The Cancer Surgeon’s Latest Tool: Quantum Dots
Now used to brighten displays, quantum dots could one day guide a surgeon’s hand


The bits of semiconducting material that are lately brightening the colors on television screens hold a much greater promise—that they will extend lives. These tiny crystals are far too small to be seen with the naked eye, measuring just one ten-thousandth the width of a human hair, which is one reason they’re called quantum dots. When you shine a suitable light on such a dot, it becomes luminous, emitting a very pure color that is determined by its size.
My colleagues and I at Nanoco Technologies, in Manchester, England, have been working with a team of researchers led by Sandy MacRobert at University College London to explore quantum-dot technology for various medical treatments. While there are many exciting possibilities to be explored, the most important one we’re pursuing aims to help delineate malignant tumors and the pathways along which they spread, which could improve the safety and effectiveness of cancer surgery.
The quantum dots we’re discussing are engineered from a blend of elements drawn from groups III and V, or II and VI, of the periodic table—elements like cadmium, lead, zinc, arsenic, indium, tellurium, selenium, sulfur, and phosphorus, which are used in pairs or as alloys. In that way quantum dots are similar to various optoelectronic devices. When exposed to a relatively shortwave light source, quantum dots absorb some of its energy and reemit light of a longer wavelength (which is to say, a different color), a process known as fluorescence. The size of the dots can be finely tuned so that they will give off any color of the spectrum you might want: The bigger the dot, the longer the wavelength it emits.

The most common commercial use of quantum dots by far has been in the backlights for LCD television screens, where they help produce two of the three colors needed. Blue comes directly from an LED, which illuminates quantum dots that in turn produce red and green light. Because quantum dots emit very pure colors, the television pictures they create are particularly vivid, showing upwards of 50 percent more color range than the average display. Their use in TVs also lowers power consumption, because less of the backlight’s energy is absorbed by the color filters in the LCD screen.
One hurdle for this technology has been that quantum dots typically contain cadmium, a heavy metal that’s extremely toxic. Cadmium plays the role of a group II element here, often being combined with the group VI element selenium. Because of environmental concerns, more and more countries are outlawing the use of cadmium in electronic products, which spurred our company to devise quantum dots that are free of this element as well as other toxic heavy metals. Along the way, we realized that such cadmium-free quantum dots might also have important uses in medicine.
Most exciting in this regard is that quantum dots have several advantages over the organic dyes now employed during certain cancer surgeries. Of course, quantum dots also present some special challenges, which we’ve been working hard to address.
To understand how quantum dots might help improve outcomes for some cancer patients, you need to know a little about how surgeons treat this disease. They typically remove both the malignant tumor and the surrounding lymphatic vessels and nodes, thus preventing them from carrying the cancer to distant sites in the body. The more lymph nodes removed in surgery, the lower the risk that the cancer will reach other organs. But the lymphatic vessels and nodes near the tumor may be healthy, in which case their removal is certainly unnecessary and possibly harmful. So one of the challenges in cancer surgery is to figure out exactly how much lymphatic tissue to cut out and how much to leave intact.
One strategy makes use of the fact that the membranes that surround cancerous cells are more penetrable and reactive than healthy tissue. So surgeons inject the area of the primary tumor with dyes or radioisotopes. After a few minutes the surgeon can then determine which of the surrounding lymphatic tissues are likely to contain cancer simply by noting the presence of the colored dye or identifying them with a radiation detector.
The main problem with this approach is that the dyes that are normally used for this purpose (isosulfan blue or methylene blue) migrate quickly from the site of injection through the cancer-containing lymph nodes and on to the entire lymphatic system. So surgeons typically take a picture of the tumor and surrounding areas soon after injection of the dye, before it has migrated very far, and then operate based on what that picture shows. As you can imagine, it’s hard to be accurate when you have to refer to memory or a picture to guide your actions.
Another issue doctors can run into when using organic dyes is premature fading: The dyes bleach quickly under the strong lights used to illuminate them. Still another problem is that, even before any fading occurs, those blue organic dyes often cannot be seen through the skin or fatty tissue. There is a different organic dye—something called indocyanine green—that does show through skin and fat because it fluoresces in the near infrared, which penetrates better through living tissue. Because the dye’s emissions are outside the visible range, though, the surgeon must use special imaging equipment to see where the tracer has gone.
When a radioactive tracer is employed for such surgery, it’s typically a metastable form of technetium that emits gamma rays when it decays and has a half-life of about 6 hours. So time is not such a factor. But like dyes, it may migrate too quickly. In any event, pinpointing where the technetium has moved is hard to do with a handheld gamma probe. As a result, radioactive tracers give a very coarse picture of how cells shed from a tumor might travel through the patient’s lymphatic system.
It’s evident that current methods need improvement, as shown by a 2013 clinical trial intended to determine the effectiveness of a technique called sentinel-lymph-node surgery for women with a particular kind of breast cancer. Normally, 10 or more lymph nodes in the woman’s armpit would be removed and analyzed for cancer. With sentinel-lymph-node surgery, however, a dye or radioactive tracer (or both) is injected at the site of the tumor, which can lead the surgeon to the first few armpit nodes along the line of lymphatic drainage. If those “sentinel” nodes do not contain cancer, there is a strong likelihood that the cancer has not spread from the tumor site. This approach avoids having to remove a large number of lymph nodes in the armpit, which often causes lymphedema (arm swelling), numbness, and restricted arm movement.
So this seems like a smart strategy for treating breast cancer, right? The question is how well it really works. The 2013 study found that 12.6 percent of the women got false-negative results. That is, their sentinel nodes, as identified using both a blue dye and a radioactive tracer, showed no cancer, but cancer was, in fact, present in these women’s other armpit nodes. The study authors considered a false-negative rate above 10 percent to be problematic. That doesn’t mean that the medical community should abandon sentinel-lymph-node surgery—not at all. Physicians just need to figure out how to do it better.
Here’s where quantum dots could really...well, shine. Unlike organic dyes, quantum dots migrate relatively slowly and can maintain their ability to fluoresce for many hours. So the surgeon could perform the procedure based on what the dots reveal in real time. The surgical team would simply inject the quantum dots and then illuminate the area with either ultraviolet or blue light. The quantum dots would fluoresce brightly and could do so for the duration of the operation. They could be engineered to emit light at whatever wavelength the surgeon desired, be it one that can be seen directly or one that emits in the infrared, allowing its light to pass through skin and fatty tissue. The surgeon could take as much time as needed to determine the locations of sentinel nodes, which would presumably make such procedures more accurate.
What exactly arethese magical quantum dots? Quantum dots are made up of tiny chunks of semiconducting material, which typically measure between 2 and 10 nanometers across. Because they are so small, their electronic and optical properties are distinct from those normally associated with whatever material they are made of. They straddle an odd middle ground between molecules and bulk material, a fact that becomes most evident when you consider how they fluoresce, absorbing a broad range of wavelengths and then reemitting that energy as light of a very specific wavelength—an ultrapure color.
The beauty of quantum dots is that you can tailor their optical properties simply by changing their size: The larger the dot, the redder (longer wavelength) its optical emissions. For most semiconductors, you have to change their composition to change their optical properties. No wonder the engineers making displays are keen to take advantage of quantum dots.
Beginning in the mid-1990s, scientists began devising various ways to produce these tiny crystals in quantity. In 2001, Nanoco was spun off from Paul O’Brien’s research group in the School of Chemistry at the University of Manchester to commercialize a process for quantum-dot synthesis called molecular seeding, which allows very good consistency and reproducibility. The challenge for us then became making quantum dots that don’t cause any ill effects. You certainly wouldn’t want to use the typical kinds of quantum dots that have been created in the past: The cadmium they contain is a huge threat to human health. You wouldn’t want to allow that cadmium to be released into the environment, much less be injected into a patient.
Anticipating that this issue can be overcome, medical researchers around the globe have been investigating the possible uses of quantum dots in the body. Working with breast-cancer tumors in mice, for example, scientists have found that, unlike conventional dyes, quantum dots are less likely to migrate through the lymphatic system beyond the regional lymph nodes around the tumor.
Soon after Nanoco was established, it began engineering a new type of quantum dot that is free of cadmium and other toxic heavy metals. Other companies quickly followed suit. These cadmium-free quantum dots are attractive for making displays, because they help them conform to various environmental regulations. The first TV sets to use cadmium-free quantum dots, such as Samsung’s SUHD TV, have just recently appeared on store shelves. But there’s an even greater payoff coming: While definitive tests have yet to be done, my colleagues and I believe that modified versions of these cadmium-free quantum dots are likely to be safe for clinical use inside the human body.
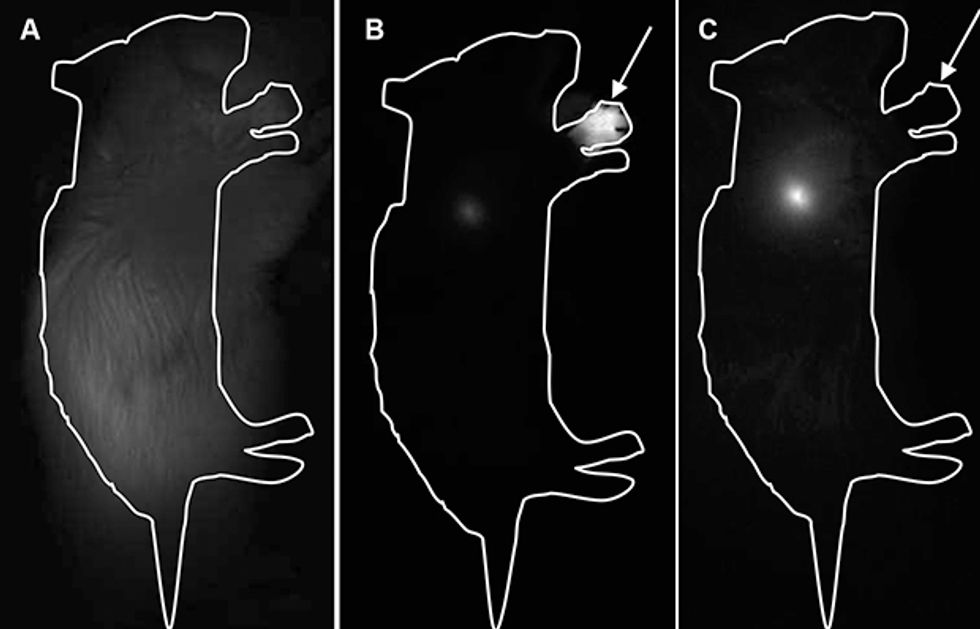
In addition to eliminating the heavy metals, researchers at our company have developed a special polymer coating to render the dots biocompatible, meaning that they are not likely to engender inflammation or any other sort of immunological response. Nanoco’s research partners at University College London have used these dots successfully in surgery to remove tumors and surrounding tissue from mice, with no detectable toxic side effects in the animals.
Of course, even if these quantum dots prove perfectly safe, it will take a long time to determine just how much of an advance they represent over conventional dyes and radioactive tracers. It seems reasonable to suppose that they will improve outcomes in cancer surgery, but clinical trials will have to be carried out to determine whether surgeons actually remove more cancerous tissue with quantum dots guiding their decisions, whether that lowers the risk that the cancer will spread, and whether there will be less need for repeat surgeries.
In addition to delineating tumors, the quantum dots may have other important uses in cancer treatment. In particular, physicians may someday be able to use quantum dots to destroy cancerous tumors in place. That’s because tumors typically absorb and accumulate nanoparticles faster than do normal healthy tissues, a phenomenon called enhanced permeability and retention. So quantum dots could, in principle, also be used as an integral part of chemotherapeutic drugs that are preferentially absorbed by a tumor.
Another exciting possibility involves the application of quantum dots to something called photodynamic therapy, which can be used instead of surgery to treat cancer by killing malignant cells through exposure to light. For this, physicians first inject special sensitizing drugs, which are preferentially taken up by cancerous tissues. When light is shone on the tumor, the sensitizing molecules absorb energy from the light and cause a toxic form of oxygen to be produced, which then kills the surrounding cancerous cells.
Special quantum dots could be created to help with such procedures. The dots would serve to transfer energy from the light to attached sensitizing molecules, thus enhancing the ability of traditional photodynamic-therapy drugs to kill malignant cells.
Cancer treatment is just one of many promising applications for quantum dots in medicine, but the question that is holding back all this research is whether it’s safe to use them inside a human being. Injecting substances into the body is never without risk, and quantum dots have long been a worry in that regard. But the elimination of toxic heavy metals and the use of biocompatible coatings should go a long way in alleviating concerns.
The most likely problem posed by the quantum dots my colleagues and I have been investigating is allergic reaction, an all-too-common phenomenon in medicine. Indeed, an organic dye commonly used now for cancer surgery elicits dangerous allergic responses in 1 to 3 percent of patients.
We won’t know how allergenic our quantum dots are until we conduct human trials, which are probably two to three years away yet. But if all goes as we hope, the new dots won’t prove any worse in this regard than today’s organic dyes. If so, nontoxic quantum dots could begin helping to extend the lives of cancer patients perhaps just a few short years after that. It will be a triumph well worth remembering while enjoying the especially vivid picture on the television you have at home, where the same fundamental technology will by that time probably be at work.
This article appears in the October 2016 print issue as “Fighting Cancer With Quantum Dots.”
About the Author
Imad Naasani is chief technology officer, life sciences for Nanoco Technologies, in Manchester, England, a maker of quantum dots.
