In the expanding universe of two-dimensional materials, perhaps none have been more tempting than silicon and its 2D version, known as silicene. The attraction is obvious: Silicon is the boat on which the computer age has floated for more than 50 years. And its proximity on the periodic table to carbon, whose 2D version is the wonder material graphene, has led researchers to investigate whether it might have the same attractive properties.
If researchers can overcome more of the obstacles standing in the way of silicene production, it has some pretty attractive qualities. First, unlike graphene, it has an intrinsic bandgap, which makes it attractive for digital electronics to stop and start the flow of electrons. Its semiconductor nature also comes with many of the same properties that make graphene so attractive—including, potentially, superconductivity.
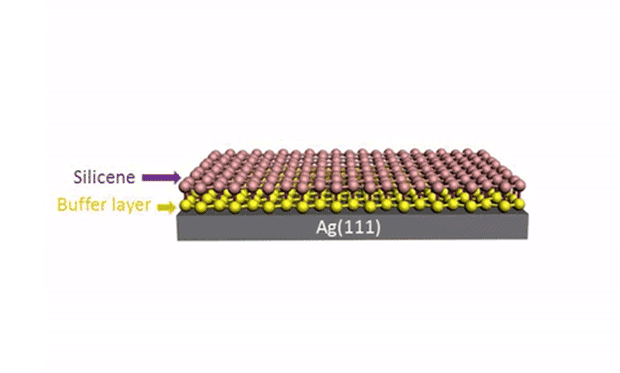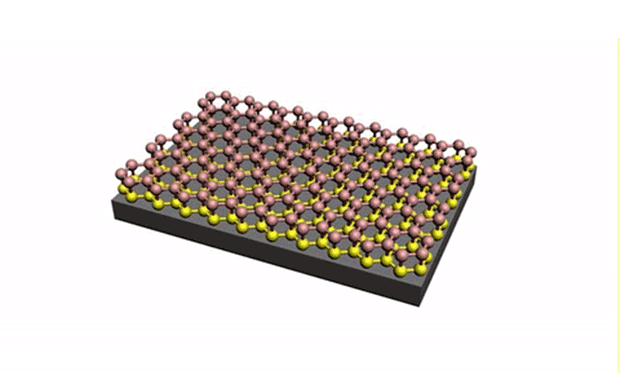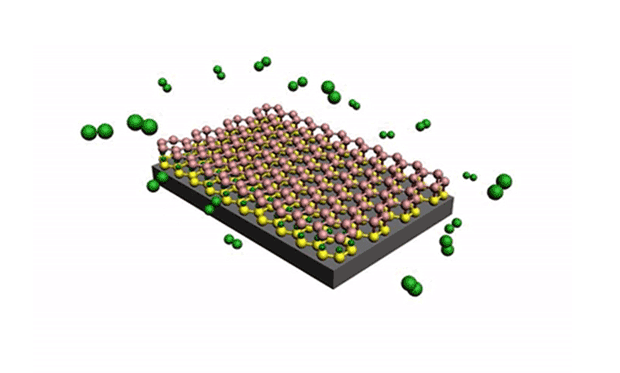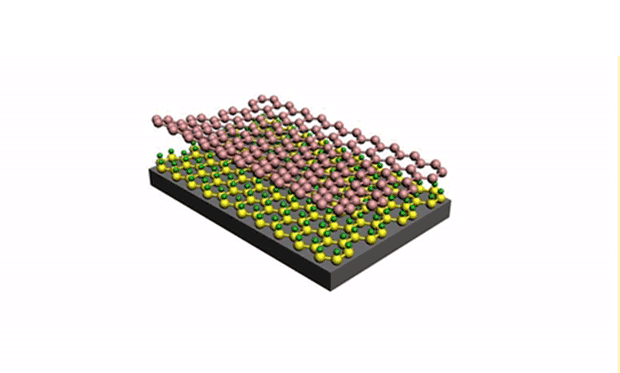
While silicene is indeed tempting, it has proven frustratingly difficult to produce. Now researchers at the University of Wollongong, in Australia, have overcome one of the main obstacles: separating it from its substrate.
In research described in the journal Science Advances, they developed a technique for reliably separating the silicene from the metal surface it is grown on. The trick is to get oxygen molecules to intercalate between the underlayer of silicene, effectively isolating the upper layer of silicene from the metal substrate. (Intercalation is the reversible inclusion of a molecule or ions between two other molecules in multilayered structures or compounds.)
“We know silicene crystals prefer to firmly attach on the metallic substrate, and because they are too thin to be peeled off by any mechanical tools, it’s impossible to remove them from the substrate,” said Yi Du, who led the research, in a press release.
In the experiment, the silicene layers were produced by the deposition of silicon atoms from a silicon wafer onto a gold substrate. A scanning tunneling microscope (STM) was used to introduce the oxygen molecules because of the ultrahigh vacuum used by the STM. While the silicene was still in the deposition chamber, the oxygen molecules were introduced at 200 ºC.
“Because the vacuum levels are so high, we can inject the oxygen molecules into the chamber, and they become a ‘molecular flux’ that follows a straight pathway,” said Du. “This allows us to direct these molecules precisely into the silicene layers, acting like scissors to separate the silicene.”
Researchers have made some pretty significant progress with silicene over the past four years, not the least of which has been overcoming its tendency to self-destruct and turn back into silicon.
The Wollongong researchers feel pretty confident that this work could be a significant breakthrough in silicene reaching its potential.
Du added: “This work solves the long-lasting problem of isolating this super material for further device development. It challenges the entire scientific literature on silicene since its discovery. These findings are relevant for the future design and application of silicene-based nano-electronic and spintronic devices.”
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.