Three-dimensional bioprinting—building tissues by putting down layers of cells and other materials—has led to the manufacturing of human tissues including corneas, skin, and blood vessels.
Now, a team at the University of California, San Diego, is raising the bar. In a paper published this week in the journal Nature Medicine, they describe a 3D-printed spinal cord implant that restored function in the hind limbs of rats with spinal cord injuries.
It is the first 3D printing of a complex central nervous system structure, according to the authors. It was made possible by an advanced bioprinting method called microscale continuous projection printing (μCPP), which prints biological materials roughly 1,000 times faster and at higher resolutions than traditional 3D-printing methods.
The ultimate goal is to create a personalized implant that helps repair an injured spinal cord and then degrades away, says study coauthor Shaochen Chen, vice chairman of the nanoengineering department at UCSD and cofounder of Allegro 3D, a startup commercializing the bioprinting tech. “At the end of the work, we hope that you have exactly a functional spinal cord there—and nothing else,” he says.
The spine is not an easy thing to repair, which is why spinal cord injuries are so devastating and often lead to permanent paralysis. Part of the challenge is that the slender, bundled axons of spinal cord nerves are delicate and finicky. These axons are highly sensitive to materials used in implants or other repair systems. In past studies, the axons will even avoid a material by growing around it or returning back the way they came.
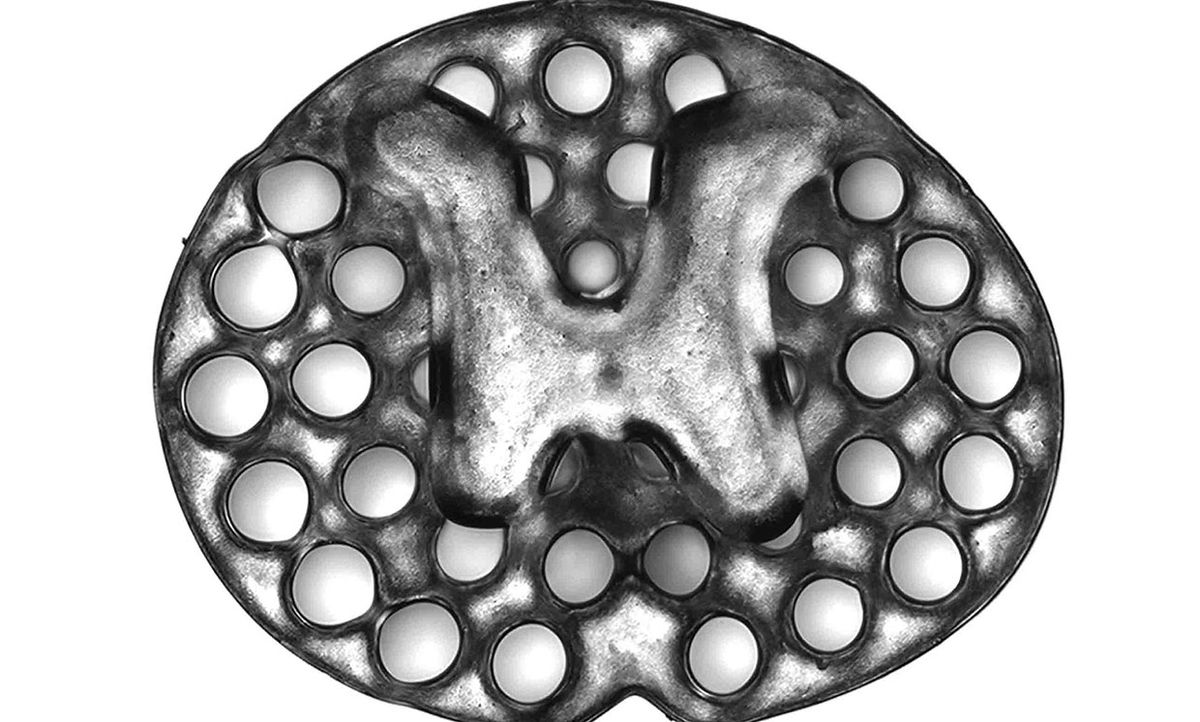
Chen, neuroscientist Mark Tuszynski, and colleagues tested numerous materials to see which would be the most compatible with spinal axons, and settled on a hydrogel called polyethylene glycol–gelatin methacrylate. Using that hydrogel, they designed and printed a rat spinal cord scaffold that looks like a piece of oval Swiss cheese about the thickness of a penny.
Video: Shaochen Chen lab/UC San Diego
To mimic axon-free gray matter, the inner, H-shaped part of the scaffold is solid. To mimic white matter, through which the axons run, the outer part of the scaffold contains 200-micrometer microchannels, like tunnels, to guide the nerves.
Such channels simply can’t be achieved with traditional inkjet or extrusion bioprinting methods, says Chen, as those methods print down to a resolution of only 200 μm. “When you lay down a droplet, that’s already 200 microns. You won’t be able to form a 200-micron pore,” he says. μCPP, on the other hand, prints down to the resolution of a single micrometer.
After the printing process, the team filled the microchannels with neural stem cells to encourage axon growth.
And remember how I mentioned the speed? The μCPP method is fast. A traditional nozzle printer might take several hours to produce a personalized 2-millimeter implant. μCPP does it in 1.6 seconds.
Here’s how: Instead of laying down individual droplets of material like an inkjet 3D printer, μCPP involves shining UV light across an entire plane of material—in this case, a mix of hydrogel and neural stem cells. Guided by digital images, the light fuses together the liquid material to create a solid shape of one’s choice. So instead of forming a structure drop by drop or bit by bit, μCPP creates a complex scaffold of hydrogel and cells in one continuous print.

To prove the technique is scalable to humans, Chen’s team also printed scaffolds to match the size and shape of human spinal cord lesions using real MRI scans. The human implants, about 4 centimeters tall, took just 10 minutes to print.
But back to the rats. With the implants, the rat spines slowly repaired over four weeks as axons grew through the microchannels and into the lower spinal cord. The scaffolds retained their shape at the four-week mark, says Chen, then began to degrade over the following five months, with no sign of inflammation.
The rats with the stem-cell-rich implants regained significant function in their hind limbs compared with animals having an empty scaffold or no scaffold at all.
The team has also used the μCPP method to make human liver tissue, which has a sophisticated microarchitecture, and blood vessels.
As for the spinal implants, “we’d like to do more fundamental studies of the materials and structures, then we’ll move onto larger animal experiments before planning clinical trials,” says Chen. He estimates that human tests could begin in five years.
Megan is an award-winning freelance journalist based in Boston, Massachusetts, specializing in the life sciences and biotechnology. She was previously a health columnist for the Boston Globe and has contributed to Newsweek, Scientific American, and Nature, among others. She is the co-author of a college biology textbook, “Biology Now,” published by W.W. Norton. Megan received an M.S. from the Graduate Program in Science Writing at the Massachusetts Institute of Technology, a B.A. at Boston College, and worked as an educator at the Museum of Science, Boston.