Researchers at the U.S. Department of Energy’s SLAC National Accelerator Laboratory have leveraged a long-used microscopy tool to investigate why perovskites, the “wonder materials” in photovoltaics, have proven to be so efficient at converting light into electricity. These studies have revealed for the first time that light essentially spins the atoms inside of perovskites into a whirl, offering new clues into how scientists can make these materials even more efficient at converting light into electricity.
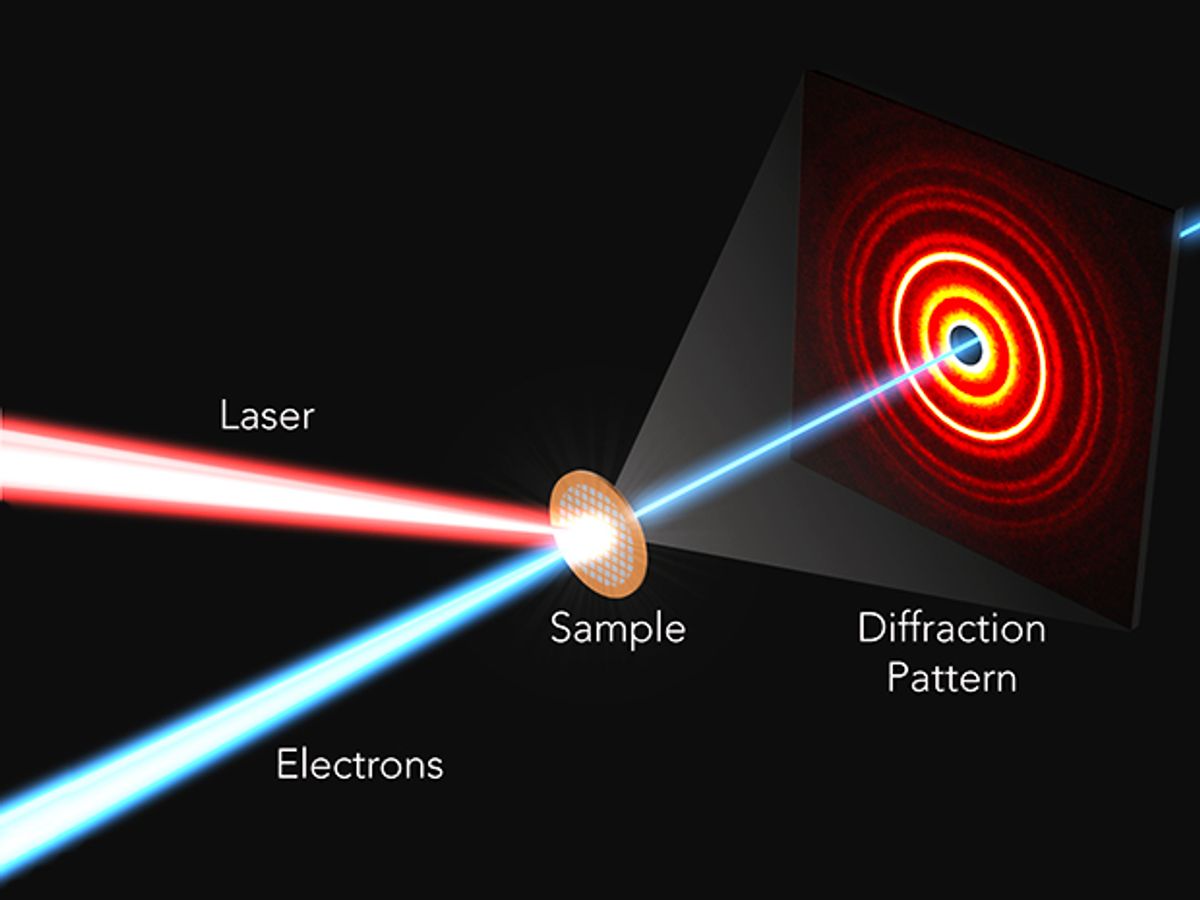
In research described in the journal Science Advances, the SLAC scientists employed the decades-old microscopy technique known as ultrafast electron diffraction (UED) to create a kind of movie of how atoms in perovskites respond within a trillionth of a second of being hit by short pulses of light.
The UED technique depends on the fact that electrons have wavelengths much smaller than the spacing between atoms in materials. By probing the interference patterns associated with how the electrons bounce off these atoms, one can effectively reconstruct and visualize the atomic-scale structure of materials.
“Because the electrons are confined within ultrashort pulses, they provide a snapshot of this process at a controllable time after a photon is absorbed,” said SLAC researcher Aaron Lindenberg in an e-mail interview with IEEE Spectrum. “By taking many of these snapshots, one can put these together and make a kind of a movie showing what the atoms are doing when a solar cell based on the hybrid perovskites is operating.”
While UED has been around for decades, the facilities at SLAC offer what is known as a relativistic device, meaning the electrons in the beam are traveling much closer to the speed of light than in traditional UED devices. This means that you can put more electrons into each femtosecond-long laser pulse, which in turn makes it possible to probe more complex, disordered, or weakly scattering structures, according to Lindenberg.
Hybrid perovskites have made a huge impact on materials scientists’ ability to investigate new materials for solar cells and other optoelectronic devices. The architecture common to many of the perovskites investigated for solar cell applications is a hybrid of iodine, lead, and an organic molecule called methylammonium. The iodine atoms are arranged in octohedra, eight-sided structures that look like two pyramids joined at their bases. The lead atoms reside inside the octohedra and the methylammonium molecules lie between the octohedra. However, to understand unique functionality of this architecture and to develop related materials, researchers need to understand what the atoms are doing when is in operation.
While previous studies have examined the role of the methylammonium ions and their motions in transporting electric charge through the material, the SLAC researchers discovered that light causes large deformations in the network of lead and iodine atoms. This, they suspect, could be crucial for the efficiency of perovskites.
“We think these ultrafast processes play a central role in determining the efficiencies of a solar cell based on these materials and this in turn requires a source like UED to enable this real-time measurement,” says Lindenberg.
While Lindenberg concedes that it is possibly too early to know how these results will impact the development of perovskites for photovoltaics, these most recent results show that very large deformations of certain atoms (the iodines) occurring on ultrafast time-scales may play a central role in the ultimate efficiency of devices based on these materials.
Lindenberg added: “Coupling between these experimental efforts, theoretical efforts, and synthetic efforts, we think, will enable the engineering of new kinds of materials for photovoltaic applications and new fundamental understanding of how these materials operate.”
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.