“Batteries are complicated.” This was the unrehearsed refrain I heard repeatedly from Mike Toney and the researchers who make up the Toney research team at the Stanford Synchrotron Radiation Lightsource (SSRL), part of the SLAC National Accelerator Laboratory in Menlo Park, Calif.
During a visit to to their laboratories, I learned that not only are the complex inner workings of batteries being revealed by the assortment of X-ray microscopy tools used at SSRL, but that the latest innovations in photovoltaics are being examined and characterized with the aim of making sure that both energy storage and energy generation technologies can meet the demands of future generations.
“Characterization is perhaps an underappreciated term,” explains Toney. His take on it?
I see it as more that we’re involved in characterization directed at understanding how things work, or how they’re put together. One example of this is batteries, which are presently a quite popular topic. We’re involved in understanding how the lithium ions shuttle back and forth between the anode and cathode and the resulting changes on a very small level. Right now, we are looking at an atom-sized level and how those changes make an impact on the nanoscale level and then eventually understanding how that leads to changes on the electrode level that lead to failure.
While Toney’s team does spend some time working in collaboration with other groups at Stanford—such as the researchers at Yi Cui lab at Stanford Institute for Materials and Energy Sciences—another big part of their research is working with commercial battery and photovoltaics manufacturers who need to know how their devices work on the atomic scale. These measurements are often only possible with the X-rays that are produced when high-energy electrons speed around the synchrotron at SLAC.
Toney calls this work “foundational science research” and involves determining the scientific underpinnings of how batteries and photovoltaics work. This kind of work can take the form of looking at a nanostructure electrode, mixed together with a bunch of carbon in an electrolyte and then reducing that down to a single crystal.
While this structure is not a realistic battery geometry, it does make it possible to simulate a realistic environment in which lithium would move in and out of the nanostructured electrode and reaction layers would form. Combining it with highly sensitive X-ray probes makes it possible to acquire detailed information on how the battery would operate in a real-world conditions. This makes it possible to determine what kinds of charging protocols or chemistries in the electrolyte can be used to basically encapsulate—or passivate—the surface.
Chris Takacs, a post-doc research fellow at SLAC who is a member of Toney’s team, has been testing one company’s batteries using one of these small battery packages and the X-rays from SLAC’s synchrotron. Takacs has devised a special measurement technique using X-ray topography diffraction that he has dubbed depth-resolved X-ray diffraction.
You can see Takacs describe his measurement arrangement in the video below.
Chris Takacs on X-ray Topography Diffractionyoutu.be
“We’re trying to understand how Li-ion concentration gradients build up on commercial battery cells as you charge and discharge them fast,” says Takacs. “We are trying to uncover the limits in these types of performances. So, right now we’re just looking at the cathode. We’re trying to understand if there is an enrichment of the lithium ions near the separator or near the current collector when you’re charging very quickly. This is considered one of the major limitations for the rate at which you charge.”
One of the most promising materials for improving the charge life of Li-ion batteries has been nanostructured silicon. Silicon has been discovered to improve the charge capacity of anodes (negative electrodes) in lithium-ion batteries by as much as ten times over standard graphite-based anodes. Unfortunately, silicon anodes crack and become unusable after a few charge/discharge cycles. This occurs because the material swells and shrinks as the ions shuttle back and forth. It has been hoped that nanostructuring the silicon can reduce or eliminate this rapid cracking. Toney and his team are looking at this problem and others assocated with silicon in batteries.
“One of the problems with silicon is you grow this all-electrolyte interface right at the surface of the silicon that consumes the electrolyte and consumes lithium,” says Toney. “If that grows uncontrollably, all the lithium ends up there and not in silicon where you want it. So we want a passivating layer there that is compliant in the sense that it can stretch. And so we’re in the process of providing some knowledge to guide people and get other researchers to think about what additives we want to add to electrolyte to kind of tune the properties of this layer.”
While polymers have been used as a passivating layer, Toney says that you can also tune the reactions that form to serve as an extremely effective passivating layer.
A great example of this is not in the battery space, but in stainless steel manufacturing, where the the alloying elements are designed to create a passive film on the surface that basically prevents the stainless steel from corroding and gives long life, according to Toney. “This passivating layer has allowed stainless steel to be used ubiquitously in our society,” he adds.
The Li-ion battery has achieved the same ubiquity. As a result, battery research has continued to take on an ever greater share of Toney’s research. He marks the beginning of this surge in battery research with the introduction of the Tesla all-electric vehicles.
“If you had asked me 10 years ago if electric vehicles were possible at a reasonable price, I think I probably wouldn’t have believed it,” says Toney. “But once you start to see the first of the Teslas that came out, you start to realize these are real. And that is kind of about the same time that I think a lot of people start to get interested in battery technology.”
The Li-ion batteries that are currently used in Tesla cars still have some room for improvement, according to Toney. But those improvements will likely remain incremental.
“Over the next few years, we we will continue to see the same incremental five- to 10-percent per year advancements in terms of capacities,” says Toney. “The costs actually have been going down much faster than that. So I would expect that some of those will continue over the next few years, but at some point—at least from the cost perspective—you’re going to hit a limit. There are some predictions that this will happen in two or three years. You really can’t get a battery much cheaper with the current chemistries.”
Alternative chemistries to the current dominant Li-ion battery is another line of research that has taken on increasing importance. Most notable among these is lithium-metal batteries. Johanna Nelson Weker, a staff scientist at SSRL, has been examining these chemistries with the aid of a transmission X-ray microscope.
With this device, Weker essentially takes images of anodes in lithium-metal batteries in situ across a 30-micron field of view and is able to get resolutions down to 30 nanometers.
“We can either look at how their chemistry is changing or how their morphology is changing in 3D,” says Weker. “So 3D imaging is very much like a computerized tomography (CT) scan. We rotate our sample, take images at many different angles, put them into an algorithm, and out comes a 3D image.”
In what is essentially spectral micrscopy, the researchers are to see what all the elements in the battery are doing spatially while it’s cycling. “For example, it will show you whether your lithium is going into your cathode in the core-shell manner—from the outside in—or is it going to smaller particles first and ignoring the large particles. Or it will show you whether every particle just transforming simultaneously together both in both large and small particles,” explains Weker.
In the video below, you can see Weker describe what they discovered when looking at how these particles lithiate in a lithium-metal battery sample.
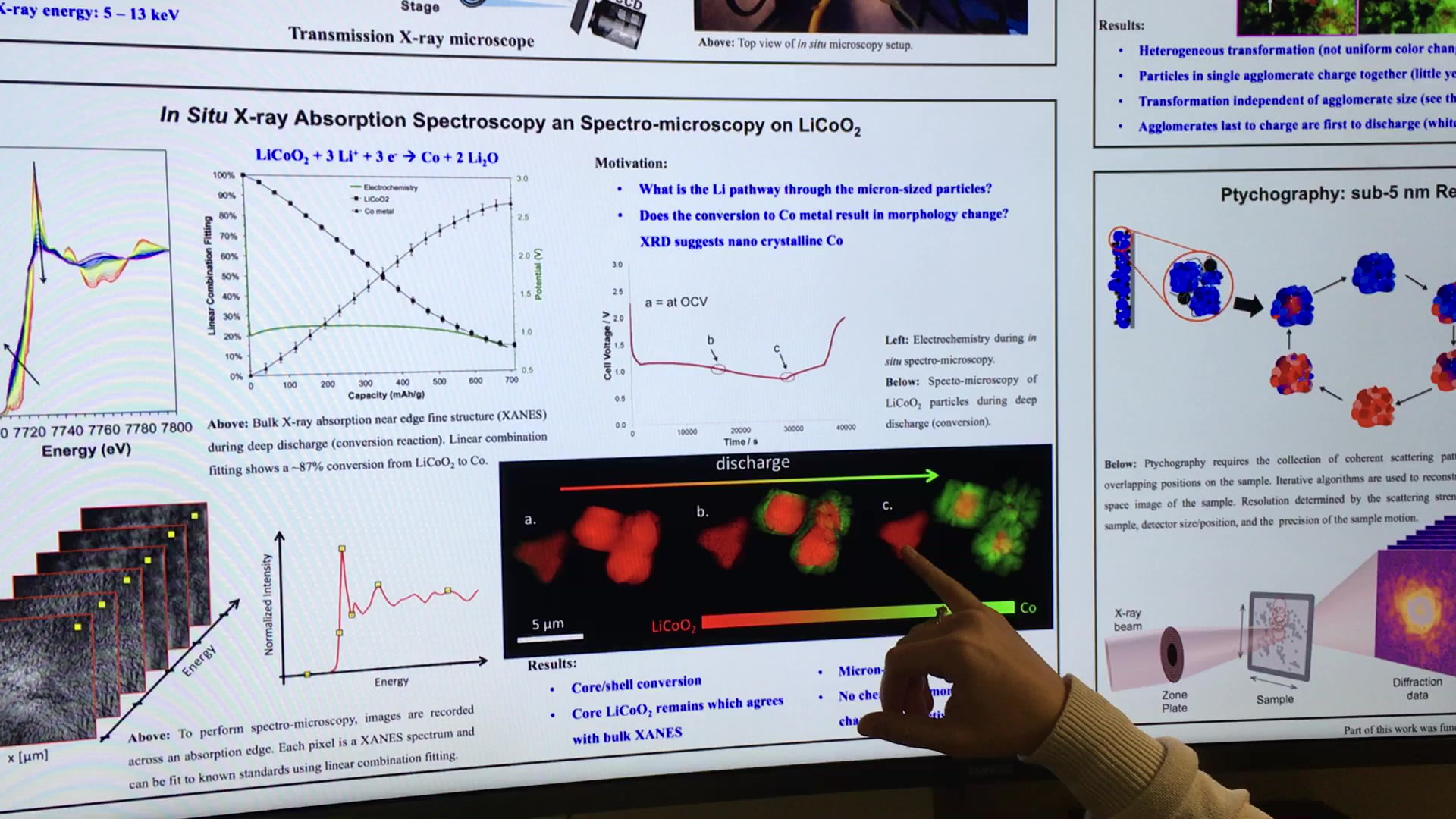
Weker’s work has revealed that a long held assumption about how these particles lithiate was wrong. The core shell of these particles does not always lithiate.
“What we found with lithium-iron phosphate is it doesn’t act that way,” she explains. “In the nanoparticles, there’s a preferential direction depending not only on the crystalline lattice planes, but also basically one particle will start to lithiate. And all the neighboring particles will not and they actually donate their lithium to that particle so the lithiation occurs one particle at a time.”
Not all the members of Toney’s teams are using X-rays for examining energy storage technologies. Some are looking at the cutting edge materials used in photovoltaics, such as halide perovskites.
Aryeh (Ari) Gold-Parker, a PhD student at Stanford working with Toney’s team, is employing an X-ray absorption spectroscopy technique to identify certain elements that are in the sample and identify the relative quantities of those elements in order to understand something about the chemical environment in which the atoms of those elements reside.
The starting material for halide perovskites involves quite a bit of chlorine in addition to iodine, but it’s widely understood that once you’ve fully prepared the film by heating it, almost all of the chlorine is gone. So what you’re left with is primarily iodine. What Gold-Parker did was to prepare the film while heating it and keep it on the X-ray beam line.
This arrangement allowed him to monitor the chlorine leaving the film as it was being heated and also discover the fact that the chlorine atoms in the film were going from one local environment to another throughout the heating process. Gold-Parker believes that this will prove extremely useful for trying to understand how these perovskite films form and might eventually help to optimize the actual solar cell performance.
“At the end of the day, the real hope is to make high-efficiency devices,” says Gold-Parker. “There are a lot of engineers who are fine with all these different chemical compositions for this material system, just swapping in all different elements and molecules in the different sites trying to achieve the highest efficiency. But the field has gotten way ahead of the basic science. So the engineers might be two or three years ahead of the actual understanding of why these atoms are making a difference.”
This is what Toney might describe with his term “foundational science research”. Instead of hit-and-miss iterative processes, Toney and his team are trying to uncover the fundamental chemistry and physics that make our next-generation energy storage and generation systems operate. This will likely make them better in just about every performance metric and also make them cheaper to produce.
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.



