Tapping the Power of 100 Suns
Concentrated solar power will keep future armies on the march

Napoleon’s dictum no longer applies: These days, an army marches not so much on its stomach as on its batteries. Without them, soldiers can’t see in the dark, work their radios, or determine their positions. But even the best storage batteries—accounting for one-fifth of the load in a typical infantryman’s 45-kilogram pack—can’t last the week or so that field soldiers require. The same problem is coming to afflict the rest of us, as we become ever more dependent on our smartphones and GPS navigation.
While we wait for better batteries we must find new ways to recharge the ones we have when we’re far from a wall socket. What we need is a really good, portable photovoltaic system, one that can take in a huge gulp of sunlight and convert most of it into electricity. Such an advance could also help drive down the cost of solar electricity in sunny climes around the world and prevent great quantities of carbon emissions.
The most promising effort to create such superefficient photovoltaics began in 2005, when Doug Kirkpatrick, a veteran of the optics industry, kick-started the Very High Efficiency Solar Cell (VHESC) program for the U.S. Defense Advanced Research Projects Agency (DARPA). He wanted a way to build modules from solar cells that would convert a full 50 percent of the solar energy they receive into direct current. That’s a jaw-dropping number when you consider that in 2005 the best laboratory devices were still shy of 40 percent efficiency and were improving by less than one percentage point per year.
Kirkpatrick considered the needs of the end user—the soldier. He noted that each electrical gizmo offered a different-size area for placing solar cells. This would make it possible to put more cells on bigger gadgets and use them to charge up other gizmos, such as flashlights, which have very little real estate. But that was a risky strategy, because the failure of one piece of equipment would put the others out of commission, too. Therefore, Kirkpatrick decreed that each piece of gear would have its own solar power, limiting the solar module’s area to 10 square centimeters and setting cell efficiency at a minimum of 50 percent. The only other key specification for the module was power, which had to be at least 0.5 watts to recharge the batteries of the more common gadgets in an acceptable time.
The program started on the wrong foot by encouraging researchers to build on DARPA’s existing programs to use biological processes to build solar cells. However, the agency abandoned that tack when two electrical engineers, then at the University of Delaware, wrote a white paper outlining a new way of using existing photovoltaic cells.
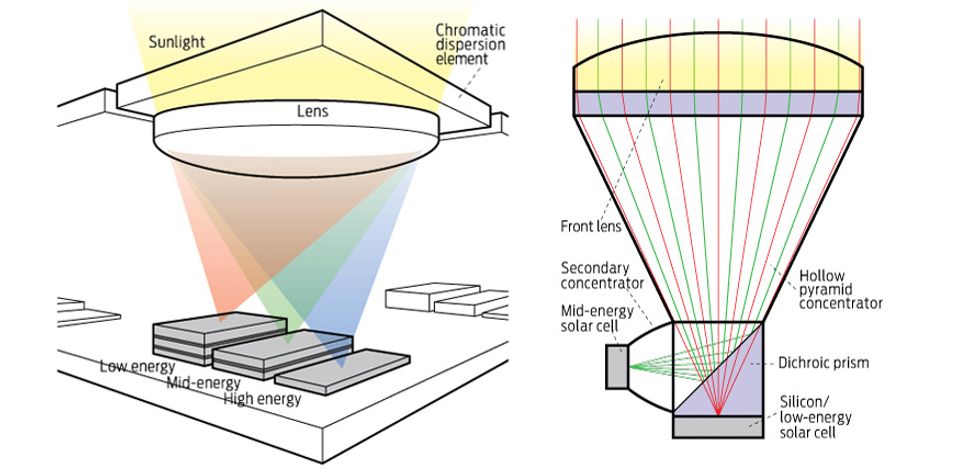
Christiana Honsberg, now at Arizona State University, and Allen Barnett, now at the University of New South Wales, in Australia, argued that the best approach would be to concentrate sunlight, break it into its constituent colors, and project each color onto the kind of solar cell best suited to it. This meant placing the cells side by side instead of the conventional way, in a vertical stack. Their paper went down very well, and DARPA invited the Delaware duo to a meeting where they could pitch their idea alongside 10 others. “A lot of people there were saying that we can’t do 50 percent, but we could do 33 percent,” says Kirkpatrick. “We were not interested.”
Because the Delaware team wasn’t banking on the success of a particular material but rather on a way of exploiting a diversity of materials, there could be many different paths to success. That was another reason DARPA put Honsberg and Barnett in charge of VHESC, which has become one of the largest solar-energy research programs ever. Initially backed by US $53 million, this effort, which commenced in September 2005, involves a consortium that at one point numbered 21 institutions, including BP Solar, Corning, and DuPont, and such universities as Georgia Tech, Harvard, MIT, Purdue, the University of California, Santa Barbara, and the University of Rochester.
There’s no better way to appreciate the merits of Honsberg and Barnett’s approach than by considering how far short of the 50 percent target you fall using conventional tricks. It all comes down to an optimization problem, rather like that of a coffee-shop manager who has to decide how much to charge. If you set the price high, you make a good amount on each cup but sell few of them. If you set it low, you’ll have a slim margin but a high volume. You need to find the sweet spot, the price that maximizes the product of the margin and the volume—that is, your profit. It’s a similar story with solar cells, though here the variables are current and voltage, whose product, of course, is power.
At the heart of every photovoltaic cell is a pair of semiconductor layers. One layer has more negatively charged electrons than needed to form the material’s crystalline bonds; thus it’s known as n-type. The other, a p-type material, has a deficiency of such bond-forming electrons, creating what are known as holes. In a small region around which the layers meet—the p-n junction—the excess electrons move to fill in the deficiencies. This flow of electrons and holes leaves the p-type and n-type regions with negative and positive charges, respectively, creating an electric field.
When the cell absorbs photons, it creates pairs of electrons and holes. Then there is a net movement of holes in one direction and electrons in the other, due to both the electric field itself and to diffusion—the tendency of charge carriers to move to where their concentration is lower. An electric current is thus created in the circuit attached to the cell.
Solar-cell designers might be tempted to pick a semiconductor that best absorbs light of short wavelengths, like gallium nitride. Those photons carry the most energy and thus produce the highest voltage. Trouble is, there aren’t many of them in sunlight, so the output current will be meager. Alternatively, designers can build cells from mercury cadmium telluride, a material that can absorb the solar spectrum all the way from the high-energy photons in the ultraviolet range to the low-energy photons in the short-wave infrared. Such a cell can indeed generate a lot of current, but its voltage—and hence its output power—is very low. So like the coffee-shop owner, solar-cell designers need to find a good compromise.
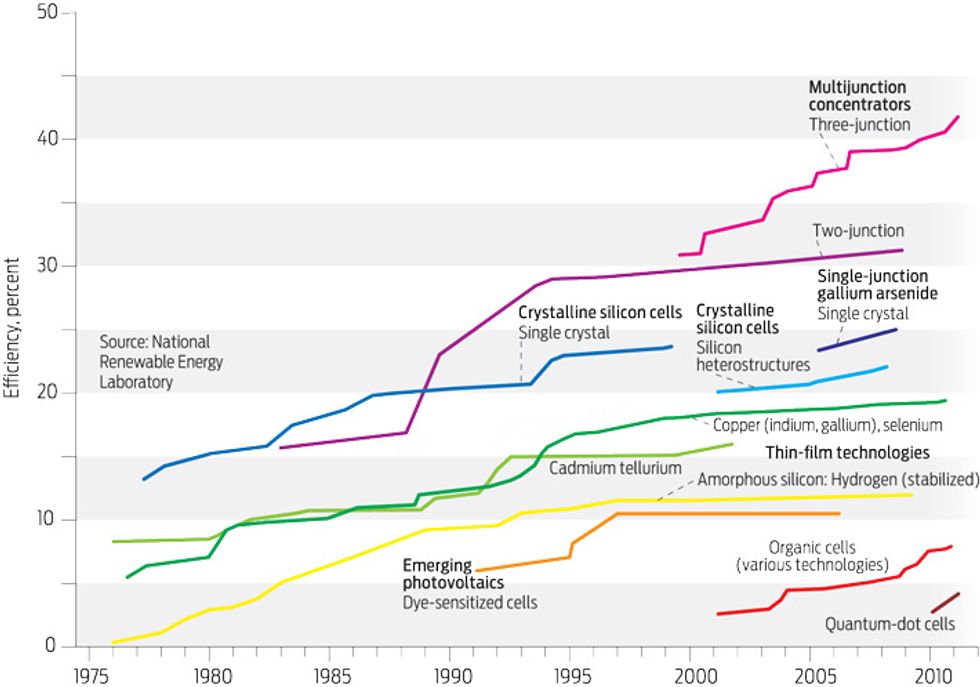
Theoretically, a semiconductor that absorbs light at infrared wavelengths greater than 910 nanometers will come closest to the sweet spot where power is maximized. Gallium arsenide fills the bill, and back in June 2005 researchers at Radboud University Nijmegen, in the Netherlands, were using this material to claim a single-cell efficiency record of 26.1 percent. Today, Alta Devices of Santa Clara, Calif., holds the record with a 28.2 percent cell. That’s still a long way from Kirkpatrick’s 50 percent target.
To close the gap requires a diversified strategy. Instead of using one material, a solar-cell maker can build cells from several. It’s rather like offering coffee in three grades—a deluxe brand, a bargain brand, and a house brand—so as to hit profit-maximizing sweet spots for the rich customers, the poor ones, and everyone in between. The practical limit—for use in photocells, at least—is three different materials, given the traditional way of stacking them up.
To capture photons of relatively low energy, cell makers most often use a layer of germanium. For photons of intermediate energy, they include a gallium arsenide layer, and for high-energy photons, they create a third layer made up of indium gallium phosphide. The voltages simply add.
In theory, this strategy would work even better if you added more layers. But in practice, the material quality of the device tends to deteriorate, although some companies, such as Stanford University spin-off Solar Junction, have been working on approaches that promise to produce up to six stacked junctions.
The efficiency of triple-junction solar cells has been slowly creeping up. When the VHESC program kicked off in 2005, Spectrolab (a Boeing subsidiary in Sylmar, Calif.) led the way with a device boasting 32 percent efficiency, which the company had developed to power satellites. Now the Japanese firm Sharp holds the crown, with a device operating at 36.9 percent efficiency. It uses indium gallium arsenide, rather than germanium, for the bottom junction.
The other big factor governing the financial success of any coffee shop is advertising. Good signage will pull in more traffic and should lead to more sales. In the case of solar cells, that means replacing a large triple-junction device with a lens or mirror that captures sunlight over an area just as great and focuses it on a photovoltaic chip that’s much smaller and far cheaper to produce. Just as important as the reduction in cost is the increase in the efficiency and output power of the cell. When light is concentrated by a factor of 20, it doesn’t merely create 20 times as many photons—it also boosts the output voltage, though it takes rather a bit of math to explain why this is the case.
In 2005, Spectrolab led the way with this approach, producing 39 percent efficiency with a triple-junction cell under the equivalent of the light coming from 236 suns. This bar has been raised many times. In 2011, Solar Junction claimed the most recent record of 43.5 percent cell efficiency at the equivalent of 400 suns, and this June, Sharp matched this figure with a cell operating at an undisclosed concentration.
To hit these figures, the cells must be mounted on systems that track the sun’s position in the sky with tremendous precision. The smallest of such systems are several meters high and wide, which makes them suitable for solar farms but not as attachments to a soldier’s gear. If concentration is to be used in portable solar systems, the factor of magnification must be far more modest.
Forty-three percent is pretty good, but it’s not the desired 50. Further progress required a radically different approach, the one that Honsberg and Barnett had suggested to DARPA five years earlier. Rather than stack cells vertically, you tile them laterally, then split sunlight into several energy bands and focus each of them on different tiles.
Switching from a multijunction stack to a side-by-side array increases the maximum theoretical efficiency. That’s because in a stacked system each layer is normally connected in series and thus has to produce the same amount of current. Designers can adjust layer thicknesses and other parameters to try to meet this requirement, but that’s hard to do perfectly because the solar spectrum varies throughout the day—it’s redder in the morning and late afternoon, bluer at midday. So a triple-junction cell’s current is often limited to whatever the least productive layer can produce.
Also, stacked cells are limited by the way the stacks must be put together. To form these devices, crystalline layers are deposited atom by atom, and the spacing of the atoms in each layer must be very similar to avoid crystal imperfections. This limits the number of material combinations—and therefore the range of absorption wavelengths—that can be used in a multijunction cell. By avoiding stacks, Honsberg and Barnett’s approach allows materials with vastly different atomic spacing to be used alongside one another without any compromise in quality.
Calculations show that without concentrated sunlight, efficiency should rise from 51.5 to 55.6 percent when you shift from three junctions to four. And moving to five or six junctions should raise efficiency to 58 percent and 59.6 percent. Real-world gains would of course be lower, but still, upping the concentration should assure good results. Barnett and Honsberg estimate that for a six-junction cell operating at 20 suns you get an efficiency of 54.3 percent, and at 100 suns, 55.6 percent. With much more concentration than that the cell might overheat, and it would be difficult to point the mirrors precisely.
Barnett and his colleagues initially developed tracking-free solar modules using concentrations of 20 suns. A mobile user could manually point the cells toward the sun.
Although in theory it should improve efficiency, splitting the solar spectrum into six portions and focusing each of them onto a separate cell is actually taking things too far, because substantial losses result from steering that many beams of light. “Ideally, we like to separate them into three ‘buckets’—high energy, mid-energy, and low energy,” explains Barnett. Each bucket contains up to three cells, which means it’s also a stack, but one that isn’t connected in series. That way, you don’t have to worry about matching the current. Stacks are required because it’s the only way to get to the five or six junctions you need to achieve 50 percent efficiency.
Deciding on the best way to get the sunlight into these buckets is tricky. At a meeting in 2006 at the University of Rochester, in New York, Kirkpatrick proposed to do it by using coatings that consist of finely built-up layers with different refractive indices that together create the desired interference patterns, much like those seen on the surface of a thin film of oil. Such dichroic designs let a range of wavelengths through while reflecting others with nearly perfect efficiency. This strategy would allow you to send the reflected light into one bucket and transmit light elsewhere, either onto cells or through another dichroic film for further spectral splitting. So far, designs have used only two buckets because the performance of the high-energy bucket isn’t yet good enough to bother including it.
“Everybody was saying, ‘No, no, no, that cannot work. You’re wrong,’ ” Kirkpatrick says. The naysayers’ skepticism reflected their background: They were aware only of incredibly precise dichroic coatings used in lasers. Kirkpatrick, however, knew that dichroics are also used in ceiling lamps in Europe to prevent filaments from overheating and thus failing prematurely.
Kirkpatrick demonstrated the capability of dichroic coatings by arranging for some of them to be made by Fiberstars (now Energy Focus) of Solon, Ohio. This firm joined the VHESC program shortly after the Rochester conference.
The initial design concept for the module—one that was never built—featured an optical element to split the sunlight into three spectral buckets and a lens the size of a thumbnail to focus it onto the low-energy, mid-energy, and high-energy cells. The modules were tiled, forming an array.
The choice for the mid-energy bucket was rather obvious: an indium gallium phosphide cell in front with a gallium arsenide cell behind. Together they captured wavelengths from the near infrared to the green part of the spectrum, where the high-energy bucket began. But it was harder to fill the low-energy bucket, which stretched from the short infrared to the near infrared, and the high-energy bucket, which stretched from the green through the ultraviolet. Silicon works well in the infrared and can be partnered with germanium-based compounds, but it works even better when paired with indium gallium arsenide phosphide and indium gallium arsenide, because this combination generates some of its electricity at a higher voltage. There are very few good candidates for the high-energy bucket. Materials based on gallium nitride are the most promising, but they are dogged by problems such as dislocations in the crystal lattice.
Because of the difficulties with the high-energy bucket, the VHESC program has so far developed optical modules with merely low-energy cells, together with mid-energy variants that also capture some high-energy photons, although not as efficiently. In 2007, researchers measured the performance of cells made from indium gallium phosphide, gallium arsenide, silicon, indium gallium arsenide phosphide, and indium gallium arsenide, then calculated the contribution that each would deliver to a five-junction cell operating at 20 suns. They concluded that such a device could have a 42.9 percent efficiency, well ahead of the 40.7 percent record then held by a Spectrolab cell operating at a concentration of 240 suns.
Since then the program has defined its target differently, aiming not for a cell efficiency of 50 percent but rather for a module efficiency of 40 percent. This makes sense: Modules are always less efficient than cells, partly because imperfections prevent their optics from directing all the incident sunlight on the cell. And it’s the output power of the modules that matters. The 40 percent target is still plenty tough, given that the best commercial triple-junction photovoltaic modules have efficiencies of just over 30 percent when operating at concentrations of several hundred suns.
By 2008 the consortium had set a new benchmark for efficiency with cells that collect light with one lens and then split the spectrum with a dichroic mirror onto tandem cells, one absorbing ultraviolet and visible light, the other absorbing infrared light. Operating at 20 suns, the device upped the record for module-wide efficiency from 32.6 percent to 36.7 percent when tested independently at the National Renewable Energy Laboratory (NREL). One hit 39.5 percent efficiency in tests at the University of Delaware. Differences in the values reflect disparities in the measurement of the aperture above the focusing lens, misalignment of the cells, and variations in individual cells.
A road map to the 40 percent efficiency mark and beyond followed. Options include turning to more highly reflective coatings on the focusing lens; enshrouding the optics in silicone to reduce internal reflections; adding antireflection coatings to the individual cells; and improving the optical alignment of the module as a whole. The team recently implemented some of these ideas, increasing the efficiency of the modules measured at NREL to 38.5 percent.
“We moved away from silicon to hit the big number,” admits Barnett, who is not a fan of records but acknowledges the good publicity they bring. More headline-grabbing success is now within reach, because Barnett believes that a recently fabricated, high-quality gallium phosphide cell could be inserted in front of the mirror, increasing module efficiency beyond 40 percent.
For the time being, though, the researchers are concentrating less on efficiency and more on economics, trying to beat the cost per watt that all-silicon cells now provide. According to Dan Laubacher, a research fellow at DuPont, one of a handful of firms responsible for building the first prototype modules, costs can be trimmed by using silicon germanium cells for the low-energy bucket alongside cheaper dichroic coatings that sacrifice little in terms of performance.
The VHESC program is clearly on its way to providing a portable, affordable source of solar power. Soldiers will be the first to reap the benefits, but not the last. With such modules in hand, it’ll be possible to carry the makings of a solar farm on a few trucks and set it up quickly. The result? A lot of electricity, with a relatively small footprint—not just for field soldiers but also for explorers, off-the-grid environmentalists, and isolated rural communities. It would shine wherever the sun does.
About the Author
Richard Stevenson returns to photovoltaic cells in “Tapping the Power of 100 Suns,” a topic he last covered for Spectrum in 2008. His Ph.D. in physics from the University of Cambridge centered on the compound semiconductors used in such PVs.