Silicon, graphene, and sometimes the two of them combined together have all been suggested as potential replacements for graphite in the electrodes of lithium-ion batteries.
While all three of these options bring attractive properties to the table—most importantly, a very high theoretical capacity—those properties are lost in the real world. Silicon electrodes crack and break after just a short number of charge/discharge cycles. Meanwhile, the use of graphene on electrodes is limited because graphene’s attractive surface area is only possible in single stand-alone sheets, which don’t provide enough volumetric capacitance. Layer the graphene sheets on top of each other to gain that volumetric capacity, and you begin to lose that attractive surface area.
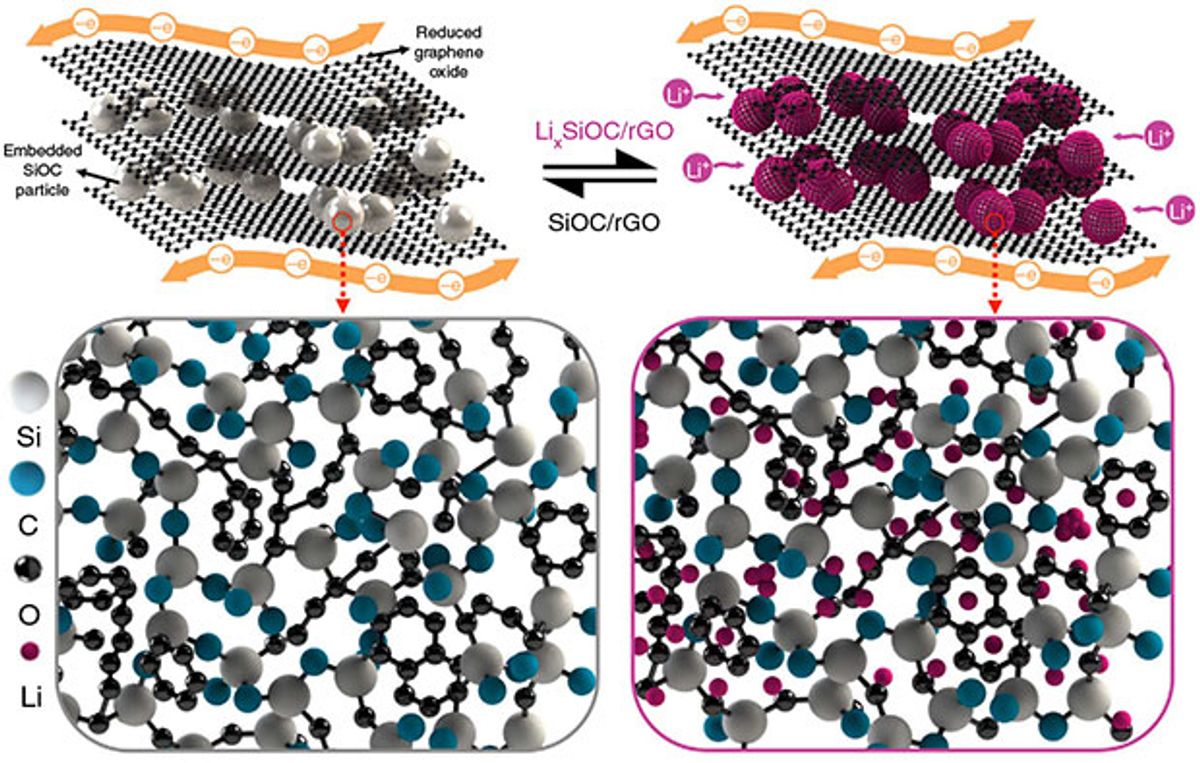
Now researchers at Kansas State University (KSU) claim to have developed a technique that uses silicon oxycarbide that makes the combination of silicon and graphene achieve its expected greatness as an electrode material.
“Silicon combined with graphene is better than a bulk silicon electrode,” explained Gurpreet Singh, an associate professor at KSU and one of the researchers, in an e-mail interview with IEEE Spectrum. “However, nano-silicon/graphene electrodes fail to satisfy key requirements for any practical applications.” Among other things, they have poor volumetric capacity, high cost, and low cycling efficiency—too much lithium is lost irreversibly with each charge-discharge cycle. What’s more, their mechanical and chemical instability that can lead to rapid capacity decay.
To overcome this, the KSU researchers turned to the high temperature glass ceramic, silicon oxycarbide. In research described in the journal Nature Communications, the KSU team created a self-standing anode material consisting of silicon oxycarbide glass particles embedded into a chemically modified graphene oxide matrix.
A heated silicon resin decomposes so that “the constituent silicon, carbon, and oxygen atoms are arranged in a random 3-D structure, and any excess carbon precipitates out into string-like or cellular regions. Such an open 3-D structure renders large sites for reversible lithium storage and smooth channels for solvated lithium-ion transportation from the electrolyte.”
This stands in stark contrast to crystalline silicon, which undergoes an alloying reaction with lithium that results in enormous volume changes and also an irreversible reaction with the electrolyte that leads to chemical instability and fading capacity as the charge-discharge cycles add up.
The KSU researchers claim that the electrode has a capacity of approximately 600 miliampere-hour per gram or 400 miliampere-hour per cubic centimeter of the electrode after 1020 cycles. The researchers expect that the power density (the maximum amount of power that can be supplied per unit mass) will be more than three times that of today’s Li-ion batteries.
In future research, the KSU team aims to produce electrode materials with larger dimensions. As a benchmark, the researchers are looking at today’s pencil cell battery that uses a graphite-coated copper foil electrode, which is more than 30 cm in length.
Singh added: “We are also looking at batteries as structural materials, such as load bearing batteries that can be charged and discharged while under dynamic loads.”
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.