With researchers still struggling to open a band gap in graphene at room temperature sufficient for transistor applications, it’s sometimes good to remember what makes graphene such an appealing material that they think it's worth the struggle—after all, an awful lot of effort has gone into engineering a band gap into a material that intrinsically doesn’t have one. High among those benefits is the promise of high electron mobility and simpler chemical doping techniques—with a far easier path to interconnection than its cousin, carbon nanotubes.
To buoy hopes of graphene in transistor applications, researchers at Aalto University in Finland and Utrecht University in the Netherlands have demonstrated the ability to create single atom contacts between gold and graphene nanoribbons.
The research ("Suppression of electron–vibron coupling in graphene nanoribbons contacted via a single atom"), which was published in the journal Nature Communications, showed that contacts between graphene and gold could be established without significantly modifying the very electrical properties of graphene’s honeycomb lattice that make it so attractive in the first place.
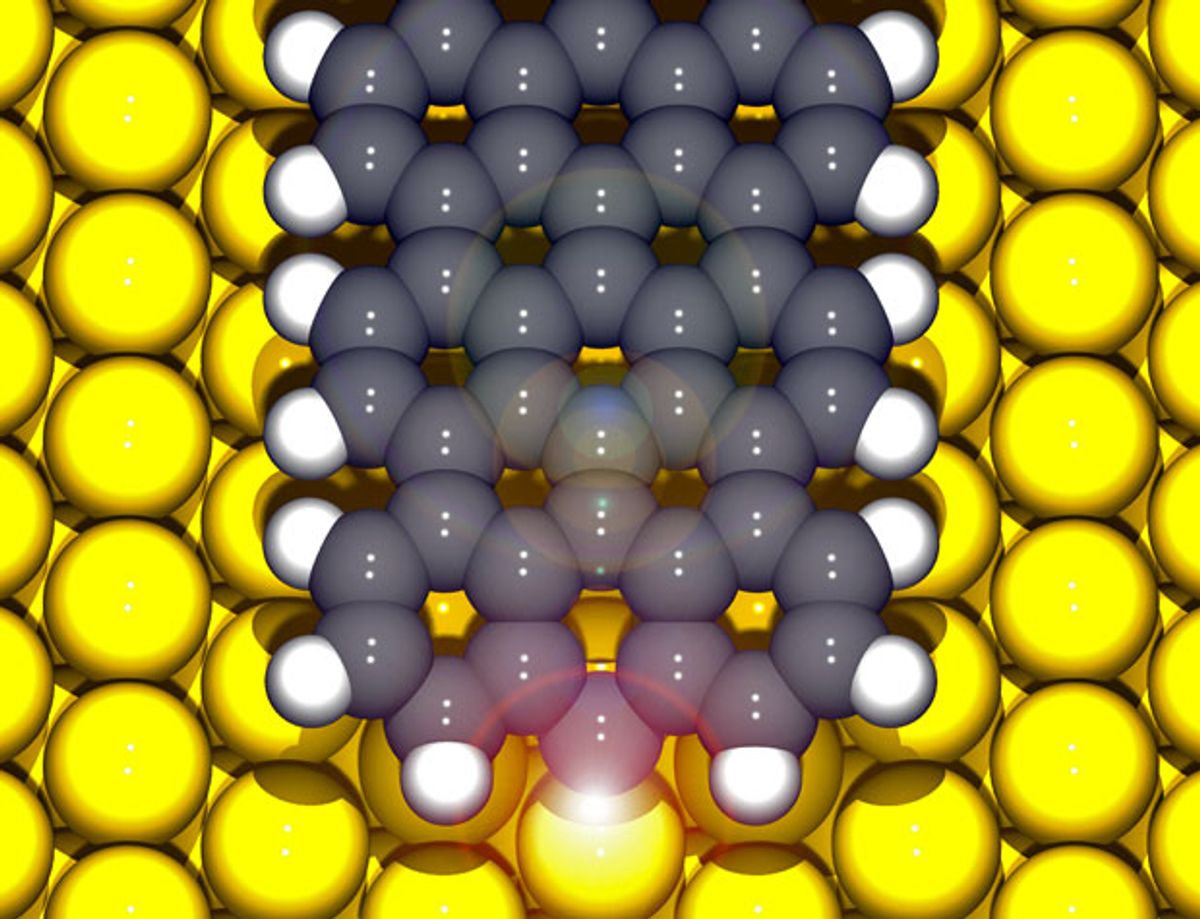
The process starts with an atomic-scale mapping of the graphene using atomic-force microscopy (AFM) and a scanning tunneling microscope (STM). Then a chemical bond is achieved by sending voltage pulses from the tip of a STM to create single bonds to the graphene nanoribbons at precisely determined locations. The pulse from the STM removes one hydrogen atom from the end of the graphene nanoribbon, initiating the bond formation.
"The edges of the chemically synthesized ribbons that we use are hydrogen terminated just as you would have in a molecule (e.g., pentacene)," Professor Peter Liljeroth, who heads the Atomic Scale Physics group at Aalto University, explained to me in an e-mail. "We can use bias voltage pulses from the STM tip to knock off the hydrogen atoms one-by-one. When you remove a single hydrogen, you form what a chemist would call a radical and a physicist would call a dangling bond: the carbon atom without the hydrogen has an unpaired electron that would like to form a bond with something. It does this with one of the atoms of the underlying gold substrate. So we remove the hydrogen, the carbon atom becomes more reactive and forms a bond spontaneously with one of the gold atoms."
“Combined AFM and STM allows us to characterize the graphene nanostructures atom by atom, which is critical in understanding how the structure, the bonds with the contacts and their electrical properties are related,” says Dr Ingmar Swart, who leads the team concentrating on STM and AFM measurements at Utrecht University, in a press release.
By employing advanced computer modeling with their physical experiments, the research team determined that bonds do not affect the overall electronic structure or the intrinsic ribbon properties of the graphene nanoribbons.
In addition to demonstrating that graphene could be bonded with gold on this atomic scale, the research looked into more theoretical concepts for the kind of electronics that graphene could enable. One quite often mentioned in the literature but not yet realized in experiment is an idea called “valleytronics” in which low energy electrons in graphene can have two different momenta (there are two valleys, K and K’, in the graphene band structure) and the idea would be to use this valley degree of freedom in much the same way spin is exploited in spintronics. In this theoretical design a graphene device would be required that could force the electrons only to occupy one of the valleys; this would be called a valley filter.
"There are also some other ideas on how you could use graphene to make electronic devices that don’t have an analogue in existing silicon technology," Liljeroth also told me in his e-mail. "To realize these experimentally, one would need atomically well-defined graphene structures (certain type of edges, widths etc.). This could be either made entirely out of the graphene (and then talking of a “contact” between the leads and the active part of a device is a bit funny) or one could have a graphene nanostructure made by other means (e.g., some kind of bottom up approach) and then connect it to external leads. In this case, the contact would have to be well-defined at the atomic scale. This would be important not to alter the properties of the graphene nanostructure (or to alter them in a controlled manner)."
While this research has helped to open physical experimentation for theoretical uses of graphene in electronics, the recent development of being able to produce graphene nanoribbons in bulk means that the ability to make chemical bonds between graphene and gold promises to be significant in electronic applications of today.
Image: Peter Liljeroth (Aalto University) and Ingmar Swart (Utrecht University)
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.