In emerging technologies—of which, nanotechnology is a leading example—it's important to recognize and encourage innovation wherever it exists, because the ecosystem in which it flourishes is so delicate. There is the so-called funding—or innovation—gap that the President’s Council on Science and Technology (PCAST) tried to tackle a couple of years back. But even if an emerging technology does get the funding to go commercial, it seems the chances of its success are so remote that it’s amazing that any new technologies come to market at all. One need only look at the examples of A123 Systems or the UK-based Oxonica to see how large amounts of funding are no guarantee that an emerging technology—and a nanotechnology in these cases—will result in a successful business.
Nanomagnetics’ primary application focus was in the data storage market. But I came to know Eric by inviting him to speak at a conference on how Nanomagnetics’ use of an iron-storage protein ferritin to make nanoscale magnetic particles could be exploited to enable forward osmosis for water purification. It was really an elegant approach and captured my interest as it did the audience of the conference.
Like other small companies that have dared to challenge the big data storage behemoths it was a rocky nearly-decade-long road for Nanomagentics that ended when the company finally closed its doors in 2006.
Last year I saw that Eric was leading a new company—Endomagnetics. It has a novel medical diagnostic tool for detecting the likely sites for cancer-infected lymph nodes to help provide early diagnosis for breast cancer. He and his new company were highlighted in a BBC News Horizon feature on nanotechnology. A video of the interview can be found here.
So, I decided to catch up with Eric and ask him about his new venture and for him to discuss the innovation landscape for nanotechnology and how it has evolved over the last 15 years.
Q: How does the Endomagnetics diagnostic tool improve on current diagnostic techniques for breast cancer?
A: Our tool supports surgeons in determining whether or not cancer has spread (also known as ‘staging’). In addition to surgery to remove a tumor, the current standard of care involves a procedure called ‘sentinel lymph node biopsy’ (SLNB).
When cancer spreads, metastatic cells separate from the tumor and are transported by the lymphatic system, then filtered by the lymph nodes first in line to drain from the tumor – the ‘sentinel’ nodes. By identifying these lymph nodes and checking for the presence of metastases, surgeons can determine whether cancer has started to spread and whether additional intervention is required.
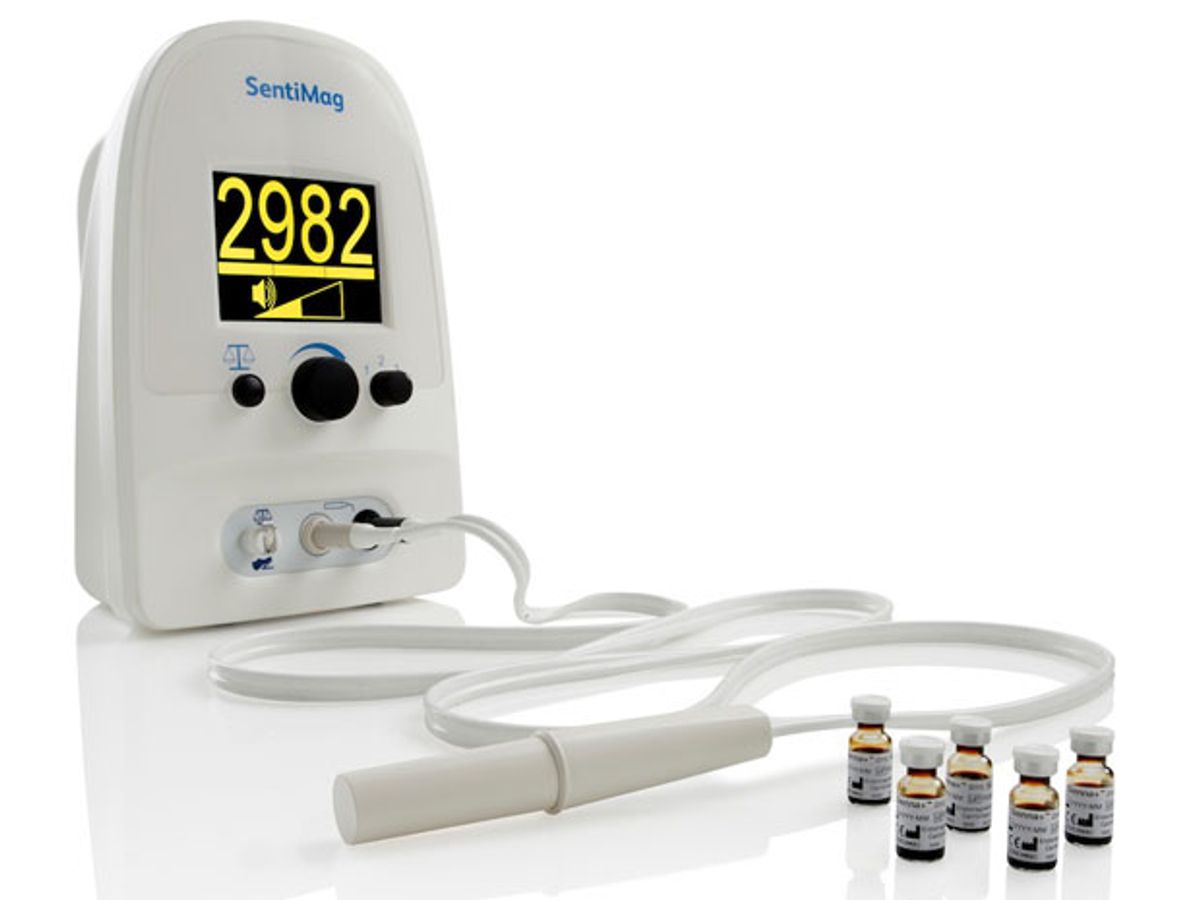
The Endomagnetics’ tool isn’t a diagnostic technique in itself, but a key part of this diagnostic procedure. The system that Endomagnetics developed comprises two components—a detector (SentiMag) and a magnetic nanoparticle tissue marker (Sienna+). Sienna+ is injected into the tissue near the tumor, transported by the lymphatic system, and then filtered out by the sentinel lymph node(s). The SentiMag detects the minute magnetic signature of Sienna+ in the lymphatic tissue to locate the sentinel lymph nodes for subsequent excision and analysis.
The current standard technique relies on radioisotope-labeled nanoparticles, such as sulfur colloid or albumen, which are used in conjunction with a gamma detector. The downside of this technique is that the radioisotope (99mTc) has a 6-hour half-life and isn’t broadly available. This limits access to the standard of care, and regulations around the use and storage of radioactive materials create workflow and cost challenges. The transition to a magnetic technology resolves all of these issues, improving availability, workflow and costs.
Q: Your previous company, NanoMagnetics, used nanometer-scale magnetic particles within hollow protein spheres. It appears magnetic particles are the basis of your new products at your new company. How is your new technology different from that of NanoMagnetics?
The basis for Sienna+ is an iron oxide nanoparticle that is superparamagnetic at room temperature, whereas NanoMagnetics’ cobalt-platinum particles were ferromagnetic at room temperature. In short Sienna+ cannot ‘store’ a magnetic orientation at room temperature, which is important so that particles do not stick together and precipitate.
In addition, the coating on the Sienna+ particles is a well-established organic coating to ensure biocompatibility and stability, whereas the NanoMagnetics protein coating was used as a carbon source for encapsulating the particles post-heat treatment.
Finally, a more strategic difference is that Endomagnetics sourced the Sienna+ particles from a third-party manufacturer rather than developing its own. The advantage was that we could select ‘best of breed’ to meet our design requirements and also could achieve the scaled manufacturing and quality standards essential for medical device applications quickly.
Q: At NanoMagnetics your initial and primary target market was data storage. What are the advantages and challenges of targeting the medical device and diagnostics market instead?
A: Maybe the best way to answer this question is to talk about how ready our customers are/were to adopt the products. With Endomagnetics, we have solid clinical data that demonstrates our system’s performance versus the standard technique, and we designed the system to match the existing procedure as closely as possible. As surgeons are our customers, they appreciate that the SentiMag/Sienna+ system solves a problem and does so without upsetting existing workflow—it actually improves it. For the customers, the technology is transparent as they can achieve their objectives and enjoy the benefits.
A challenge for Endomagnetics has been the regulatory environment, particularly along the border between the definitions for drugs and devices. We have successfully negotiated this border to achieve device approval for our particles, but the sands are always shifting and vary from one country to another.
With NanoMagnetics, our target customers were hard disk media companies. We had developed good lab demos to demonstrate how well we addressed the challenge in increasing storage density, but our materials and related manufacturing techniques were pretty foreign. So there was a much wider gap to readiness—we needed something more advanced than laboratory demonstrations and also needed to find a way to readily integrate the manufacturing processes. On reflection, our strategy should have been to focus on integrating our nanoparticles and manufacturing hard disk media, rather than on selling nanoparticles and manufacturing techniques. Had we done this, we could have more easily slotted into the supply chain for disk drives. Regardless, the hard disk drive industry is just a really tough place to be, let along trying to introduce a completely new technology from outside the existing players.
Q: Endomagnetics has been selected as one of the “UK’s most exciting, high-potential new businesses" by the Real Business Future 50 project. In the last few years, the UK has come under criticism—for example from Nanoco, which threatened to move abroad—for its lack of support of small start-up companies. How would you describe your relationship with the UK government in supporting getting your company from the lab to the fab, so to speak?
A: Endomagnetics probably wouldn’t exist without support from the UK’s Technology Strategy Board. The TSB provided a development grant that was matched with the company’s first seed capital round, and the result was an engineering prototype of the current SentiMag. Prior to that, the system had been based on a cryogenically cooled magnetic sensor that was impractical for use both in the clinic and in reproducible manufacturing. The company has since been supported by two further grants that help expand our product pipeline and extend the indications for the current system, so I’d say our relationship with the UK government has been very supportive.
I believe the Nanoco example was related to significant funding to support larger-scale manufacturing. I don’t know enough to comment on Nanoco’s challenges, but I know the government has to walk a fine line regarding State Aid rules. I am not a fan of governments propping up industries, but am a fan of smaller, strategic interventions that catalyze innovation and then leaving the market to sort the rest.
Q: There has been some frustration in the innovation process for bringing emerging technologies to market and some new frameworks are being examined to streamline and enhance the current processes. How have you seen the innovation process evolve since you first launched NanoMagnetics in 1997?
A: I think the only ‘new’ framework I’m really familiar with is ‘open innovation.’ Indeed, this is how Endomagnetics has innovated to its current products so quickly. As mentioned before, we designed our requirements for a particle then went out to source it. Had we developed our own, it would have taken significant time and could have locked us into one platform without the flexibility to respond to changing customer requirements.
Being ‘closed’ was a limitation of NanoMagnetics in that we were always trying to force-fit our fantastic material into the customer’s requirements. While our material did have compelling features, we would have been better to focus on the customer requirement and then sample from a number of possible solutions (even from outside) rather than spending lots of resource trying to adapt our own.
Q: Finally, how will Endomagnetics’ diagnostic tool—and others—improve treatments for a range of diseases and how will the doctor-patient relationship evolve with these new technologies?
A: One thing I’m really pleased by is that we are introducing a new technology that optimizes the surgical process for all involved. The patients are happier as our material can be injected following anesthesia, so we remove the pain of injection and fears over radioactive materials. The surgeons are happier as they can make the injections themselves and not bother with arranging schedules with the nuclear medicine department or managing radioactive tissue waste. The hospitals and clinics are happier as the technology makes the technique available where it wasn’t before, and less expensive where it was.
Resolving these issues has a direct impact on the doctor-patient relationship, and I am certain that similar technology developments to improve efficiency, efficacy and safety will achieve the same.
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.