Bubble Power
Tiny bubbles imploded by sound waves can make hydrogen nuclei fuse—and may one day become a revolutionary new energy source

For more than half a century, thermonuclear fusion has held out the promise of cheap, clean, and virtually limitless energy. Unleashed through a fusion reactor of some sort, the energy from 1 gram of deuterium, an isotope of hydrogen, would be equivalent to that produced by burning 7000 liters of gasoline. Deuterium is abundant in ocean water, and one cubic kilometer of seawater could, in principle, supply all the world’s energy needs for several hundred years.
So why haven’t we built any such reactors? Basically, because after spending billions of dollars on research, we have yet to identify an economically viable fusion-reactor technology that can consistently produce more energy than it consumes. Today, researchers are using enormous lasers or powerful magnetic fields to trigger limited fusion reactions among deuterium and other hydrogen isotopes. Results are promising and yet still modest—and so the challenge remains.
For several years our research groups—at Purdue University in West Lafayette, Ind.; Rensselaer Polytechnic Institute in Troy, N.Y.; and the Russian Academy of Sciences branch in Ufa—have been working on a new way to create fusion reactions. By applying sound waves to a deuterium-rich liquid, we create pressure oscillations that implode tiny bubbles filled with deuterium vapor. The bubbles’ violent collapse can cause some of the deuterium nuclei to undergo fusion.
It is hard to imagine that mere sound waves can possibly produce in the bubbles, even briefly, the extreme temperatures and pressures created by the lasers or magnetic fields, which themselves replicate the interior conditions of stars like our sun, where fusion occurs steadily. Nevertheless, three years ago, we obtained strong evidence that such a process—now known as sonofusion—is indeed possible.
Since then, we have been working to improve and scale up our apparatus, investigating the possibility that it can produce a sizable surplus of energy. If this proves possible—and it’s still a big “if”—sonofusion could become a revolutionary new energy source.
To explore this enticing possibility, early this year our research team joined forces with others to create the Acoustic Fusion Technology Energy Consortium, or AFTEC. Its five founders are Boston University; Impulse Devices Inc. in Grass Valley, Calif.; Purdue University; the University of Mississippi in Oxford; and the University of Washington in Seattle. Its goal is to promote the development of sonofusion and its related science and technology.
The Idea of Sonofusion (technically known as acoustic inertial confinement fusion) was derived from a related phenomenon, sonoluminescence, which has been known for more than 70 years. In sonoluminescence, a process widely used by chemists and also a science-fair staple, loudspeakers attached to a liquid-filled flask send pressure waves through the fluid, exciting the motion of tiny gas bubbles. The bubbles periodically grow and collapse, producing visible flashes of light that last less than 50 picoseconds.
About 20 years ago, researchers studying these light-emitting bubbles speculated that their interiors might reach such high temperatures and pressures that they could trigger fusion reactions. Since then, several groups have been trying to achieve fusion using sound waves, most of them with a kind of enhanced sonoluminescence. This method, called single-bubble sonoluminescence, involves a single gas bubble that is trapped inside the flask by a pressure field and yields light flashes during repetitive implosions.
Our own efforts began in 1996 at the Oak Ridge National Laboratory in Tennessee [see photo, "Bubble Maker”]. In those first attempts, we tried many different configurations for single-bubble sonoluminescence, all without success. We finally concluded that excitation pressures higher than about 170 kilopascals would always dislodge the bubble from its stable position and disperse it in the liquid. That, we thought, was a fundamental problem for single-bubble sonoluminescence, because we calculated we would need at least 10 times that pressure level to implode the bubbles strongly enough to trigger thermonuclear fusion.
To overcome that limitation, we began seeking a different approach. After a lot of brainstorming and many experiments, we came up with a promising idea: remove virtually all the naturally occurring gas bubbles dissolved in the liquid and then, somehow, create our own bubbles, much smaller, precisely when we needed them. That way, we could increase the bubbles’ maximum size before their collapse, thereby tremendously increasing the implosion’s energy. We easily removed the gas from the liquid by attaching a vacuum pump to the flask and acoustically agitating the liquid. But then how could we create the bubbles we needed? We owe our success—as often occurs in science—to some fortunate happenstance, perhaps more than we had any right to expect.
The idea came from one of our colleagues, who was then working on a largely unrelated project. The colleague was trying to use a sonoluminescence flask as a neutron detector. To test his idea, he fired high-energy neutrons at the flask and then analyzed the light emissions. Upon learning about that, we figured we could do the same thing, not to produce light but to create tiny vapor bubbles that we could later grow and implode.
Our apparatus has evolved since those first experiments in 1996, but it continues to be relatively simple [see illustration, "How Sonofusion Works"]. It consists of a cylindrical Pyrex glass flask 100 millimeters high and 65 mm in diameter. We first attach a lead-zirconate-titanate ceramic piezoelectric crystal in the form of a ring to the flask’s outer surface. This piezoelectric ring works like the loudspeakers in a sonoluminescence experiment, although it creates much stronger pressure waves. When a positive voltage is applied to the piezoelectric ring, it contracts; when the voltage is removed, it expands to its original size.
We then fill the flask with commercially available deuterated acetone, in which 99.9 percent of the hydrogen atoms in the acetone molecules are deuterium (this isotope of hydrogen has one proton and one neutron in its nucleus). The main reason we chose deuterated acetone is that atoms of deuterium can undergo fusion much more easily than ordinary hydrogen atoms. Also, the deuterated fluid can withstand significant tension (“stretching”) without forming unwanted bubbles. The substance is also relatively cheap, easy to work with, and not particularly hazardous.
To initiate the sonofusion process, we apply an oscillating voltage with a frequency of about 20 000 hertz to the piezoelectric ring. The alternating contractions and expansions of the ring—and thereby of the flask—send concentric pressure waves through the liquid. The waves interact, and after a while they set up an acoustic standing wave that resonates and concentrates a huge amount of sound energy. This wave causes the region at the flask’s center to oscillate between a maximum (1500 kPa) and a minimum (-1500 kPa) pressure. During the positive pressure cycle, the liquid is being compressed, and during the negative pressure cycle, it is being stretched.
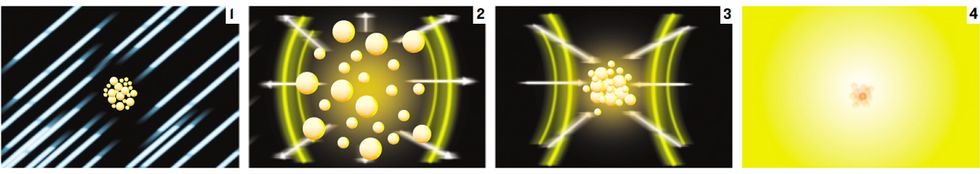
Precisely when the pressure reaches its lowest point, we fire a pulsed neutron generator, a commercially available, baseball bat-size device that sits next to the flask. The generator emits high-energy neutrons at 14.1 mega-electronvolts in a burst that lasts about 6 microseconds and that goes in all directions. Some neutrons go through the liquid, and some collide head-on with the carbon, oxygen, and deuterium atoms of the deuterated acetone molecules. In these collisions, the fast-moving neutrons may knock the atom’s nuclei out of their molecules. As these nuclei recoil, they give up their kinetic energy to the liquid molecules. This interaction between the nuclei and the molecules creates heat in regions a few nanometers in size that results in tiny bubbles of deuterated acetone vapor. Our experiments, along with computer simulations, suggest that this process generates clusters of about 1000 bubbles, each with a radius of only tens of nanometers.
By firing the neutron generator during the liquid’s low-pressure phase, the bubbles instantly swell—a process known as cavitation. In this swelling phase, the bubbles balloon out 100 000 times from their nanometer dimensions to about 1 mm in size. To grasp the magnitude of this growth, imagine that the initial bubbles are the size of peas. After growing by a factor of 100 000, each bubble would be big enough to contain the Empire State Building. Then, as the pressure cycle rapidly reverses, the liquid pushes the bubbles’ walls inward with tremendous force, and they implode with great violence.
The implosion creates spherical shock waves within the bubbles that travel inward at high speeds and significantly strengthen as they converge to their centers. The result, in terms of energy, is extraordinary: our hydrodynamic shock-wave computer simulations show that the shock waves create, in a small region at the center of the collapsing bubble, a peak pressure greater than 10 trillion kPa. For comparison, the atmospheric pressure at sea level is 101.3 kPa. The peak temperature in this tiny region soars above 100 million degrees centigrade, about 20 000 times that of the sun’s surface.
These extreme conditions within the bubbles—especially the bubbles at the center of the cluster, where the shock waves are more intense because of the surrounding implosions—cause the deuterium nuclei to collide at high speed. These collisions are so violent that the positively charged nuclei overcome their natural electrostatic repulsion and fuse. The fusion process creates neutrons, which we detected using a scintillator, a device in which radiation interacts with a liquid that gives off light pulses that can be measured. The process is also accompanied by bursts of photons, which we detected with a photomultiplier. And subsequently, after about 20 microseconds, a shock wave in the liquid reaches the flask’s inner wall, resulting in an audible “pop,” which can be picked up and amplified by a microphone and a speaker.
Increasing the pressure by an order of magnitude, firing neutrons at the flask to seed the bubbles on demand, and choosing a liquid rich in deuterium are the three key differences between single-bubble sonoluminescence and our sonofusion method.
We gather two kinds of evidence that the deuterium is fusing. The first is neutron emissions detected by the neutron scintillator. The device registers two clearly distinct bursts of neutrons that are about 30 microseconds apart. The first is at 14.1 MeV, from the pulsed neutron generator; the second, however, is at 2.45 MeV. This is the exact energy level a neutron produced in a deuterium-deuterium fusion reaction is expected to have. What’s more, these 2.45-MeV neutrons are detected at about the same time that the photomultiplier detects a burst of light, indicating that both events take place during the implosion of the bubbles.
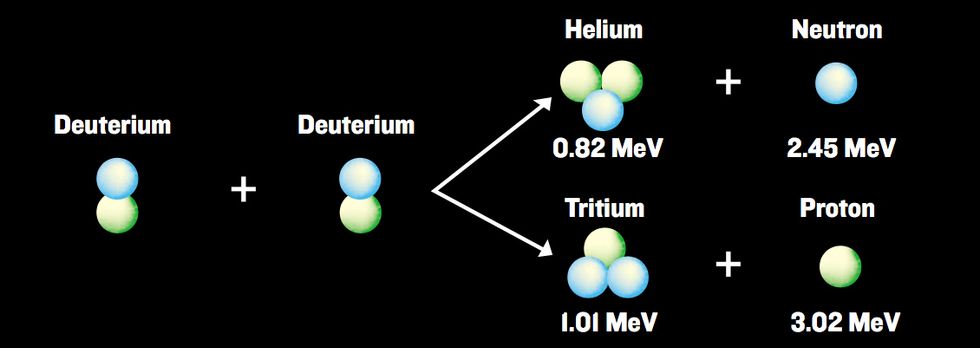
We found a second fusion “fingerprint” by measuring levels of another hydrogen isotope, tritium, in the deuterated acetone. The reason is that deuterium-deuterium fusion is a reaction with two possible outputs at almost equal probability [see illustration, "The Action in the Flask"]. One possibility gives you 2.45-MeV neutron plus helium, and the other gives you tritium plus a proton. Thus, the build-up of tritium above the measured initial levels is an independent, and strong, indication that fusion has taken place, since tritium cannot be produced without a nuclear reaction.
The Action in the Flask
When we first published our results, in the 8 March 2002 issue of Science, many scientists were skeptical. Critics argued that the neutron detection system wasn’t accurate enough to distinguish between the 14.1-MeV and the 2.45-MeV neutrons; they argued, in fact, that the neutrons we thought were coming from fusion might actually be coming from our own neutron generator. Confusion grew to the point that some even said sonofusion could be a repeat of the cold fusion fiasco of the late 1980s. Our experiments, however, are based on thermonuclear fusion, which involve extremely high temperatures and pressures. In contrast, cold fusion invoked some new physical processes to explain fusion at room temperature.
We addressed those criticisms when we repeated the experiment last year, with results published in the journal Physical Review E in March 2004. This time we used a detector that registered neutrons and also gamma rays. It ran over the entire duration of the procedure, while the detector used before made measurements only in a few shorter intervals. This time it became even clearer that the detector was registering two separate bursts of neutrons. The first one corresponds to the 14.1-MeV neutrons of the generator, and it lasts less than 10 microseconds, after which it quickly decays. The second one corresponds to the 2.45 MeV of deuterium-deuterium fusion, and it appears 30 microseconds after the first burst, accompanied by characteristic gamma rays. We also ran several control experiments, such as using regular acetone instead of the deuterated version or firing the neutron generator during the positive pressure cycle rather than during the negative. In such cases we observed no signs of fusion at all.
Now at least five groups—three in the United States and two in Europe—are working on reproducing our sonofusion results. Some have apparently already succeeded and are now preparing to publish their findings. As these groups have learned, it is not an easy experiment to run, despite its apparent simplicity. Inside that glass flask, there are many kinds of processes going on—the dynamics of the fluid, shock wave propagation, plasma formation, chemical reactions, nuclear processes—and you need to understand and treat them carefully. It took us two and a half years of painstaking experimentation—and dozens of broken flasks—to observe fusion. It is encouraging that other groups may soon present their results and help advance the field.
Now that fusion reactions are taking place, can we somehow harness the energy they are releasing? Well, not so fast. Each individual fusion reaction is very brief—it lasts only about a picosecond—and it is confined to a very small region. (Think of it as “fusion sparks” rather than a steady fusion burn.) As a result, the energy output is relatively small, and that’s why, despite the miniature “stars” within the bubbles, the fusion reactions don’t melt down the whole apparatus. To obtain something interesting in terms of energy, the next step is to scale up the apparatus and make the fusion reactions self-sustaining. This is the greatest challenge not only for sonofusion but also for all other fusion methods.
Research groups throughout the world have concentrated on two approaches. In one, extremely energetic laser beams converge on a tiny solid pellet of deuterium-tritium fuel. The result is a shock wave that propagates toward the center of the pellet and creates an enormous increase in temperature and density. The world’s largest experiment using this method, called inertial confinement fusion, is at the Lawrence Livermore National Laboratory’s National Ignition Facility, in California. One of the drawbacks of this approach is the amount of power the lasers require. At the National Ignition Facility, for example, the capacitor banks that store the energy for the 200 lasers used require an area equivalent to that of a few U.S. football fields. Also, the facility’s main goal is not producing energy but rather conducting thermonuclear weapons studies.
The second approach, called magnetic confinement fusion, has been under investigation since the 1950s. It uses powerful magnetic fields to create immense heat and pressure in a hydrogen plasma contained in a large, toroidal device known as a tokamak. The fusion produces high-energy neutrons that escape the plasma and hit a liquid-filled blanket surrounding it. The idea is to use the heat produced in the blanket to generate vapor to drive a turbine and thus generate electricity. Magnetic confinement has been demonstrated experimentally on tokamaks at the Joint European Torus, in Abingdon, England, and at the Princeton Plasma Physics Laboratory, in New Jersey. Yet tremendous challenges remain, such as holding the plasma in place while increasing temperature and pressure. It’s a very unstable process that has proved difficult to control. Imagine trying to squash Jell-O with your hands without letting it escape between your fingers.
Recently, a consortium of institutions from China, Japan, South Korea, the European Union, Russia, and the United States said it was ready to start building the ITER—International Thermonuclear Experimental Reactor—a US $5 billion, 500-megawatt reactor based on magnetic confinement. The consortium is now deciding between Cadarache, France, and Rokkasho, Japan, as a home for the reactor. ITER is not expected to begin operating until 2015, and a commercially viable version will be even further away—some say 2050, give or take a few decades.
For sonofusion ever to compete with those other approaches, it will have to overcome a number of challenges. The first is making the fusion reaction self-sustaining—in other words, arranging the setup so it produces a continuous neutron output without requiring the external neutron generator. A possible way of doing that would be simply to put two complete apparatuses side by side so that they would exchange neutrons and drive each other’s fusion reactions. Imagine two adjacent sonofusion setups with just one difference: when the liquid pressure is low in one, it is high in the other. That is, their pressure oscillations are 180 degrees out of phase. Suppose you hit the first apparatus with neutrons from the external neutron generator, causing the bubble cluster to form inside the first flask. Then you turn off the neutron generator permanently. As the bubble cluster grows and then implodes, it will give off neutrons, some of which will hit the neighboring flask. If all is right, the neutrons will hit the second flask at the exact moment when it is at the lowest pressure, so that it creates a bubble cluster there. If the process repeats, you get a self-sustaining chain reaction.
Next, it would be necessary to scale up the apparatus so it could produce more energy than it consumes. So far, a single apparatus yields about 400 000 neutrons per second. The neutrons are an important measure of the output of the process, because they carry most of the energy released in the fusion reaction. Yet that yield corresponds to a negligible fraction of a watt of power. How much would we have to scale things up to create, say, a full-size electricity-producing nuclear reactor?
Such reactors—a billion times as large in volume as our current apparatus—normally operate at a few thousand megawatts of thermal power. In terms of neutron-per-second output, that means 1022 neutrons per second. It is a long way to go, but our experiments to date have shown that with many relatively small steps we could eventually get there. For example, in our experiments, as we increased the pressure by about a factor of two, from 700 kPa to 1500 kPa, we detected an increase in the neutron output by a factor of about 100 000. With improvements to several parameters of the sonofusion process, such as the size of the liquid flask, the size of the bubbles before implosion, and the pressure compressing the bubbles, we should be able to reach a neutron output sufficient for net power production.
The total neutron output would include not only the neutrons from deuterium-deuterium fusion, but also neutrons from deuterium-tritium fusion, since the tritium produced in sonofusion remains within the liquid and can fuse with deuterium atoms. Compared with deuterium-deuterium fusion, deuterium-tritium fusion occurs 1000 times more easily, produces more energetic neutrons, and could increase the neutron yield by about three orders of magnitude. Then, if we installed a blanket system around the reactor like the one in a tokamak, all those high-energy neutrons would collide with it, raising its temperature so that its heat could then be used to boil a fluid to drive a turbine.
As an alternative, we are also exploring the possibility of using different liquids in the reaction flask. If the energy from the fusion products could be retained by the liquid, then the liquid itself could be used to transfer heat to another fluid, such as water vapor, which would then drive a turbine. To do that, however, you would need a fluid that works at much higher temperatures than our chilled acetone (our current experiments are running at 0 degrees centigrade). However, there’s no need to use a hydrocarbon such as acetone to produce sonofusion. We’re currently looking at many other fluids, including some exotic silicon- and carbon-based liquids that can work at much higher temperatures. Ideally, the fluid would run at the temperatures of the pressurized water used in fission nuclear reactors—about 340 degrees centigrade. That way the same heat-exchange technology found in those reactors could be used with little modification. In this case, you would only have to replace a standard fission reactor core with a sonofusion-based core, which would then generate the steam to spin a turbine generator.
Even though this and other sonofusion applications may be years or decades away, at least two companies are betting they are not only feasible but commercially viable. General Fusion Inc., in Vancouver, B.C., Canada, has come up with an approach that combines sonofusion and laser inertial confinement. The plan is to achieve high pressures inside a vessel 1 meter in diameter filled with liquid lithium, within which a small hydrogen-based shell is positioned. A pressure wave of 1 million kPa is launched all around the shell. As this spherical pressure wave converges toward the center, according to the company, its peak intensity grows to 10 billion kPa as it reaches the shell. The subsequent collapse, much like a laser-induced collapse, will generate fusion energy in excess of the energy invested—or so the designers hope.
Meanwhile, Impulse Devices, in California, has chosen an approach similar to our sonofusion method. The main difference is that, instead of a glass flask, the company is using a stainless steel spherical chamber, about 24 centimeters in diameter, that can resist extremely high pressures [see photo, "Sonocapsule”]. Piezoelectric crystals are mounted on cylindrical attachments distributed on the chamber’s surface. Using various test liquids and different methods to seed an initial cluster of bubbles, Impulse Devices has been successful in consistently imploding the bubbles and in observing the resulting light emissions. The company hasn’t detected any signs of fusion to date, but it is repeating the experiment with different adjustments.
The Sonofusion Apparatus we developed, as is, might be useful for various applications. One is the study of thermonuclear fusion itself; the method is considerably simpler and cheaper than other means of studying fusion in the laboratory. Another possible application is as a low-cost pulsed neutron generator. While a commercial device can cost up to $250 000, our setup could be produced for about $1000 (instead of an external neutron generator, small quantities of thorium or natural uranium salts, both of which emit alpha particles, would seed the bubbles). As a neutron-emitting device, the apparatus may be useful, for example, to probe the molecular structure of materials or to activate certain anticancer drugs, as in an experimental therapy called boron neutron capture.
Nevertheless, the Holy Grail of all fusion research is the development of a new, safe, environmentally friendly way to produce electrical energy. Fusion produces no greenhouse gases and, unlike conventional nuclear fission reactors, it produces no noxious radioactive wastes that last for thousands of years. With the steady growth of world population and with economic progress in developing countries, average electricity consumption per person will increase significantly. Therefore, seeking new sources of energy isn’t just important, it is necessary. Much more research is required before it is clear whether sonofusion can become a new energy source. But then there is just one way we can find out—we will continue making bubbles.
About the Authors
Richard T. Lahey Jr. is a professor of engineering and physics at Rensselaer Polytechnic Institute in Troy, N.Y.; Rusi P. Taleyarkhan is a professor of nuclear engineering at Purdue University in West Lafayette, Ind.; and Robert I. Nigmatulin is the president of the Russian Academy of Sciences branch in Ufa.
To Probe Further
The computer simulations used to study sonofusion are discussed at https://www.rpi.edu/~laheyr/DARPA_Slides_Nigmatulin-Lahey.ppt (5.3 mb download).
Details on laser- and magnetic-induced fusion are available at https://www.llnl.gov/nifand at https://www.iter.org.
An interesting summary of sonofusion’s brief history can be found at https://www.washington.edu/research/scienceforum/pdfs/Crum.pdf (1 mb download).