19 September 2012—When utilities burn fossil fuels to produce electricity, roughly two-thirds of the energy in the feedstock is lost as waste heat. As far back as the preparation for the first moon landing, researchers have been working to efficiently convert waste heat into electrical energy. But so far, the conversion efficiency of thermoelectric materials has been way too low to yield a commercially viable device. This week, however, researchers at Northwestern University, in Evanston, Ill., report in Nature that they have smashed through the old efficiency record, and they claim that further improvement to efficiencies previously thought impossible are within reach.
Materials scientists have long known that in order to improve the performance of a thermoelectric material, they must inhibit its thermal conductivity. For decades they were able to reach but not surpass the long-standing figure of 1 ZT. (ZT, the figure of merit for thermoelectric energy conversion, refers in this case to the amount of electricity generated for a given amount of thermal energy applied.) They did so by replacing certain atoms in the thermoelectric materials’ crystal lattice. This doping created enough atomic-scale disorder to disrupt the movement of phonons, the waves that carry thermal energy.
Then in 2004, Mercouri G. Kanatzidis’s group at Northwestern University realized that they were disrupting only the phonons with short wavelengths. By arranging the placement of these atomic-scale lattices into nanometer-scale structures, they were able to interrupt medium-wavelength phonons too and boost ZT as high as 1.8.
But Kanatzidis’s goal had been 2 ZT.
In the research reported this week, his lab finally surpassed that goal. The breakthrough came when Kanatzidis’s group figured out how to disturb the propagation of long-wavelength phonons across the semiconductor lead telluride, dramatically improving its conversion efficiency. They did so by fine-tuning the grain boundaries in the material at the so-called mesoscale, up to about 1 micrometer. Disturbing the flow of short-, medium-, and long-wavelength phonons—and the addition of just the right amount of sodium into the material’s matrix—resulted in a ZT of 2.2 at 642 °C. The researchers predict that based on this level of improvement, a thermoelectric device with a cold-side temperature of 77 °C and a hot-side temperature of 677 °C will convert roughly 20 percent of waste heat into electricity.
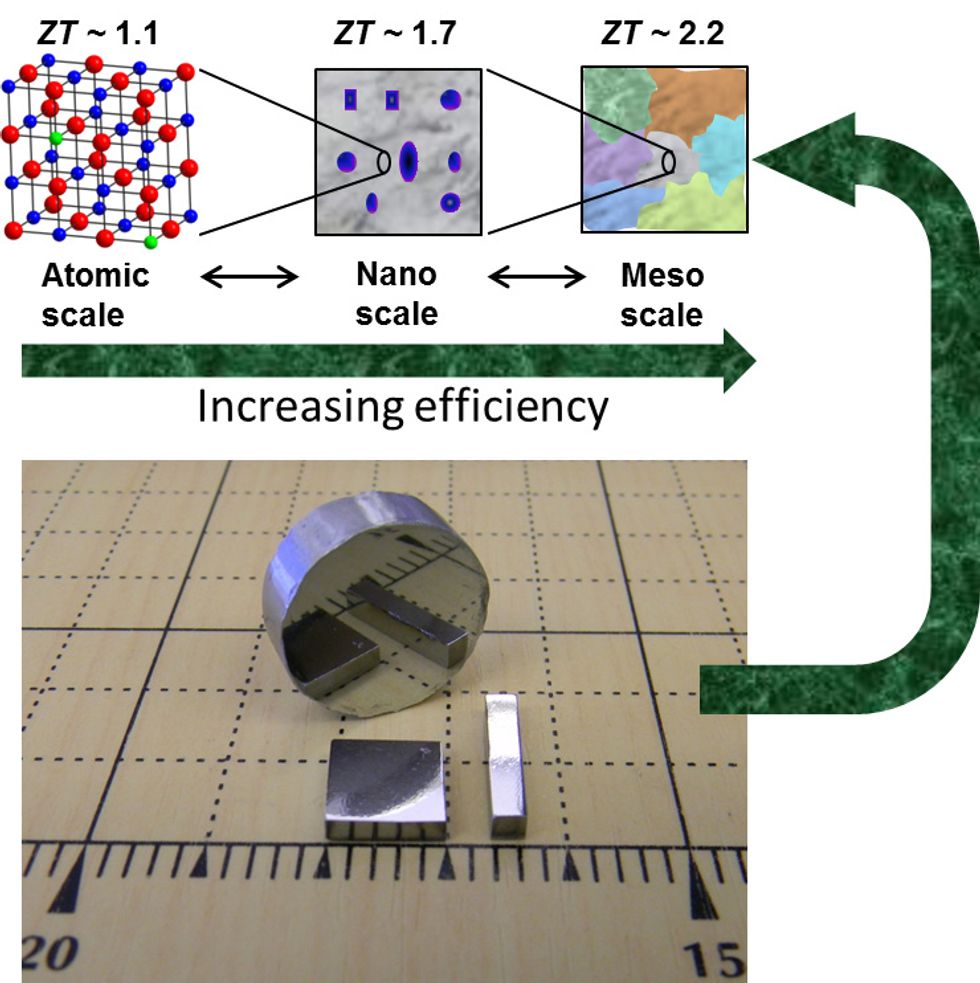
A good analogy is a chocolate chip cookie, says Kanatzidis, who is a professor of chemistry. The field “got to 1 ZT by refining the formulation of the chocolate chips. That is the atomic scale. More recently, we improved things by determining where on the cookie the chips should be placed. That is the nanoscale that improved the state-of-the-art to about 1.8 ZT. And here, now we have adjusted the size of the cookie and how it is stacked in the package,” says Kanatzidis.
“The brilliance of [the Northwestern team's] study of lead telluride is that they have [shoehorned] almost every known strategy for optimizing thermoelectric materials into one system,” writes Tom Nilges, a chemistry professor at the Technische Universität München in Garching, Germany, in the News & Views article accompanying the Nature paper. Nilges describes the Northwestern group’s technique with another analogy: “The embedding of progressively smaller subunits within the material reminds me of matryoshka (Russian) dolls.”
Because it took so long to break the 1-ZT barrier, it was not immediately evident that 2 ZT was reachable, says Kanatzidis. But now, having shown that it is achievable, the goalposts have moved to 2.5 ZT, and eventually 3 ZT, he says.
“I am sure that the authors’ findings will trigger exponential progress in the performance of thermoelectric materials,” writes Nilges.
The Northwestern researchers note that their multiscale approach is applicable to all bulk thermoelectric materials, not just lead telluride. In addition to that semiconductor, they are working with lead selenide and lead sulfide because of concerns about the availability of tellurium, a by-product of copper production.
