Back in 2009 IBM pushed the boundaries of surface microscopy when they developed a technique for noncontact atomic force microscopy (AFM) that enabled the resolving of single atoms in molecules. Since then IBM has been working on this foundational work to develop a Kelvin probe force microscopy (KPFM) technique that enabled the first imaging of the charge distribution within a molecule.
Now the team, based in Zurich, that have been at the forefront of this research have just completed some collaborative research with the Royal Society of Chemistry (RSC) and the University of Warwick in which they have imaged the synthetic molecule Olympicene (named after its resemblance to the five-ring design of the Olympic symbol) to the point where they could not only image individual hydrogen atoms but also manipulate them. Here's a video describing the research:
While IBM’s noncontact AFM had shown the ability to image hydrogen atoms previously, this latest research made that capability more concrete, according to Leo Gross, Scientist, at IBM Research Zurich.
A unique feature of the IBM-developed AFM technique is that it can image individual molecules whereas other techniques require that the molecules be collected into an aggregate crystal form. But perhaps even more importantly, what this line of research has demonstrated rather clearly is that they can use the tip of the AFM to induce reactions, such as forming a bond or extracting an atom.
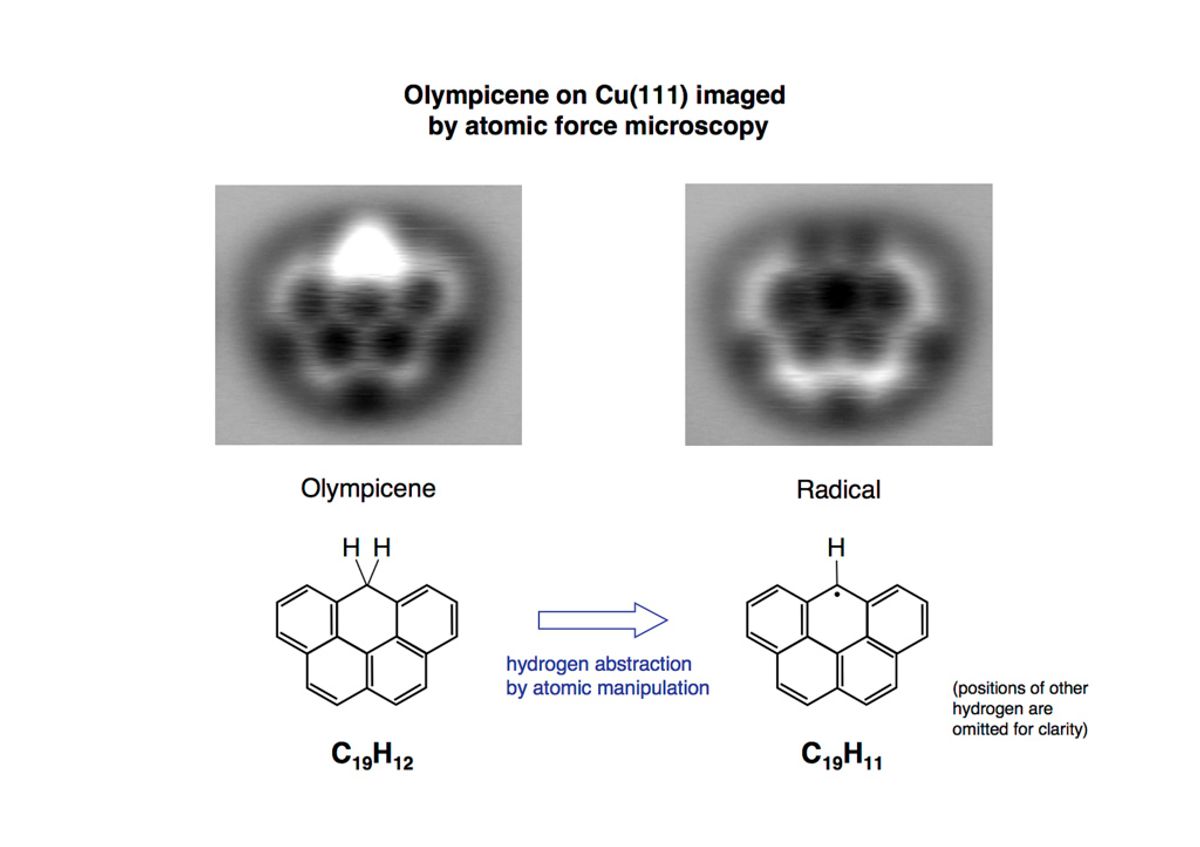
This can be seen most clearly in the images the researchers created of the Olympicene molecule. In one you can see the bonds between the links of the molecule is much brighter. This is where there are two hydrogen atoms attach to the olympicene molecule. In another image you can see that same site is no longer as bright because they have removed one of the hydrogen atoms, creating what they dubbed the Olympicene radical.
While Gross believes that the imaging and manipulation of hydrogen atoms marks the limits in scale for this technique, there are still things at this size they would like to investigate. “We would like to look at things such as the adsorption, bonding and bonding angles, position, distances,” says Gross.
In addition to this line of research, Gross sees a great deal of potential is the KPFM technique. “We want to use this ability to image charge distribution and charge separation to study molecules for organic solar cells where charge separation is very important, and also for molecular electronics, such as single-molecule devices, which have a functionality depending on single electrons. The combined techniques of AFM and KPFM give us a very good tool to study these single-electron devices.”
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.