Doctors are typically conservative about new technologies, and often skeptical when engineers promise that their gear will revolutionize health care. But when pediatrician Amy Paller heard a conference talk about flexible, wireless, stick-on biosensors that could be used on infants, her first thought was: “Wow.”
“I thought, this could be transformative to medical care for babies,” says Paller, an expert in pediatric dermatology at Northwestern University’s Feinberg School of Medicine. Her second thought was, “I can help make this happen,” she tells IEEE Spectrum.
The conference talk was by Northwestern professor John Rogers, the founder of the university’s Center for Bio-Integrated Technologies. Rogers has been working for years on stretchable and bendable electronics that stick to the skin (see some of Spectrum’s coverage here).
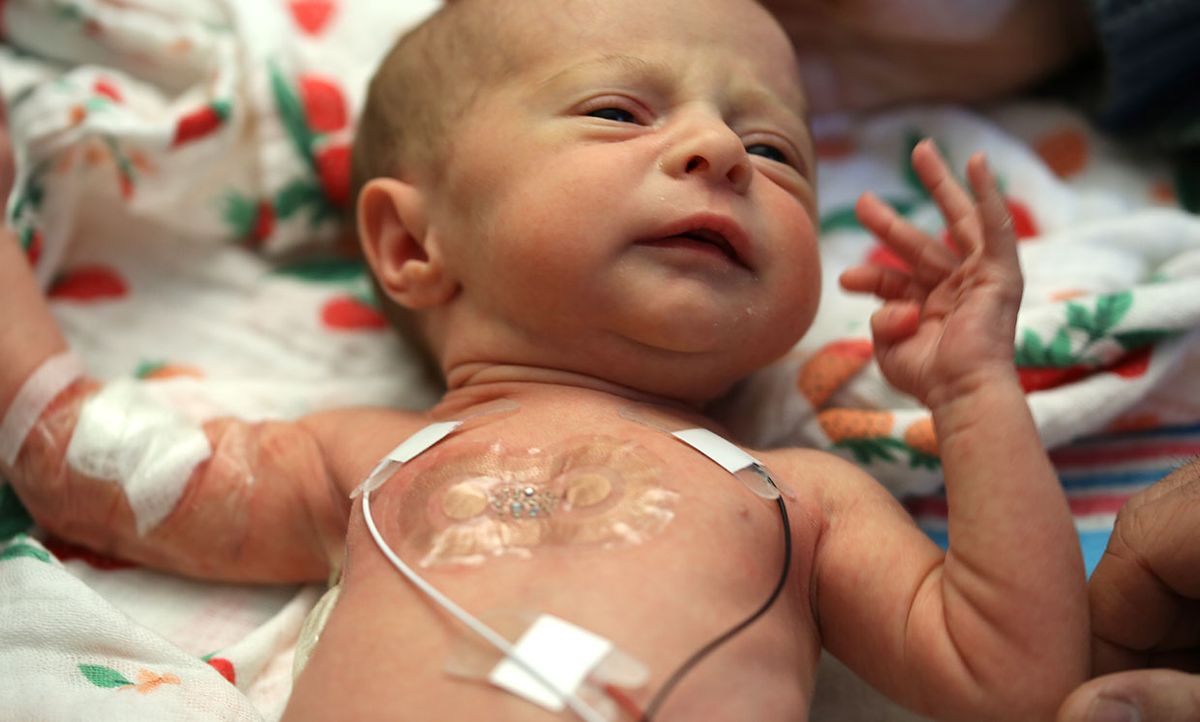
The latest application of his technology, described today in Science, is wireless biosensors for fragile infants in the neonatal intensive care unit (NICU). Rogers and Paller worked together to develop stick-on sensors that can safely adhere even to the underdeveloped skin of premature infants, and which provide continuous vital sign readings superior to those furnished by today’s standard monitors.
In today’s standard NICUs, the tiny babies are wired up to a host of sensors to measure things like heart rate, temperature, and blood oxygenation levels. Getting rid of the wires helps with both the practical aspects of care and the medical outcomes for the babies, the researchers say.

The practical benefits would be obvious on a daily basis. Nurses need to lift the babies for routine tasks like diaper changes, and “these wires are always in the way,” Paller says.
Going wireless also makes it much easier for parents to pick up and cuddle their delicate newborns, and for mothers to breastfeed them. “All those wires tether the baby to the bed, and they’re definitely intimidating for the parents,” Paller says. “It’s hard to hold the baby, let alone do skin-to-skin contact.”
Putting a naked (well, usually diapered) baby directly on a parent’s bare chest, a practice sometimes called “kangaroo care,” has proven to have a wide range of benefits: It helps with bonding, improves breastfeeding, helps stabilizes the newborn’s heart and respiration rates, and seems to improve brain development and decrease the incidence of disease. “It really translates into improved health and survival for these babies,” Paller says.
The engineering challenges involved in designing biosensors for newborns were significant, says Rogers. “The skin is very fragile, so the sensors need to maintain robust skin contact with very gentle adhesives,” he tells IEEE Spectrum. The key, he says, is to make the electronics so flexible that they conform entirely to the skin, and don’t need a strong adhesive to hold them on. The electronics also had to be fully encased, so they wouldn’t be damaged during baby baths, and compatible with X-ray and MRI machines.

The final system he devised is composed of two sensors, one that sticks to the chest to record electrocardiograms (providing heart rate), another that sticks to the foot to record photoplethysmograms (measuring blood oxygenation) and skin temperature. The foot sensor required the engineering team to create software that could compensate for movement artifacts in the data: “These babies kick a lot,” Rogers notes.
Time-syncing these two sensors also provides a continuous measurement of blood pressure; the system knows when the heart pumps out a pulse of blood and when it arrives at the foot, and that time measurement correlates well with blood pressure. “In that way, we can go beyond the clinical standard measurements done today,” Rogers says. Today, continuous monitoring of blood pressure requires an invasive arterial probe.
The sensors use near-field communication (NFC) to connect to a module that can be attached to the baby’s bed, and which both receives the data and sends wireless power to the sensors. That module transmits the data via bluetooth to a mobile phone or tablet.
The researchers have tested the technology in clinical trials in two NICUs in Chicago. They’re expanding their tests to other hospitals while simultaneously beginning to pursue FDA approval for their system, which would make it commercially available.
To make mainstream adoption possible, Rogers says his team has been adapting the handcrafted devices described in the Science paper, which were “produced in a lab using sophisticated instruments and processes,” he says. They’ve now designed a version that’s suitable for consumer electronics manufacturing processes, he says. “We know now how to build thousands of devices.”
They’ll soon test that capacity, as they have a mandate to produce 20,000 devices that will be tested in India, Pakistan, and Zambia by the end of 2019. Rogers received funding from the Bill & Melinda Gates Foundation and Save the Children to test the technology in developing world clinics, to determine if both the hardware and software function in those settings. Rogers says he’s hopeful that the technology will be affordable; he currently estimates manufacturing costs at $20 per unit.
While the economics seem favorable and the medical benefits seem clear, Paller says she’s well aware of the difficulty of translating a great idea into clinical practice. But she thinks hospitals will be keen to improve on the status quo. “It’s so devastating to new parents to see their baby surrounded by all the the wires and beeping monitors, and then to not even be able to hold their baby,” she says. “That’s something we really hope to change.”
Eliza Strickland is a senior editor at IEEE Spectrum, where she covers AI, biomedical engineering, and other topics. She holds a master’s degree in journalism from Columbia University.