I have detailed in the past how biologists have felt forlorn in the world of imaging on the nanoscale.
Dejected biologists have continued to seek out some tools to aid them in seeing the nanoscale world. This search found some sympathetic researchers at the University of California Santa Barbara.
In an article over at Nanowerk, which delves into the details of the research that was first published in Nature Nanotechnology, the lead researcher on the project explains the motivation for developing what is being described as a “high-throughput label-free nanoparticle analyzer”.
"Our collaborators informed us of the significant need for similar technologies for particle analysis on the nanoscale for studying blood and other potential clinical applications, and we set to work," explains Jean-Luc Fraikin, the first author of the paper. "Only once we became more deeply involved in the project did we begin to appreciate the broader context for this work, given the range of applications for which nanoparticles in this size range are being developed, and the lack of practical sizing technologies that were available."
The device they developed is able to measure both the size of nanoparticles and their concentration in solution at a rate of 100,000 particles per second. This stands in stark contrast to the other methods that have been available for measuring these metrics, namely averaging and electron microscopy. With averaging there was no way of determining the varying sizes of the particles and electron microscopy was time consuming and expensive.
"The low-cost, scalable fabrication method, as well as the simple readout electronics, make this analyzer potentially useful in a wide range of applications," noted Fraikin in the Nanowerk article.
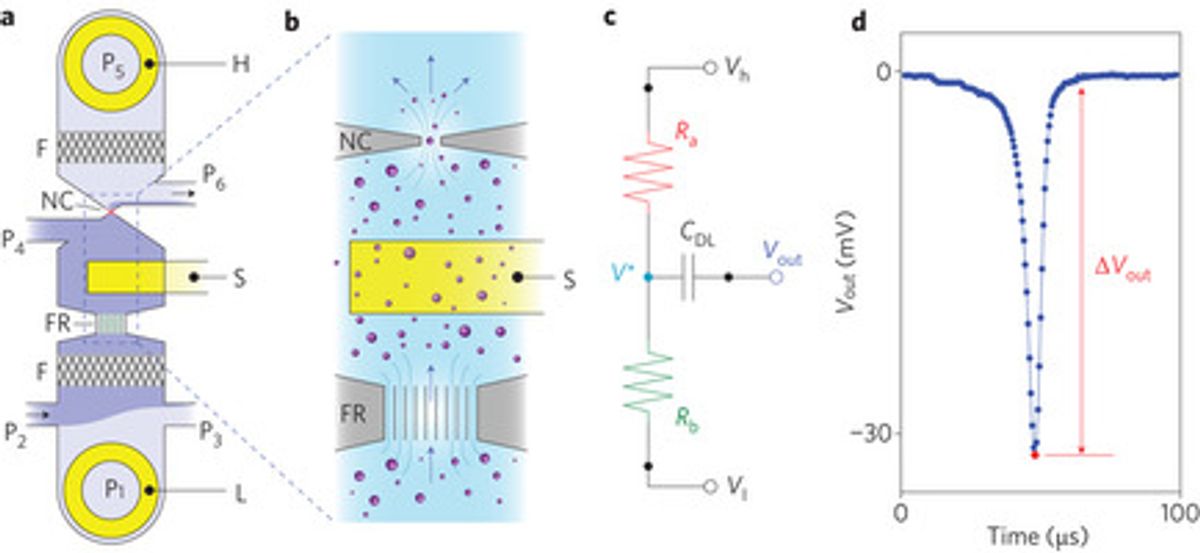
The analyzer is a micro-fluidic design with a micro channel that leads the analyte through a sensor consisting of “two voltage-bias electrodes and a single, optically lithographed readout electrode embedded in the microchannel.” "Together, these components form a fluidic voltage divider that yields wide-bandwidth electrical detection of particles as they pass through the nanoconstriction," Fraikin explains in his Nanowerk interview. "The sensing electrode is embedded in the channel between the fluidic resistor and the nanoconstriction. As a particle enters the nanoconstriction, it alters the ionic current and, because of the voltage division between the fluidic resistor and the nanoconstriction, changes the electrical potential of the fluid in contact with the sensing electrode."It would seem the time is right for having a cheap and easy-to-use device for measuring the size and concentration of nanoparticles in all sorts of solutions, not just biological ones. If Geoffrey Ozin is correct in his estimation that nanomaterials are at a saturation point where some more measurement of what we have is needed, then this could be tool to help in that regard.
But as the Nanowerk article points out, “While the nanoparticle analyzer discussed here measures two very basic physical properties of nanoparticles – their size and concentration in solution – many other characteristics of nanoparticles, such as surface charge, surface roughness, detailed particle shape and electric or magnetic polarizability are critical to their applications and will require tools for the high-throughput measurement these properties as well.”
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.