF is using ML and computer vision technology to allow embryologists to discover which embryos have the most potential for success during intrauterine implantation. AiVF is working with eight facilities in clinical trials around the world, including in Israel, Europe and the United States. It plans to launch commercially in 2021.
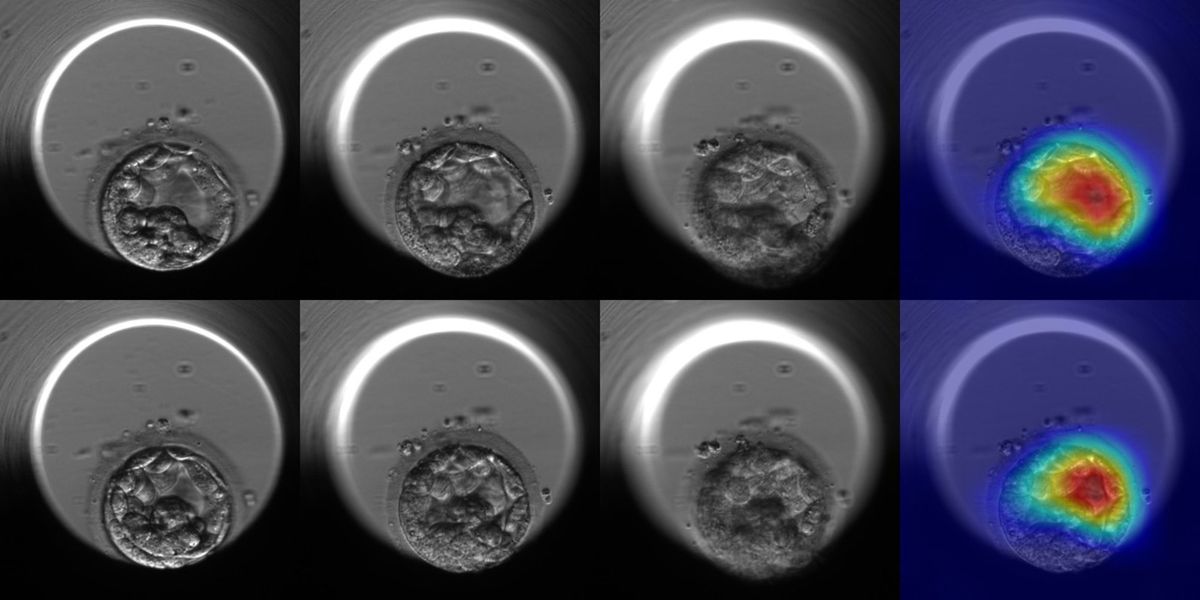
Ron Maor, head of algorithm research at AiVF, says that AiVF has built its own “bespoke” layer on top of various off-the-shelf AI, ML and deep learning applications. These tools “handle the specific and often unusual aspects of embryo images, which are very different from most AI tasks,” Maor says.
AiVF’s ML technique involves creating time-lapse videos of developing embryos in an incubator. Over five days, the video shows the milestones of embryo development. Gilboa explains that previous methods yielded just one microscope image per day of the embryo compared with computer vision’s greater image-capturing success.
“By analyzing the video, you could dig out so many milestones and so many features the human eye cannot even detect,” Gilboa says. “Basically you train an algorithm on successful embryos, and you teach the algorithm what are successful embryos.”
Likely only one embryo out of 10 can be implanted in the uterus. Once a physician implants the embryo, the embryologist will know within 14 days whether the patient is pregnant, Gilboa says.
“As an embryologist I look at embryos, and I understand what happens to them,” Gilboa says. “If I learn on maybe thousands of embryos, the algorithm would learn on millions of embryos.”
As AiVF’s initial results suggest, computer vision and ML could potentially drive IVF’s prices down—in turn making it less expensive and burdensome for a woman to become pregnant.
“Once you have a digital embryologist, then you could set up clinics much easier,” Gilboa says. “Or each clinic could be much more scalable. So many more people could enjoy IVF and achieve their dream of having a child.”