In January, regulators in Japan and the United States grounded the worldwide fleet of Boeing 787 Dreamliners after lithium-ion batteries caught fire in two of them—one in the air over Japan and the other on the ground in Boston. AsIEEE Spectrum went to press, authorities were tracing the proximate cause of the Boston fire to a short-circuit in one of the batteries. The ultimate cause remained unclear.
What is clear, though, is that lithium-ion batteries in commercial airliners are probably here to stay. Airliners are going to keep electrifying previously mechanical systems like those used for braking and de-icing, because doing so makes the aircraft much lighter and more efficient, says Cosmin Laslau, an electrochemistry expert and technology analyst for Lux Research.
A heavy reliance on electrical systems, together with extensive use of ultralight carbon composites, is what cuts the 787’s fuel consumption by 20 percent versus the competition’s—its main selling point. (The batteries are mere backup to the six—count ’em, six—generators that supply the 787 with a whopping 1.5 megawatts, about five times as much as in any comparable airliner.) And lithium-ion batteries are likely to remain the technology of choice for new airliner designs, says Laslau. They’re valuable not just for the great amount of energy they can store in comparatively little space and at a lighter weight but also for the big spike of current they can kick out. What’s more, lithium-ion technology is getting better and safer by the year, while older battery technologies, like nickel metal hydride, are essentially standing still.
That said, figuring out what went wrong and how to fix it will be crucial to the future of the technology—and of Boeing. Damage to the batteries in Japan and Boston was extensive, suggesting that there were mishaps in the first two layers of the redundant safety system. The first layer contains two independent electronic control circuits, which manage the batteries’ operation, keeping voltage within bounds and preventing overcharging. This control system is also supposed to prevent full discharging, which would keep a lithium-ion battery from ever being recharged again.

The second layer of protection involves the thermal sensors inside the battery itself, which are supposed to detect overheating and halt it by disconnecting the batteries. It was the third and final layer—shielding meant to contain a fire—that saved the day. Even so, some flammable electrolyte leaked from the batteries.
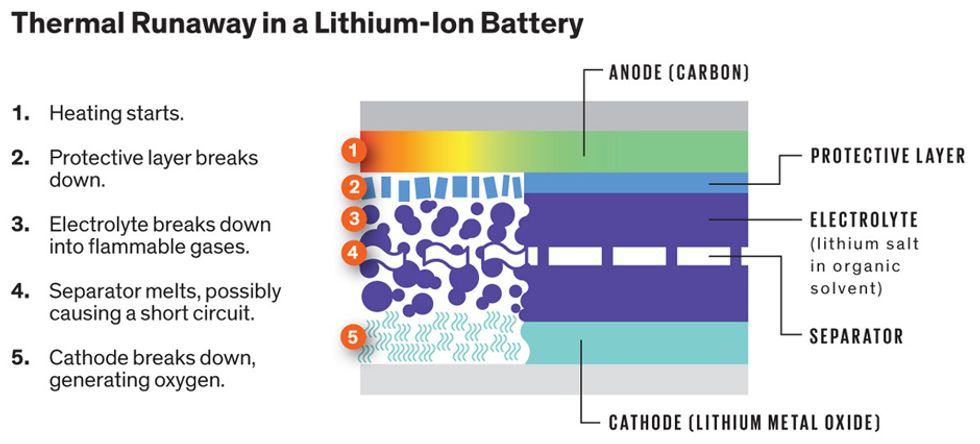
When lithium-ion batteries were introduced in mobile electronics, they gained infamy for incineration. The issue stems from a self-accelerating process called thermal runaway.
A lithium-ion battery typically has a carbon anode, a metal oxide cathode, and, sandwiched between them, an electrolyte consisting of a lithium salt dissolved in an organic liquid. When current flows into the battery, lithium ions move from the cathode through the electrolyte to the anode; when current flows out, they return to the cathode.
Too much voltage, too high a charge, too high an ambient temperature, or a combination of these things can trigger thermal runaway. According to the U.S. National Transportation Safety Board, it was heat, caused by an unexplained short-circuit in one cell, that started thermal runaway, which then cascaded through neighboring cells.
In one scenario, Laslau says, “as the temperature goes up, a protective layer on the surface of the anode breaks down.” Next, heat makes its way across to the electrolyte, breaking it down and releasing flammable gases like ethene and methane. That can cause pressure to build up in the cell. Then the heat begins to melt the separator, a polymer membrane that normally keeps the cathode and anode apart. If that happens, the cell can short-circuit, releasing much more heat. Finally, the cathode breaks down, releasing oxygen. Because the fire creates its own oxygen, it can’t be put out easily and must therefore be contained.
The battery, made by Japan’s GS Yuasa, was Boeing’s choice for the 787’s design in 2005, which Laslau says is unfortunate because its cathode was based on cobalt oxide. “The cobalt oxide chemistry that Boeing chose has a fantastic energy density, but it’s not the safest. Lithium iron phosphate would be safer,” he says. But the iron phosphate alternative wasn’t well developed seven years ago.
Perhaps Boeing’s reliance on relatively old technology reflects the many hiccups that slowed the development of the airliner, which went into service in 2011, about two years later than planned. “You could call it teething pains, but there was an awful lot of it, the worst in many decades,” says Richard Aboulafia, an analyst with the Teal Group, an aerospace consultancy in Virginia.
Boeing has itself placed some of the blame for missed deadlines on its unprecedented decision to employ vendors around the world—not only to provide critical systems but in many cases to design them. “This arrangement is called working with ‘risk-taking suppliers,’ ” says physicist Hans Weber, who runs Tecop International, a San Diego aviation consultancy. “Detailed design is up to the suppliers in many cases, who use their own money and get paid only when [the system is] delivered.” But, he adds, Boeing underestimated how much management attention would have to be given just to keep suppliers up to speed.
As the project stretched out, the suppliers felt the economic pressure. Reuters reported in January that whistle-blowers working for some of those companies had gone to the National Transportation Safety Board with allegations of cutting corners, including one case involving the battery chargers.
Boeing bet big on a daring concept and, despite the 787’s production delays, won contract after contract from airlines, at least before the battery imbroglio. It could be that Boeing’s example will save its competitors from teething pains of their own. Its archrival, Airbus, is developing a fuel-conserving alternative to the 787 called the A350.
It, too, will use a lot of carbon composites—and a lot of electricity. And its design, too, relies on lithium-ion batteries.
In mid-February, Airbus said that it would design that airliner's electric system around nickel-cadmium batteries, instead of the lithium-ion ones it had originally planned.
This article was updated on 27 February 2013.