Getting clean water for drinking and agriculture to a burgeoning population is one of the most pressing challenges of this century. A natural place to turn to is the world’s oceans, but desalinating seawater has so far proven to be costly and energy-intensive.
Engineers at MIT have come up with a new desalination system that uses a shockwave to get the salt out of seawater. It could be a practical and energy-efficient method for desalination; water purification in remote locations and emergencies; and for cleaning brackish wastewater generated from hydraulic fracturing, the researchers say.
The most common desalination method involves boiling seawater, which takes a lot of energy. A slightly less energy-intensive method is reverse osmosis, in which seawater is pushed through a thick membrane that blocks sodium and chloride ions and lets fresh water through. But reverse osmosis is limited by the rate at which water molecules pass through the membrane. Plus, you still need a substantial amount of energy to force water through the membrane.
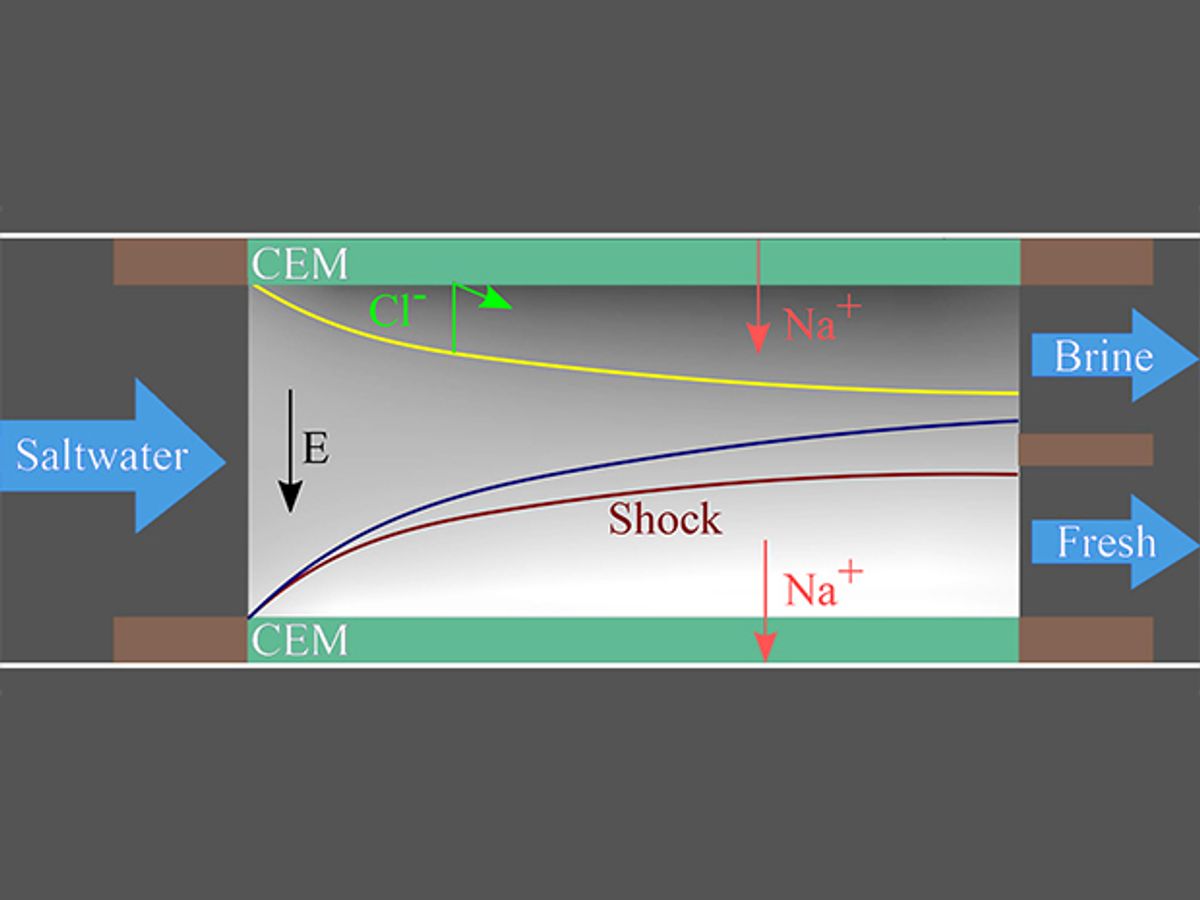
So Martin Bazant, a professor of chemical engineering and mathematics at MIT have turned to a process called shock electrodialysis that doesn’t require membranes and uses very little energy.
In the process, water flows through a charged porous material made of tiny glass particles that are sintered together. When a small electric current is applied across the porous glass, the salt ions accumulate on one side of the flow, creating an ion-rich side and an ion-deficient side. When the current is increased to a certain point, the charged surfaces of the porous media generate a shockwave that sharply divides the flowing water into two streams, one with fresh water and the other salty. The streams are simply physically separated at the center of the flow.
The generation of a shockwave in salt water was discovered a few years ago by researchers at Stanford University. But Bazant and his team have for the first time used it in a prototype electrodialysis system, which they reported on November 3 in the journal Environmental Science & Technology. Their prototype system can remove over 99% of various salts from solutions and recover up to 79% of the water. It can also remove contaminants like dirt and bacteria.
The system should be practical to scale up since it uses a simple setup and cheap materials. The team is now working on a larger prototype system.
In a MIT press release, Maarten Biesheuvel, a principal scientist at the Netherlands Water Technology Institute who was not involved in this research, said that the new work
“opens up a whole range of new possibilities for water desalination, both for seawater and brackish water resources, such as groundwater. It will be interesting to see whether the upscaling of this technology, from a single cell to a stack of thousands of cells, can be achieved without undue problems.”
Prachi Patel is a freelance journalist based in Pittsburgh. She writes about energy, biotechnology, materials science, nanotechnology, and computing.