Researchers only recently discovered distinct temperature gradients inside cells, but the measurements were at the limits of available techniques. Now, newly reported spintronic approaches promise to drive down the limits of thermometric and spatial resolution to allow detailed mapping of cellular thermodynamics.
Last year, a group of researchers from the Universities of Tokyo and Kyoto and the Nara Institute of Science and Technology first showed that cell nuclei are relative hotbeds of activity, running at an average 0.96°C warmer than the cytoplasm—and the temperature gradient varies during the cells’ division cycle. The 2012 study, though, depended on a fluorescent polymer thermometer with a temperature resolution of about 0.2°C and a spatial resolution limited by diffraction to about 200 nm. (Human cells range in size from the red blood cells’ 8 micrometers in diameter to the ovum’s 130 microns.)
Now a team from the University of California at Santa Barbara, the U.S. Department of Energy’s Ames Laboratory, and the University of Chicago—led by David Awschalom of UCSB and UC—has shown that fluorescent defects in diamonds could furnish a tool for far more precise measurements, with temperatures accurate to within a few thousandths of a degree Kelvin and positioning resolution limited only by the size of the probe.
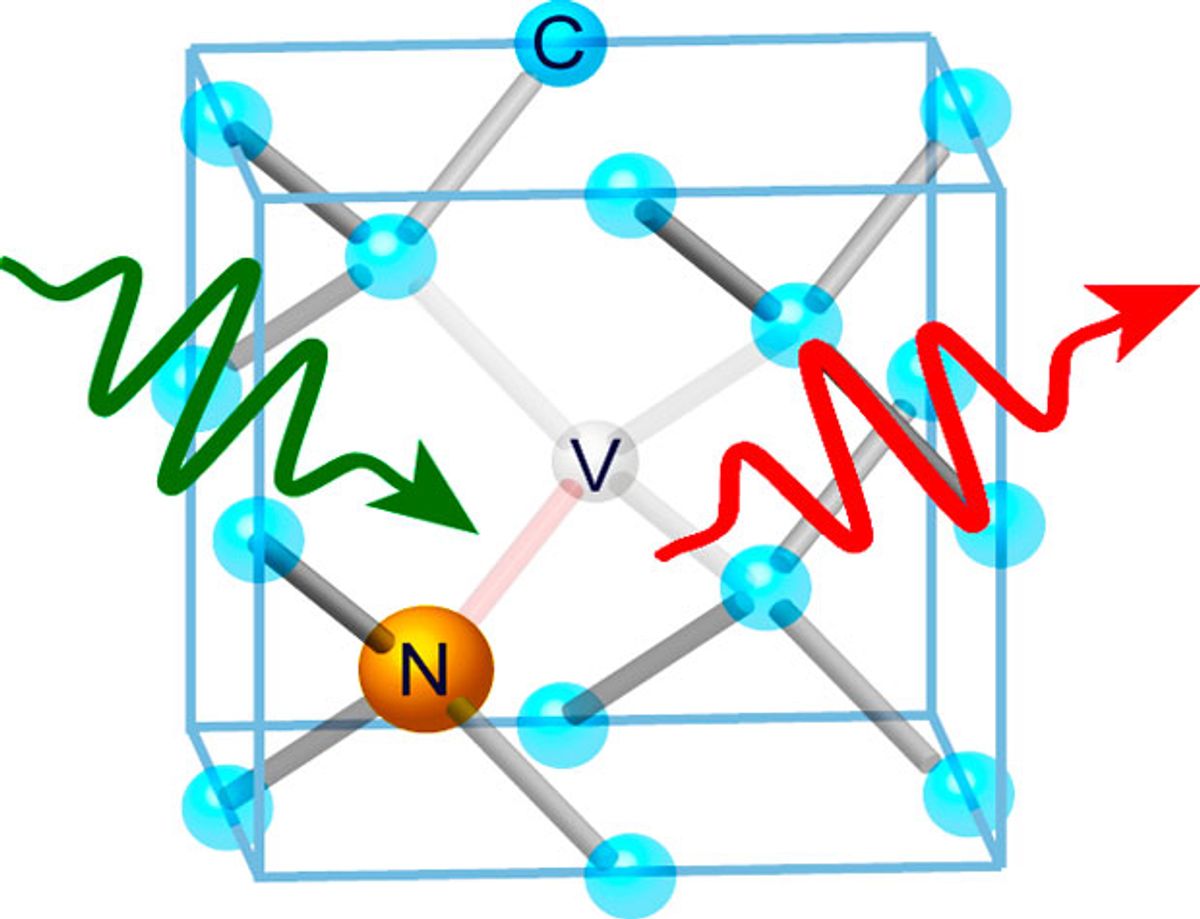
The key is a nitrogen vacancy defect in the diamond: a nitrogen atom replaces the carbon at one node of the lattice, disrupting the structure and leaving an adjacent node empty. As David Toyli (the lead author of the Proceedings of the National Academy of Sciences paper) points out, “The vacancy results in dangling bonds containing electrons from the three nearby carbon atoms and the adjacent nitrogen.”
The six dangling bonds (three from adjacent carbons, two from the nitrogen, and one trapped from the lattice) create, in effect, a negatively charged virtual molecule with three possible electron spin states (+1, 0, and -1). When stimulated by laser light, a single defect emits detectable fluorescence.
In some crystals, the bonding structure splits electron energy states (crystal field splitting), so that different spin combinations have orbital energies—and thus different fluorescence signatures. The diamond nitrogen vacancy is a case in point. This spin-dependent fluorescence is what makes diamond nitrogen vacancy defects an interesting candidate for qubit storage.
The diamond nitrogen vacancy defect’s fluorescent frequency also varies with temperature and magnetic field, and researchers can read signals from a single defect. Taken together, this also makes the defect a prime prospect for nanoscale metrology.
Around room temperature, the frequency of emitted fluorescent light drops by 74 kHz for every additional degree Kelvin. The temperature effect increases to -140 kHz/K around 500 Kelvin (226°C). Resolution, however, is a function (the square root, in fact) of the “read” time. The UCSB group used a variety of techniques to extend spin coherence times to more than 80 microseconds, a 45-fold improvement that translates in to a seven-fold increase in temperature sensitivity, to 0.1 K or less. And, says Toyli, extending measurements to 1 s would allow researchers to resolve temperatures to within about 0.01 K.
“With the same sensor,” Awschalom says, “you could measure magnetic fields, electric fields and now temperature, all with the same probe in the same place at approximately the same time. Perhaps most importantly, since the sensor is an atomic-scale defect that could be contained within nanometer-scale particles of diamond, you can imagine using this system as a thermometer in challenging environments such as living cells or microfluidic circuits.”
The authors note that their current work is based on measurements from a single nitrogen vacancy. The recent construction of good synthetic diamonds with multiple coherent nitrogen vacancy centers could improve sensitivity and reading times. (For ArXiv papers on other approaches to nanothermometry via diamond nitrogen vacancy defects, see here and here.)
Image: University of California at Santa Barbara
(Corrected 6/27 to revise 1 s temperature sensitivity and correct typo.)
Douglas McCormick is a freelance science writer and recovering entrepreneur. He has been chief editor of Nature Biotechnology, Pharmaceutical Technology, and Biotechniques.