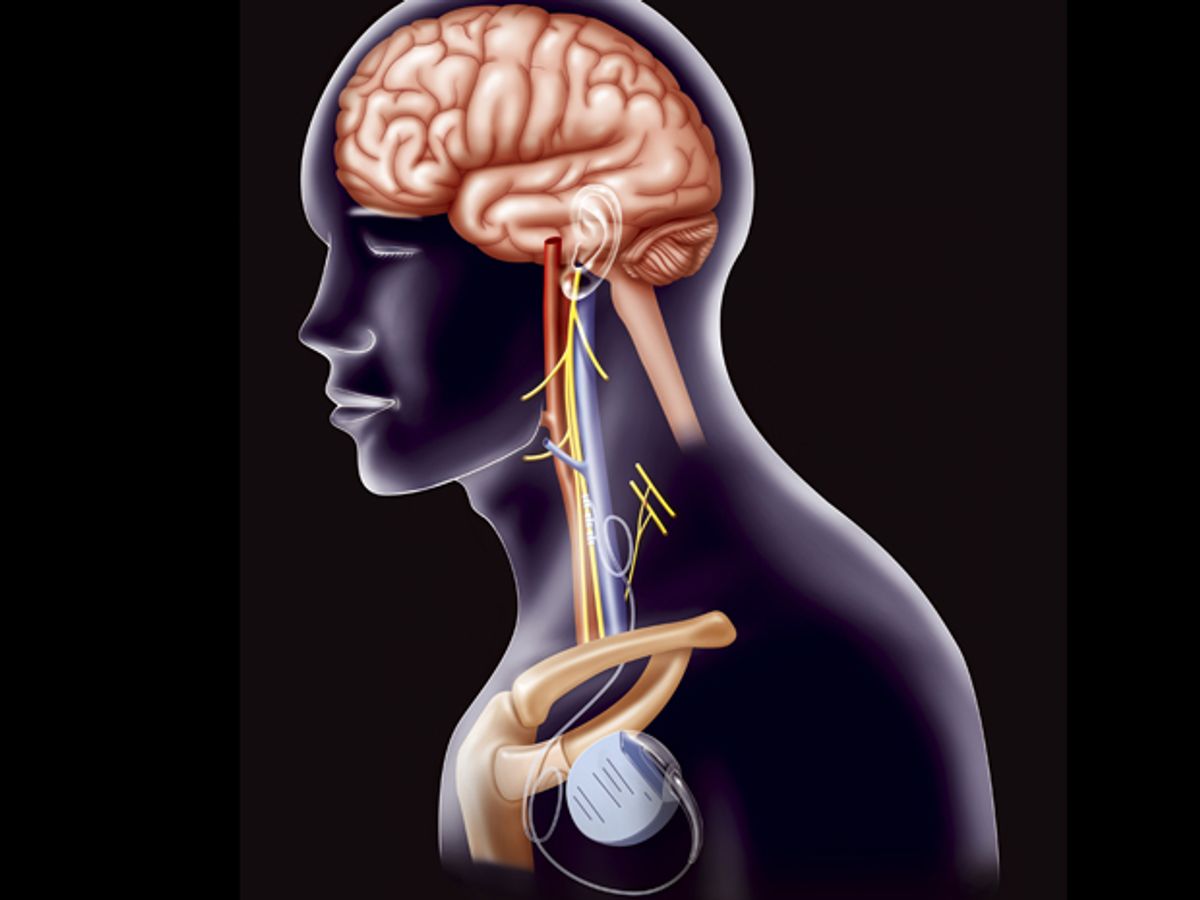
Both trials used implanted electric stimulators that attached to one of a pair of key nerves in the neck, the vagus nerve. The nerve connects the brain to the heart, stomach, and other organs. It's a key target for medical device developers because it allows convenient access to the brain to treat disorders of that organ, such as epilepsy and depression, as well as to gut organs to treat other problems, such as obesity.
Heart failure, the inability of the heart to pump enough blood to satisfy the body's needs, has also been on the list of targets. The failure of Boston Scientific's trial was a big surprise to its leaders. "There is robust pre-clinical data showing the benefit of [vagus nerve stimulation], but the NECTAR-HF trial failed to demonstrate a successful clinical translation of this protocol," Faiez Zannad an investigator in the trial at l'Institut Lorrain du Coeur et des Vaisseaux Louis Mathieu, in Vandoeuvre-lès-Nancy, France, said in a press release.
Boston Scientific was hoping to see a key marker of improvement—a decrease in the size of the left ventricle when it's fully contracted. They were also looking for other heart dimension measurements, as well as improvements in exercise capacity and in levels of the hormone tied to heart failure.
The results were especially disappointing because of the care used in designing the study. 96 heart failure patients were recruited from across Europe, and all received implants for their right vagus nerve. Two-thirds had the stimulator turned on, while one third acted as controls with the stimulator off. All received the best standard treatment for heart failure during the following six months. After that point the controls, too, had the stimulator switched on.
Zannad suggested that the failure to see results may have been because the treatment group was already pretty well managed using standard medical therapies or because six months is too short a time period. The amount of current delivered might also have been too low.
The Cyberonics trial also followed patients for six months, but it was less convincing because it lacked a control group that received implants but no stimulation. Together, the 60 patients averaged a 4.5 percent improvement in left ventricular ejection fraction—a measure of how much blood makes it out of the left ventricle when it contracts.
"I think, at six months, that [4.5 percent improvement] is a very impressive achievement," study leader Inder Anand from the University of Minnesota told reporters.
Clearly, more clinical trials will be needed to see if this therapy can make a difference. But, following the failure of Boston Scientific's trial, whether or not to pay for that research will likely be a more difficult question.
Samuel K. Moore is the senior editor at IEEE Spectrum in charge of semiconductors coverage. An IEEE member, he has a bachelor's degree in biomedical engineering from Brown University and a master's degree in journalism from New York University.