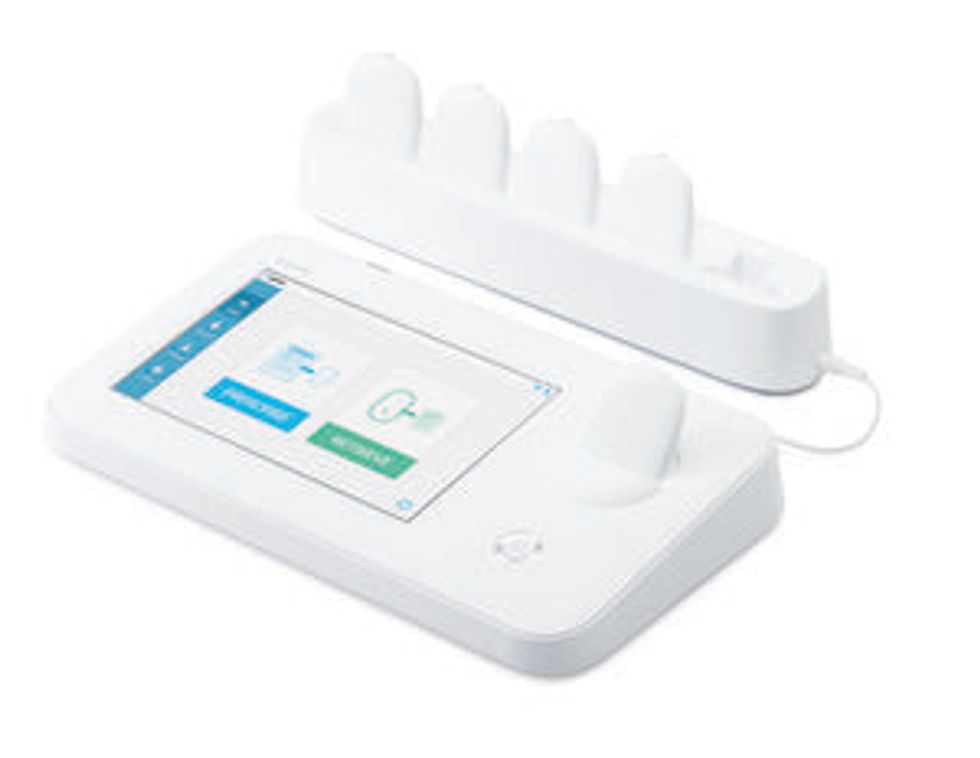
A doctor’s prescription for clinical depression could one day sound like this: In the comfort of your own home, slip on a brain-zapping headband a few times per week. For 20 minutes, send a tiny stream of electricity through your brain.
The treatment would be delivered by a user-friendly type of brain stimulation called tDCS (transcranial direct current stimulation), which has recently become a hot topic in neuroscience research. Now it’s beginning to make the transition from lab to doctor’s office. And a South Korean startup called Ybrain thinks its stylish tDCS headband, specifically designed to treat depression, will be the product that brings tDCS into the clinical mainstream.
Ybrain founder and CEO Kiwon Lee made his pitch earlier this month at the NYC Neuromodulation conference, where doctors and researchers working on the cutting edge of brain stimulation met to compare notes and chart the field’s progress. When his turn came at the podium, Lee predicted that his device will receive regulatory approval in Korea this March. He also laid out his very optimistic game plan for worldwide market domination.
Lee says Ybrain will roll out the device in 70 Korean hospitals this year to reach thousands of patients with clinical depression. The company will use data from all those patients to build a case for approval in Europe, Lee says, and then in the United States, where the regulatory requirements are most stringent. “After one device is approved by the FDA, it will be seen as a mainstream treatment,” he says.
Ybrain’s tDCS device may not be first to make it to the all-important American market; the New York-based Soterix Medical has a tDCS system for depression that’s already approved for clinical use in Europe, and the company is pursuing FDA approval now. But the European patients using the Soterix device are mostly using it in the clinic under a doctor’s supervision. Ybrain hopes to make its at-home treatment the norm.
TDCS is seen as an exciting new type of treatment in part because the gear is cheap, portable, and easy to use. The systems use electrodes placed at particular locations on the scalp to channel a few milliamps of current through a specific brain region, and they can be powered by a 9-volt battery. Last year there were nearly 700 papers published about tDCS, with studies on every topic imaginable: Researchers experimented with physical rehab for stroke patients, memory improvement for people with Alzheimer’s, and craving reduction for smokers and alcoholics.
It’s so easy to set up a tDCS stimulator that DIYers are making their own rigs, and startups are selling products directly to consumers as tools to enhance mental focus or improve athletic performance.
Yet scads of scientific questions must still be answered. The NYC conference revealed the experts’ consensus that tDCS can definitely alter the brain in meaningful ways—but they don’t know which medical conditions will truly benefit from these alterations. The journal Clinical Neurophysiology just published a comprehensive review of the evidence on tDCS effectiveness across a range of disorders; for most it concluded that there haven’t yet been enough rigorous trials to draw conclusions. However, the article singled out tDCS treatment for depression as “probably effective.”
Depression might be the first killer app for tDCS. At the NYC conference, attendees heard from Andre Brunoni, a neuroscientist and psychiatrist at the University of Sao Paulo who coauthored a 2013 study comparing tDCS to a standard antidepressant medication. Interestingly, that study found the electric and pharmaceutical therapies to be equally effective, and found that patients who received both treatments together fared best of all.
Brunoni says he’s not quite ready to forge ahead with tDCS in his clinical practice; he’s still waiting for evidence on many crucial questions that will define tDCS’s practical use. “We don’t know the best ‘dose’ to use for tDCS,” he says. “What’s the best amount of current to use? What’s the optimal number of sessions? There are a lot of parameters to explore.”
But Brunoni also tells IEEE Spectrum that tDCS may offer advantages over antidepressants as well as transcranial magnetic stimulation (TMS), which requires clinics to invest in an expensive machine and patients to travel to the clinic for each treatment session. He mentions three types of potential benefits:
- Price: Because tDCS is relatively cheap, Brunoni says, private medical plans would likely prefer it to more expensive treatments like TMS.
- Ease of use: Brunoni says that companies such as Soterix and Ybrain “are developing very safe and reliable devices that would allow home-use with almost zero risk.”
- Short-term treatment: TDCS treatment for depression would likely start with an intense phase of treatment—for example, two weeks of daily sessions—followed by less frequent “maintenance” stimulation sessions. Brunoni calls this a big advantage over antidepressant drugs, which require treatment for at least 9 months and can cause a host of unpleasant side effects throughout that time (like weight gain and sexual problems).
In South Korea, Ybrain is betting that these benefits and its slick consumer-friendly design will speed adoption of its device. “It’s designed for home use,” says Ybrain CEO Lee, “so physician can electronically prescribe the device and patients can bring it to their homes.”
To make the medical gadget easy to use and failsafe, designers kept it simple. "The device has just a power button, no interface,” Lee says. “The patient turns it on and puts the device on their head, and it automatically starts the stimulation based on the doctor’s prescription.” So if the doctor prescribed one 20-minute session of 2 milliamps per day, the gadget wouldn’t allow more than the prescribed limit.
The Ybrain system comes with a smartphone app that regularly asks the patient to rank their mood on a depression scale, and sends this info to their doctor along with device usage data. These features enable the doctor to monitor the treatment’s effects. Not incidentally, this setup also enable Ybrain to collect efficacy data the company can use when it applies for regulatory approval in Europe and the United States.
While the NYC Neuromodulation meeting showed that Ybrain isn't alone in having high hopes for tDCS depression treatments, there were also cautionary notes. In one panel discussion the distinguished neurologist and psychiatrist Mark George, who directs the Brain Stimulation Laboratory at the Medical University of South Carolina, said he’s not yet ready to prescribe tDCS to his clinically depressed patients.
George invokes the Hippocratic Oath: “First, do no harm.” While there’s ample evidence that tDCS is safe when used correctly, George notes that physical safety isn’t the only consideration. Depressed patients who come to see him are desperate, despondent, and looking to him for salvation.
“Hope is fragile,” George says, “and you want to be very careful with what you throw out there for your patients. You want to go with your best shot. And right now, I don’t see tDCS as your best shot.”
This article appears in the March 2017 print issue as “At-Home Electric Headband for Depression Could Go Mainstream.”
Eliza Strickland is a senior editor at IEEE Spectrum, where she covers AI, biomedical engineering, and other topics. She holds a master’s degree in journalism from Columbia University.