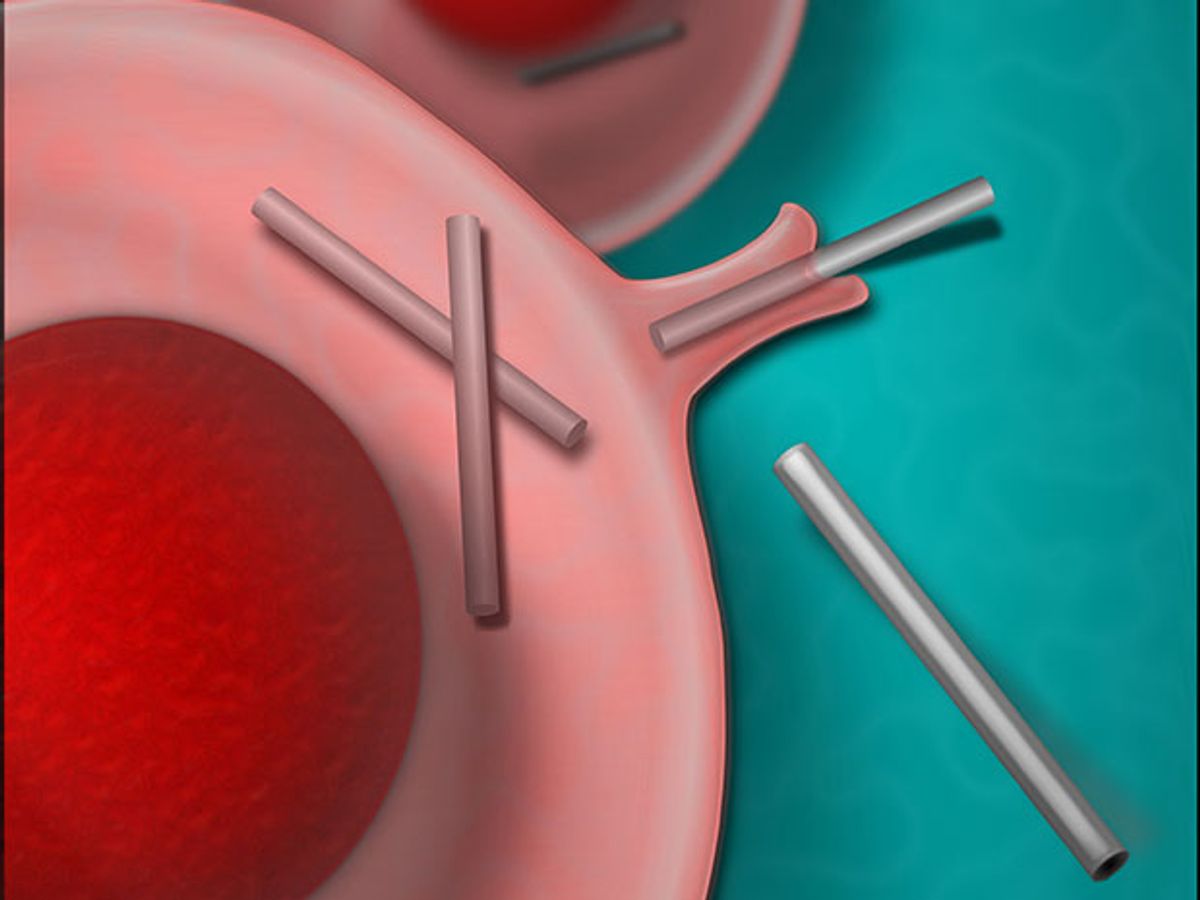
Drop silicon nanowires and human cells into the same petri dish, and you’ll see something extraordinary: The cells will eat the nanowires. Researchers at the University of Chicago recorded the phenomenon, and reported it Friday in the journal Science Advances.
The group aims to harness that knowledge to develop a new means of drug delivery or bioelectronic therapy. “We want to do something that is special,” says Bozhi Tian, a materials scientist at the University of Chicago. “We’re trying to develop a bioelectronic device that is intracellular” and can control individual parts of the cell, he says.
Silicon nanowires are biocompatible, highly conductive, and so thin they are essentially one-dimensional. Inside human cells, they could potentially be used to do a lot of things. They could record the electrical communication between structures inside the cell—signals passed from one organelle to another. They could electrically stimulate those organelles for therapeutic purposes.
Or the nanowires could carry small molecule drugs and deliver them directly to cells, bypassing some of the body’s natural barriers. The fact that the cells consume the nanowires naturally, without any kind of special treatment, and without damaging themselves, makes it that much more useful.
But before Tian’s group can turn the phenomenon into a tool, they must first understand how exactly a cell will eat a piece of silicon nanowire, and what happens to it once it’s inside the cell. That’s what the group did in today’s report.
Using both an electron microscope and a specialized optical imaging tool designed by the team, the group recorded the eating of the nanowires in detail. It appears that the cell’s outer membrane folds itself like a pocket, grabs the nanowire, and envelops it in a membrane-lined bubble. The process is called phagocytosis; it’s the same method used by immune cells to grab a bit of bacteria and swallow it up.
Once the nanowire is inside, the cell’s machinery then shuttles it through its system with sudden bursts of speed—up to 99.4 nanometers per second—and deposits it just outside the cell’s nucleus. Tian’s group made a video of the process (complete with melodramatic accompaniment).
Cellular Uptake of Silicon Nanowires Might Be the Key to Merging Electronics With Biologyyoutu.be
The specialized tool they designed to observe the cells, called a scatter-enhanced phase contrast, or SEPC, enabled them to simultaneously see not only the cellular components, but also the inorganic nanowires. John Zimmerman, who was a graduate student in Tian’s lab and is now a postdoc working with Kit Parker at Harvard, developed the tool. It uses phase contrast imaging for observing the cell and dark-field imaging for observing the nanowires.
Tian’s group observed thousands of nanowire-cell interactions, mostly in human endothelial cells, which are the cells that line the interior surface of blood vessels. Endothelial cells are known to be stubborn about taking in pharmaceutical drugs—they essentially form a biological barrier along blood vessel walls. But in Tian’s experiment, the endothelial cells took in 96% of the nanowires.
That brings hope that pharmaceutical molecules that normally couldn’t get into endothelial cells will now have a new route: on the backs of silicon nanowires. The team also envisions using the tool like optogenetics, where cells are controlled using light. “We can imagine having cells internalize light-responsive nanowires, and then using these properties to influence cell behavior,” says Zimmerman.
The tool also gives researchers a way to electrically stimulate inside a cell in precise ways—a new spin on a decades-old area of research. Most people working on electrical stimulation are experimenting with devices that send pulses of electricity to larger systems in the body—whole nerve circuits, for example. The most advanced groups are targeting a narrower set of nerve fibers within a nerve. Tian’s approach would mean stimulating in even more precise ways, by sending electrical pulses to particular organelles within a cell.
There are a number matters to address before those applications can be realized. For example, in addition to endothelial cells, Tian’s team tried the experiment in smooth muscle cells, neurons and cardiac cells. The latter two cell types rejected the nanowires, largely because they don’t have the phagocytosis mechanism that endothelial cells do. But with some surface modifications, those cells might also capable of swallowing nanowires, Tian says.
Adds Zimmerman: “While we still have a ways to go...by understanding how cells ‘eat’ these nanowires, we can really start to think of how we can actually make these applications a reality.”
Emily Waltz is a features editor at Spectrum covering power and energy. Prior to joining the staff in January 2024, Emily spent 18 years as a freelance journalist covering biotechnology, primarily for the Nature research journals and Spectrum. Her work has also appeared in Scientific American, Discover, Outside, and the New York Times. Emily has a master's degree from Columbia University Graduate School of Journalism and an undergraduate degree from Vanderbilt University. With every word she writes, Emily strives to say something true and useful. She posts on Twitter/X @EmWaltz and her portfolio can be found on her website.