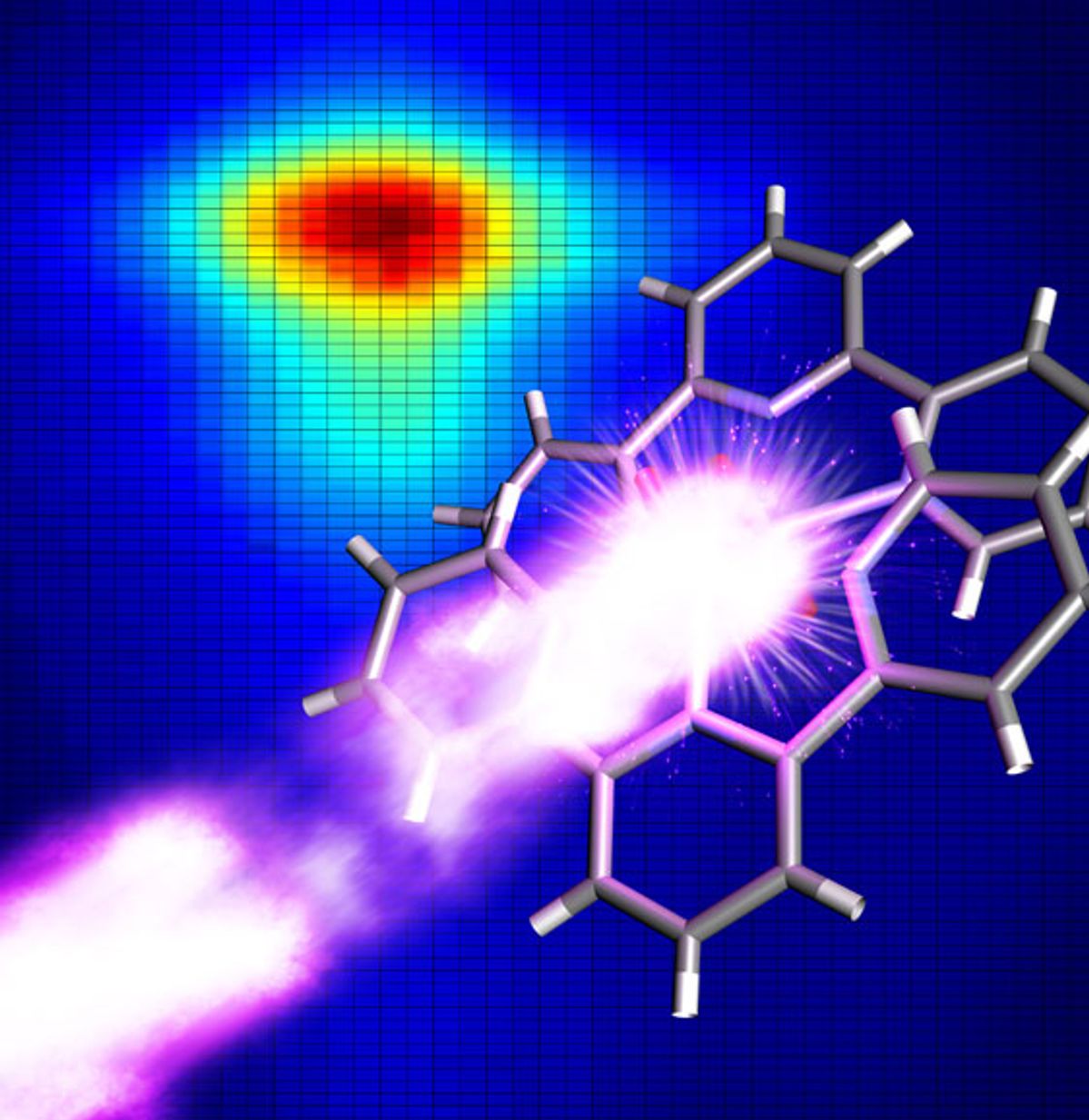
If you wanted to do an imitation—at least an accurate one—you would be well served to carefully observe the original. With artificial photosynthesis being hotly pursued by some of the most renowned nanotechnology researchers, a team at Umeå University in Sweden thought an important step for improving artificial photosynthesis would be to peer deeply into real photosynthesis to reveal the factors that make it work.
While this may sound like an approximation of photosynthesis, the research group had already used LCLS to perform structural analyses of isolated photosynthesis complexes in plants. The new wrinkle this time was to bring spectroscopy into the imaging process.
To accomplish this imaging, the LCLS emits a laser beam with wavelengths that are the breadth of an atom at pulses that last 50 femtoseconds. When the LCLS was combined with the spectrometer, the x-rays emitted from the manganese complex after being hit with the laser pulses are diffracted by the spectrometer and picked up by a detector array.
With this set-up the researchers observed detailed information about the compound’s electronic structure before the laser beam destroyed it.
"Having both structural information and spectroscopic information means that we can much better understand how the structural changes of the whole complex and the chemical changes on the active surface of the catalysts work together to enable the enzymes to perform complex chemical reactions at room temperature,” says Johannes Messinger, professor at the Department of Chemistry at Umeå University, in a university press release.
This detailed imaging of how photosynthesis splits water into its constituent parts has been held out as a way to help engineers more cheaply synthesize hydrogen gas to power hydrogen fuel cells—and possibly the automobiles powered by them. Research efforts to split water molecules into hydrogen gas have been taken on both by commercial entities and the academics. Perhaps this new information on the electronic structure of the water-splitting process of photosynthesis can further inform both these lines of research.
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.