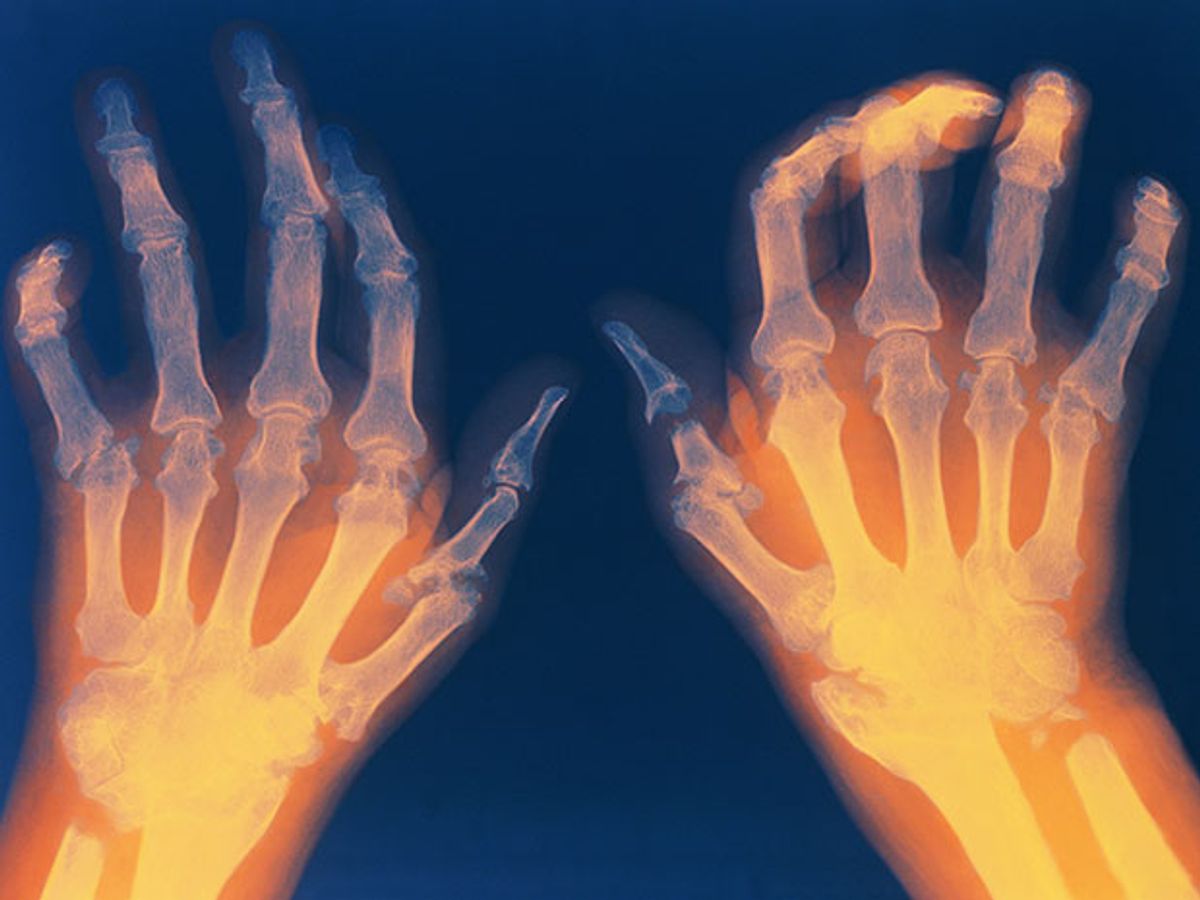
By selectively stimulating nerve fibers running from the brain to the spleen with electricity, researchers have successfully treated a small group of patients with rheumatoid arthritis. Importantly, the team showed the exact mechanism by which the procedure works: Vagus nerve stimulation activates immune system cells to inhibit the production of key inflammatory proteins, called cytokines, implicated in the disease.
“We wanted to know if we could selectively block cytokines in humans with electrons. The answer is yes,” says study co-author Kevin Tracey, president and CEO of the Feinstein Institute for Medical Research in New York and co-founder of SetPoint Medical Corporation, which funded the study published this month in the Proceedings of the National Academy of Sciences. “We began with the mechanism and ended with the mechanism.”
Using mild pulses of electricity to treat disease, so-called “bioelectronic medicine” or “electroceuticals” is not new, but knowing exactly how it works in the body for a particular disease is a step forward. Many medical devices are approved and put to use without knowledge of what downstream molecular events they cause in the body.

Vagus nerve stimulation is an approved treatment for both epilepsy and depression. The vagus nerve, which runs down the neck and to major organs, is a super-highway bundle of over 100,000 nerve fibers. Four of its five “lanes”—about 80 percent of the fibers—run information from the body up to the brain. In addition to epilepsy and depression, researchers have been exploring how to activate those fibers to treat heart failure, stroke, migraine, and numerous other ailments.
That fifth lane of nerve fibers carries information away the brain to the body, including about 4,000 fibers that travel to the spleen. In 1998, Tracey sketched an idea for a human clinical trial involving the fifth lane on a napkin. His lab had just discovered that stimulating the vagus nerve activated cells in the spleen to produce acetylcholine, a neurochemical with functions in the brain and in muscles. In the spleen, acetylcholine decreases the ability of nearby immune cells to produce inflammatory molecules called cytokines, such as tumor necrosis factor (TNF) and interleukin-1.
This month, that napkin brainstorm finally comes to fruition. Tracey, collaborator Paul-Peter Tak from GlaxoSmithKline and the Academic Medical Centre/University of Amsterdam, and other colleagues successfully treated a small group of patients with rheumatoid arthritis by stimulating those 4,000 efferent fibers in the vagus nerve. The fibers can be specifically targeted because they respond to lower thresholds of electricity than other fibers in the nerve.
Conventional therapy had failed in all 18 patients who participated in the trial, including some who had taken anti-TNF drugs. For the treatment, doctors surgically implanted a 2.5 mm electrical lead into the neck of each patient, touching the vagnus nerve, and attached the lead to a small pulse generator. After 42 days of treatment—turning on the device for 60 seconds, from one to four times daily—the patients had significantly reduced levels of TNF in their blood compared to baseline levels. When the stimulator was turned off for two weeks, those levels of TNF began to creep back up; when it was turned back on, they once again plummeted. In addition to TNF, the stimulation also decreased the production of other inflammatory proteins in the body, including interleukin-6 and interleukin-1.
This isn't the first evidence that VNS can help treat rheumatoid arthritis: In 2012 the team published a pilot study of 8 patients with rheumatoid arthritis, and this new publication includes an additional 9 patients.
Although it was a small study, many of the patients reported significant improvements in their symptoms, and no serious side effects were reported. Each patient was offered the option to have the device removed at the end of the study. None took up the offer.
The fact that the device only needs to be turned on up to 15 minutes per day—its active much more frequently for patients being treated for epilepsy or depression—is a valuable engineering feature, says Tracey, as the device won’t need much power. “We envision devices from SetPoint that are implanted once per life. There won’t be battery changes. That’s huge for a patient,” he notes.
The study also demonstrates what should be a common goal for medical devices—knowing the molecular mechanism by which they treat disease, says Tracey. By knowing how VNS improves rheumatoid arthritis symptoms, he says, “we know enough about the target and the pathway that we should be able to keep moving forward fast.”
In addition to moving ahead in rheumatoid arthritis, SetPoint is running a clinical trial testing VNS in patients with Crohn’s disease, an autoimmune disorder of the bowl that also involves TNF and other inflammatory cytokines. In 2010, an independent team in France showed that VNS improved inflammatory bowel disease in rats.
Megan Scudellari is an award-winning freelance journalist based in Boston, Massachusetts, specializing in the life sciences and biotechnology. She was previously a health columnist for the Boston Globe and has contributed to Newsweek, Scientific American, and Nature, among others. She is the co-author of a college biology textbook, “Biology Now,” published by W.W. Norton. Megan received an M.S. from the Graduate Program in Science Writing at the Massachusetts Institute of Technology, a B.A. at Boston College, and worked as an educator at the Museum of Science, Boston.