Ever since the end of 2013, when James Tour and his colleagues at Rice University created graphene quantum dots (GQDs) from coal, they have been busily looking for applications for the material.
Whenever a nanomaterial needs an application, offering it up as a replacement for platinum catalysts in fuel cells is a sure fire way to generate some attention. Late last year, Tour’s group proved to be no exception to this rule when they did exactly that.
Not to say that finding a cheaper alternative to platinum in fuel cells isn’t necessary, but isolating hydrogen and then creating a distribution infrastructure for it is a far more critical factor in realizing the so-called “hydrogen economy.”
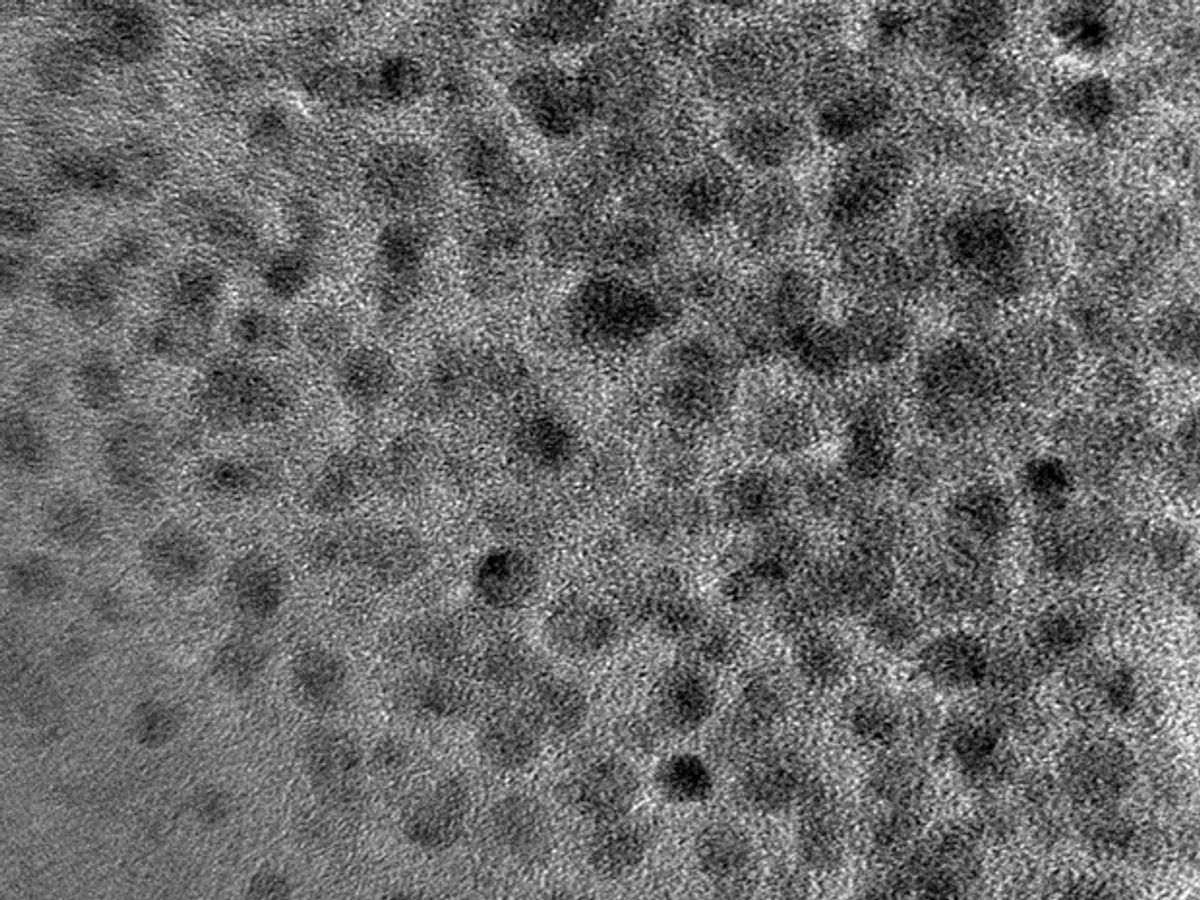
So, Tour and his team have taken a new tack with their GQDs and developed a simple manufacturing technique that can sort out the GQDs according to their size—and therefore, their semiconducting properties.
In research published in the journal Applied Materials & Interfaces, the Rice team focused renewed attention on their method for producing the GQDs from coal. They discovered that if they carefully controlled the reaction temperature in the oxidation process that turned the coal into quantum dots—the nanoscale semiconducting crystals with properties that make them attractive for several optoelectronic applications—they could control the size of the dots they produced. Hotter temperatures produced smaller dots; different size dots have different semiconducting properties. The researchers then used an ultrafiltration system, which is often used in industrial water filtration systems, to start the sorting of the quantum dots according to their size.
Quantum dots can absorb and emit photons at specific wavelengths, from visible colors into infrared. The size of the dot can determine which wavelength, or color, is absorbed. Smaller dots emit green light, while the larger dots emit light in the orange to red range. Tour and his team determined that the tiniest quantum dots, which emit blue light, were the easiest to produce from coal.
The ability to do this sorting is critical for the production of optoelectronic devices based around their fluorescence. It is for this reason that the Rice researchers are now discussing metal or chemical detectors that let engineers tune the fluorescence of the quantum dots so the devices can avoid interference with the target materials’ emissions. Of course, the Rice team still mentions catalytic reactions, but this latest production development seems to place the graphene quantum dots on a broader, if not more adoptable, application path.
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.