Currently there is just one drug that has been approved for treatment of acute stroke—recombinant tissue plasminogen activator, or t-PA. Essentially it works by thinning blood clots. Researchers at the University of Georgia (UGA) announced last week that they have developed a magnetic nanoparticle that when combined with t-PA can make the drug significantly more effective.
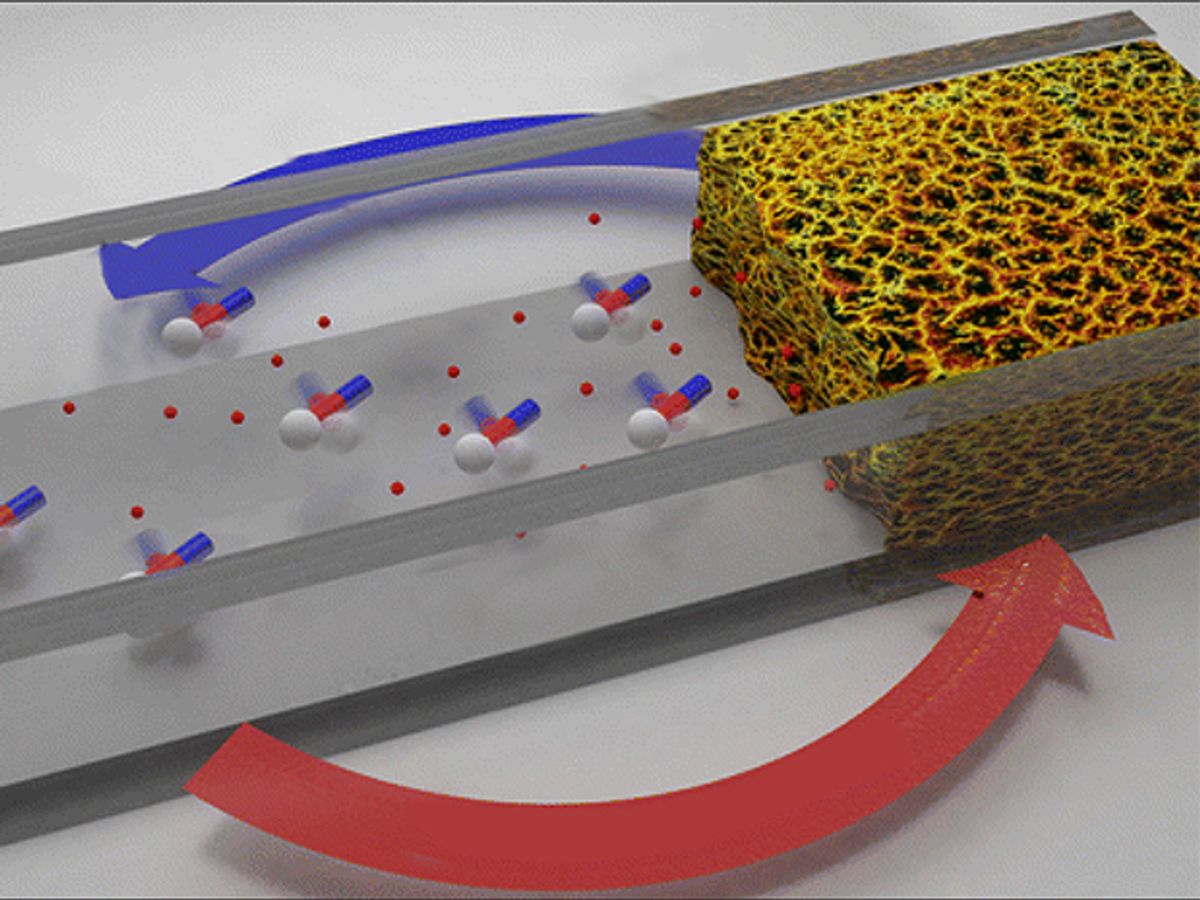
The Georgia researchers injected magnetic nanorods into the bloodstream. When stimulated by rotating magnets the nanorods act as a kind of mixing tool that shakes up blood clots that have already been thinned by t-PA.
The injected nanorods "act like stirring bars to drive t-PA to the site of the clot," said Yiping Zhao, professor of physics at UGA, in a press release. "Our preliminary results show that the breakdown of clots can be enhanced up to twofold compared to treatment with t-PA alone."
Like recent research at Penn State University in which nanorods were combined with magnets and ultrasonic waves to make them spin and thereby churn up cancer cells, the nanoparticles are referred to as nanomotors.
While this may offend the sensibility of some people’s understanding of what constitutes a motor, the term is used as well in the UGA research paper published in the journal ACS Nano.
In the research, the UGA team tested their technique on blood clots in mice. They injected a mixture of t-PA and the nanorods and then activated the nanorods with two revolving magnets once they had reached the blood stream. By triggering the spinning of the nanorods so early it helps to push the drug towards the blood clot, like fans wafting them along.
With the researchers reporting a two-fold increase to the breakdown of blood clots over just t-PA alone, the technique offers a much-needed boost to stroke treatment. While t-PA is good a thinning the blood clots, it has an unwanted side effect of thinning the blood throughout the body resulting in a real threat of hemorrhaging.
"We want to improve the efficiency of this drug, because too much of it can lead to serious bleeding problems," said Rui Cheng, a researcher on the project, in a press release. "This approach may one day allow physicians to use less of the drug, but with equal or improved effectiveness."
The next step for the researchers will be to find a nanorod material that is more biocompatible than what was used in these initial experiments. They would also like to develop a chemistry model that will connect clot dissolving speed and other parameters with the material being used.
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.