Fires resulting from the catastrophic failure of lithium-ion batteries could be prevented with chemical additives, say researchers at Stanford University.
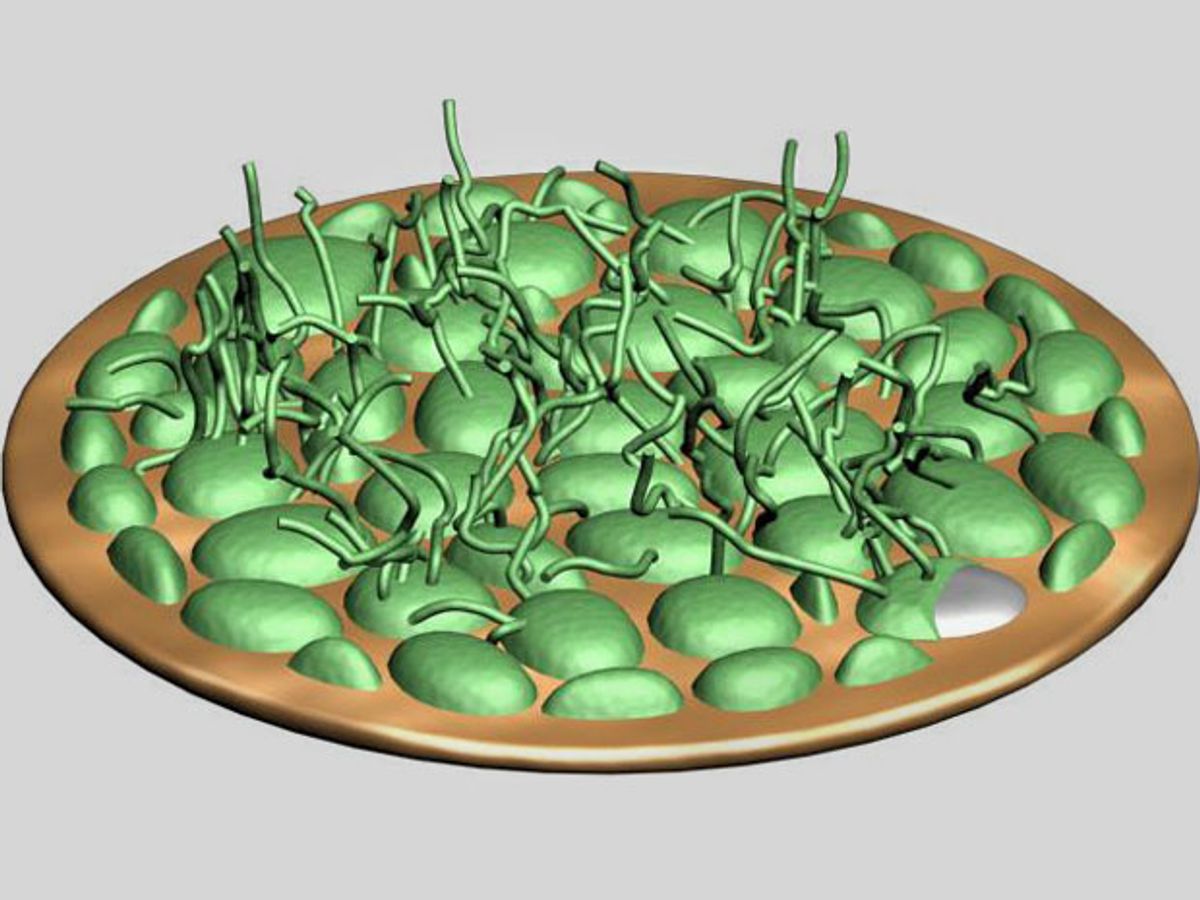
When lithium-ion batteries overheat, they can burn through internal pockets, burst into flames, and even explode. One reason such damage can occur is the formation of dendrites—finger-like deposits of lithium that can grow long enough to pierce the barrier between a lithium-ion battery's halves and cause it to short out.
Dendrites form when a battery electrode degrades and metal ions deposit onto the electrode's surface. Previously, scientists at Stanford developed a lithium-ion battery that can detect when dendrites start to puncture the barrier between its halves and warn that it needs to be replaced.
In their latest work, detailed in the 17 June online edition of the journal Nature Communications, the Stanford researchers used chemicals designed to prevent dendrite formation. These get added to the battery's electrolyte, the fluid in which electrons flow between the battery's electrodes.
One of the additives, lithium nitrate, has been researched for a long time as a way to boost battery performance. The other, lithium polysulfide, has been considered a nuisance; it forms when a sulfur electrode degrades and can wreck lithium metal electrodes. But together, these additives can synergistically react with lithium metal to form a stable coating that protects the electrode from further degradation.
In experiments with coin cell batteries similar to the ones that power calculators and watches, the additives prevented dendrite formation, with harmless pancake-like deposits forming instead. Such batteries could operate at 99 percent efficiency after more than 300 charge-discharge cycles, compared with less than 92 percent efficiency after 180 cycles for batteries doped with only lithium nitrate.
This approach could help remove a major barrier to developing lithium-sulfur and lithium-air batteries, which can store up to 10 times more energy per unit mass than batteries now used in consumer electronics and electric cars, but have a tendency to form dendrites. It may also work for batteries made with metals such as magnesium, calcium, or aluminum, which also could store much more energy than today's batteries.
Charles Q. Choi is a science reporter who contributes regularly to IEEE Spectrum. He has written for Scientific American, The New York Times, Wired, and Science, among others.