
Scientists have come up with two clever new ways to harness the programming power of DNA in living bacterial cells. In separate experiments published in Naturein July, researchers reported that they had successfully archived a movie and built a complex biological computer inside living E. coli cells.
The experiments expand our ability to exploit DNA’s encoding potential. “What these papers represent is just how good we are getting at harnessing that power,” says Julius Lucks, a bioengineer at the Center for Synthetic Biology, at Northwestern University, in Evanston, Ill., who was not involved in either report.
Researchers for both experiments relied on electrical engineering principles to achieve their feats. In the DNA storage experiments, a team at Harvard University demonstrated for the first time how to encode a movie and an image into living cells. Storage of digital data in DNA has been achieved before—as much as 200 megabytes—but until now, no one had archived data inside a living organism, says Seth Shipman, a neuroscientist at Harvard who led the experiments.
To get a movie into E. coli’s DNA, Shipman and his colleagues had to disguise it. They converted the movie’s pixels into DNA’s four-letter code—molecules represented by the letters A,T,G and C—and synthesized that DNA. But instead of generating one long strand of code, they arranged it, along with other genetic elements, into short segments that looked like fragments of viral DNA.
E. coli is naturally programmed by its own DNA to grab errant pieces of viral DNA and store them in its own genome—a way of keeping a chronological record of invaders. So when the researchers introduced the pieces of movie-turned-synthetic DNA—disguised as viral DNA—E. coli’s molecular machinery grabbed them and filed them away.
The movie they stored was a 36-by-26-pixel GIF of one of the first moving images ever recorded: a galloping mare named Annie G., by Eadweard Muybridge in 1887. The team was able to retrieve it, along with a separate image, with about 90 percent accuracy by sequencing the bacterium’s genome.
The same month Shipman announced the storage breakthrough, a separate group of researchers reported another cleverly programmed piece of synthetic DNA. This one, when introduced into E. coli, can direct the cell to produce a biological computer made of ribonucleic acid, or RNA.
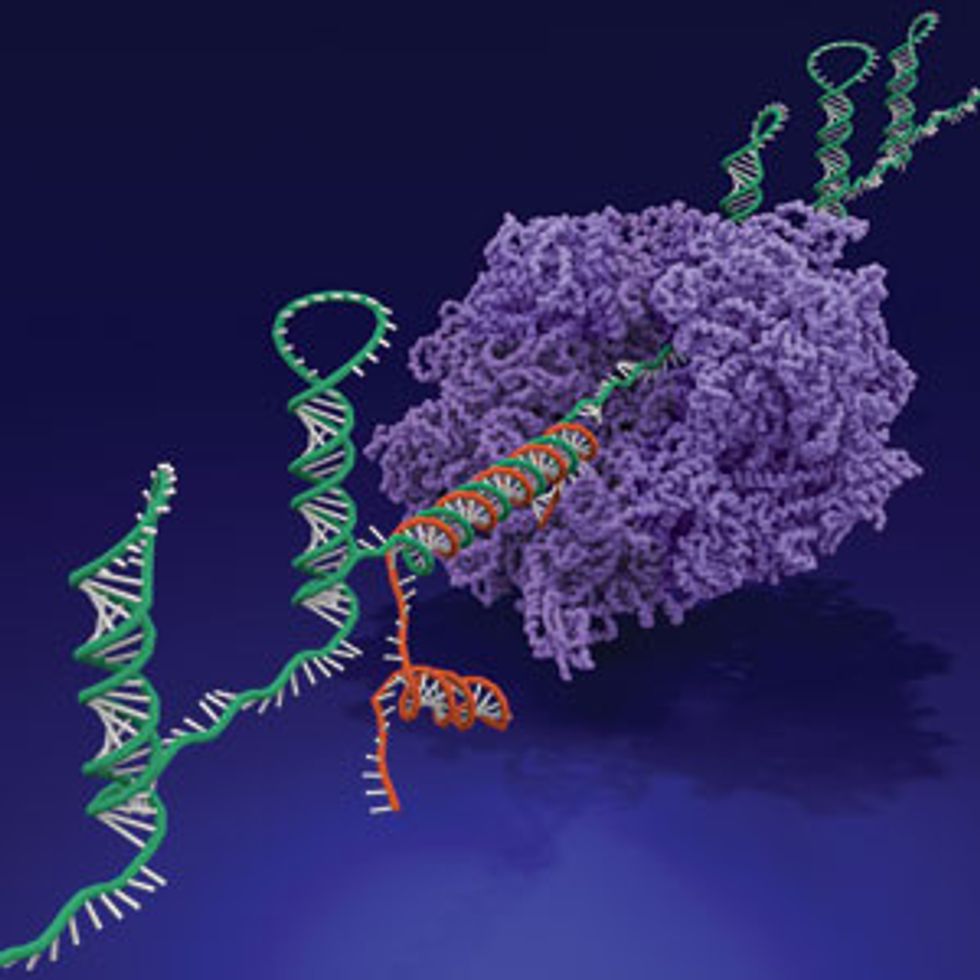
The “ribocomputer” can evaluate up to a dozen inputs, make logic-based decisions using AND, OR, and NOT operations, and give the cell commands. The system is the most complex biological computer to date and is one of the few that operates inside a living cell, says Alexander Green, an engineer at the Biodesign Institute at Arizona State University, in Tempe, who developed the technology with colleagues at Harvard’s Wyss Institute for Biologically Inspired Engineering.
The biological circuit enables researchers to program cells to respond when they receive a particular type of input. For instance, cells could be programmed to light up or self-destruct when they sense the presence of a toxin or a marker of cancer.
Taken together, these advances in DNA storage and biological computing are reminiscent of the early days of electronics, say researchers. “It’s like a golden age of circuit design,” says Timothy Lu, a bioengineer in the Research Laboratory of Electronics at MIT, who was not involved in the work. “It’s a great time for creative circuit engineers to be in the field.”
In order to build more complex biological computers, or store increasingly complex data in DNA, bioengineers will need to borrow electrical engineering concepts. “That way of thinking—the way that electrical engineers have gone about establishing design hierarchy or abstraction layers—I think that’s going to be really important for biology,” says Lu.
One doesn’t need a life sciences background to participate, adds Lucks. “We can create a layer of abstraction where you don’t need to know about RNA folding to design a circuit out of RNA,” he says.
At some point—perhaps much further down the road—the public will need to weigh in on the idea of forcing living things to perform functions that fall so far outside their normal activities. “We can make bacteria compute information and store a movie. Is that okay?” says Lucks. “I don’t think anybody would really argue that it’s unethical to do this in E. coli. But as you go up in the chain [of living organisms], it gets more interesting from an ethical point of view.”
A version of this article appeared as two posts in The Human OS blog.
