There are many ways to convert the movement of liquids into electricity, such as, in the form of hydropower or tidal power. But there’s another kind of movement available in liquids that has rarely been considered for this purpose—molecular thermal motion or Brownian motion. (Albert Einstein famously developed the foundational theory of Brownian motion, the basis of his Ph.D. thesis, at the University of Zurich in 1905.)
Molecular motion has traditionally been an impediment in engineering on a nanoscale, but recent developments in nanotechnology have also allowed scientists to use this phenomenon to their advantage. In a recent study from China, scientists posit that the internal, molecular motion of liquids could be a source of energy if only there was a way to harvest it. To demonstrate, they built a prototype of a molecular thermal motion harvester (MTMH).
The research reveals the potential of producing small currents from molecular motion.
Their paper, which was published in the journal APL Materials earlier this month, reveals the potential of producing small currents from molecular motion.
Atoms and molecules in a gas or liquid are in continuous and random motion at temperatures above absolute zero (0 kelvin, which is -273 °C or -459 °F). The resulting pushing and shoving keeps this internal movement going, resulting in untapped kinetic energy.
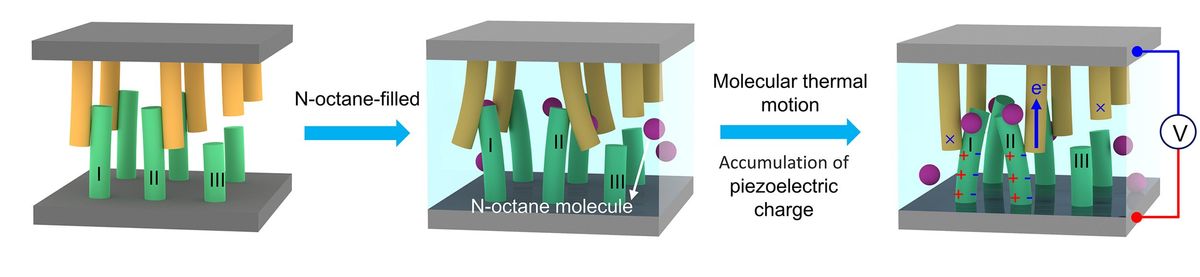
To use Brownian movement as a source of energy, the researchers had to design a device that could capture and convert the motion into electricity. To make their harvester, they used nanowire arrays made of zinc oxide (ZnO) and gold-coated ZnO as electrodes. ZnO is a piezoelectric material, which means it produces an electric charge in response to mechanical stress, such as bending or flexing, from the Brownian movement of the liquid medium, which in this case was n-octane, with a purity of greater than 99.99 percent. The gold-coated ZnO nanoarray was used as a negative electrode, the plain ZnO one as positive, and the entire device—2 by 2 centimeters in size—was sealed with epoxy.
The prototype yielded a stable output voltage of 2.28 millivolts and current of 2.47 nanoamperes at room temperature, though the latter number goes up as temperature rises. Yucheng Luan, founder of the Shanghai-based East Eight Energy, the lead investigator of the present study, says that though the energy output was very low, this experiment was a demonstration that it is possible.
The researchers chose their materials for the demo based on the fact that zinc oxide nano-arrays can be synthesized relatively easily and their morphology controlled, he says. The use of pure, electronic-grade octane was dictated by the necessity of not having free-moving ions that could leak electrons from the nano-arrays. But there are a number of ways to increase the power density.
“First of all, we can use different materials,” says coauthor Wei Li, professor at Nankai University, in China. “Also, we are going to use some different [designs], such as, a tandem structure, different fabrication methods, and we can also alter the liquids we use.” In fact, since the paper was published, he adds that the researchers have already had better outputs with other materials.
With the abundance of liquids and gases available, Luan sees untapped potential for their MTMH device. Without leaning on any notions of perpetual motion, small fluctuations in ambient thermal energy might be tapped, the researchers suggest, via a material’s Brownian motion—which is present in a liquid that is perfectly still, as long as the temperature is above absolute zero.
Luan says he’s optimistic that in the near future, the MTMH might see applications in different kinds of microwatt and milliwatt devices, either independently or with a capacitor. “We [also] believe the harvester is capable of driving devices in the watt or kilowatt range,” he says.
Payal Dhar (she/they) is a freelance journalist on science, technology, and society. They write about AI, cybersecurity, surveillance, space, online communities, games, and any shiny new technology that catches their eye. You can find and DM Payal on Twitter (@payaldhar).