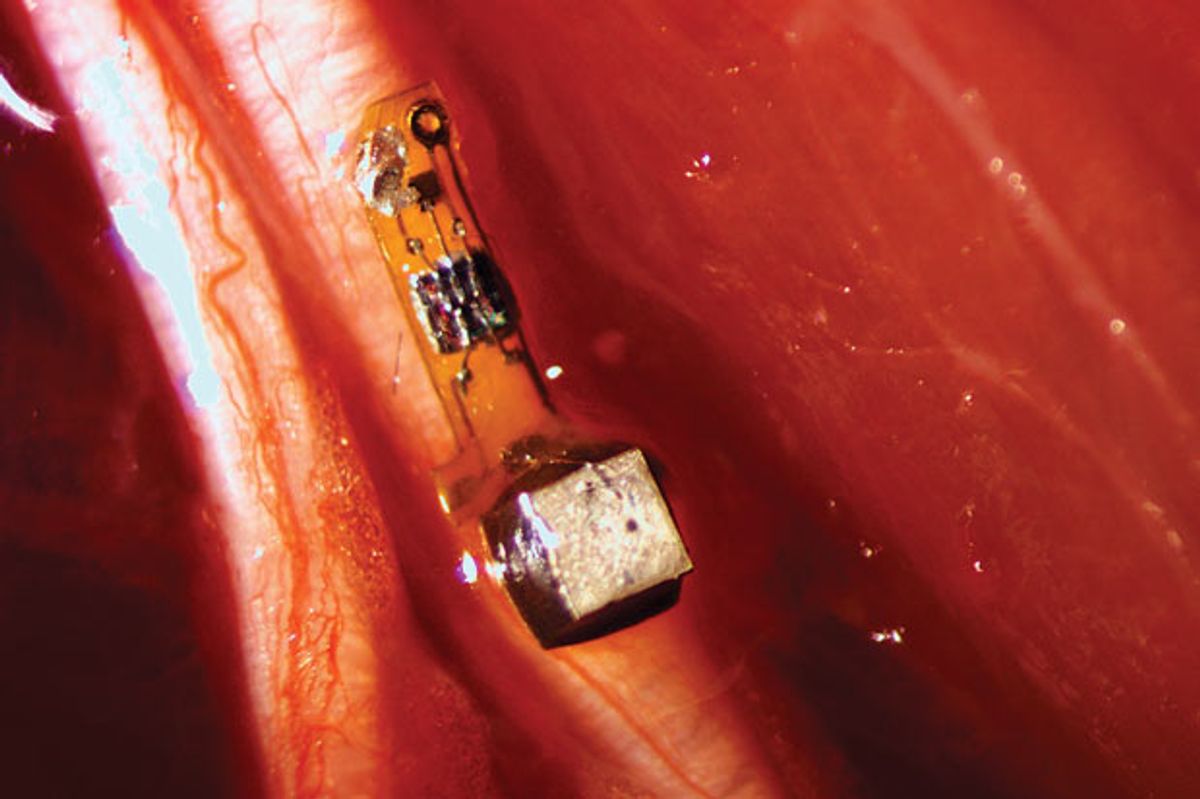
Mainstream medicine is making increasing use of electronics inside the body, deploying implanted gadgets both to measure internal conditions and to provide stimulating jolts of electricity to nerves and muscles. But turning a human into a proper cyborg will require many minuscule devices that can be scattered throughout the body. As a step toward that goal, a team of bioengineers has built speck-size wireless electrodes that can be affixed directly to nerves—and that may one day be nestled inside the brain.
The engineers from the University of California, Berkeley, implanted one mote of what they call “neural dust” inside an anesthetized rat, and demonstrated that the electrode could record signals from the rat’s sciatic nerve and wirelessly transmit the information. This experiment was a proof of concept, says Jose Carmena, who co-led the research at UC Berkeley’s Center for Neural Engineering and Prostheses, where he is codirector. If the neural dust can be adapted for the human body and brain, doctors could have an intimate new interface with the human nervous system.
But first, it had to work in a conked-out rat.
To power the neural dust, a transducer outside the animal’s body emits ultrasound vibrations that pass through skin and tissue. When the sound waves reach the implanted mote, its piezoelectric crystal converts the vibrations’ mechanical energy into electricity, providing power to a tiny transistor pressing against the nerve. As natural electrical activity in the nerve varies, it changes the current passing through the transistor, thus providing a read-out mechanism for the nerve’s signal.
To send the information back out of the body, the neural dust system also uses ultrasound. The external transducer alternates between sending ultrasound vibrations to power the mote and listening for the returning echo as some of those vibrations bounce back. The changing current through the transistor alters the piezo crystal’s mechanical impedance, thereby modulating how much bounce back the transducer receives.
To make the system viable for humans, Carmena and his colleagues are tackling four distinct technical challenges:
Challenge 1: Make the neural dust system work in rats that are awake and scurrying around.
The use of ultrasound to power the mote has the big advantage of making the system wireless, while other neural recording systems often use bulky batteries or ungainly wires that protrude through the skin. But going wireless has a downside. The external transducer must be aligned precisely with the implanted dust mote so the ultrasound vibrations hit its piezoelectric crystal dead-on. When an animal is unconscious, it’s easy to keep that alignment, but it becomes a trickier problem when the rat is awake and on the move.
So study coleader Michel Maharbiz, in Berkeley’s electrical engineering department, is now working on a rodent-wearable transducer. A tiny backpack holding the gear could be positioned over the implanted mote to keep the proper alignment, he says.
Still, some experts say the alignment problem may make the neural dust system impractical in human patients. Complicated external components could cancel out benefits gained by the simple wireless implants, says Dustin Tyler, a biomedical engineering professor at Case Western Reserve University, in Cleveland, who works on cuff electrodes that wrap around nerves. “Daily donning and doffing of arrays of [ultrasonic] transducers that need to be properly aligned can be quite daunting,” he says, “and often prevent patients from using the system or from using it properly.”
Challenge 2: Use multiple motes inside a single animal’s body.
In the proof of concept, the researchers not only recorded from a rat’s nerve, they also recorded electric signals from a muscle. By attaching motes to many muscles and nerves, the neural dust could be used as an internal sensor network, says Maharbiz. “Deep-tissue temperature sensors would enable monitoring organ function in a way not possible today,” he says.
Powering and recording signals from multiple motes will require new techniques and better signal processing. The researchers may use an array of ultrasound transducers that sweep their beams across an animal’s body, hitting all the motes in turn. The engineers are also writing new signal processing algorithms to make sense of returning echoes from multiple sources. Carmena is optimistic, noting that bats use the similar process of echolocation to navigate in caves filled with thousands of animals. “If the bats can do it,” he says, “we should be able to do it too.”
Challenge 3: Use the neural dust not only to record but also to stimulate nerves.
While recording signals from the nerves and muscles provides information about the body, researchers in the hot new field of “electroceuticals” are more interested in stimulating nerves with pulses of electricity to change the body’s operations. Electroceutical companies are developing medical treatments that involve stimulating the vagus nerve in the neck, for example, showing progress on reducing inflammation in rheumatoid arthritis and blocking the pain of migraine headaches.
But the current neural dust mote is a very low-power device, drawing only 0.12 milliwatt. Chad Bouton, who heads the Center for Bioelectronic Medicine at the Feinstein Institute, in Manhasset, N.Y., says a stimulating mote will require more power. “They have the right idea, but it’s going to take some work to develop that stimulating ability,” he says. Bouton’s lab has developed another nerve cuff with densely packed electrodes to provide powerful stimulating bursts, but that device currently requires penetrating wires.
Challenge 4: Make the motes small enough to be embedded in the brain.
The name “neural dust” is a bit aspirational: The researchers’ latest mote is about 2.4 cubic millimeters, which is far bigger than your average dust particle. If they want to scatter their dust in the human brain, they’ll need to make more-minuscule motes.
The researchers say their prototype mote used a commercially available circuit board. They think they can get the size down to 1 mm3, about the size of the piezoelectric crystal in the prototype, by developing a customized board. And they’re already working toward a more ambitious goal: a mote that measures 50 cubic micrometers. At that size, motes could be tucked into the deep folds and wrinkles of the brain, letting us peer into the mysterious, dusty corners of the mind.
This article appears in the November 2016 print issue as “4 Steps to Dust-Size Neural Implants.”