Magnetic Field Makes a Better Lithium-Ion Battery for Electric Vehicles
Magnetic field alignment enables thick-electrode batteries with a higher energy density at a lower cost
Researchers at MIT have developed a manufacturing approach for the electrode material of lithium-ion (Li-ion) batteries that should lead to a threefold higher area capacity for conventional electrodes. In the devices that they have fabricated thus far they have measured 12 milliamp hours per square centimeter (mAh cm2) versus the 4 mAh cm2 seen in conventional electrodes at normal charge-discharge rates.
The technique the MIT team developed involves using an external magnetic field to align pores in the electrode material in a particular way to achieve these much higher capacity numbers. And because of some of the unique properties of the resulting material, these Li-ion batteries may be far better suited for the requirements of electric vehicles (EVs).
One of the properties the researchers were trying to achieve with this magnetic templating approach was anisotropy, which means that the material’s physical properties behave differently in one direction than another. Wood is a good example of this in which it is strong in the direction of its grain but weaker in the direction that goes against its grain.
Research has shown that structures with anisotropic pores that are aligned in the direction of charge transport have an advantage by enabling faster transport at high pore density. This means that really thick electrode material, which can provide high storage capacity, can still have fast transport of despite its thickness. Unfortunately, Li-ion batteries of today are not designed to take advantage of this anisotropic property.
Another property that the researchers were trying to achieve in the electrode material was what is called low-tortuosity. This means reducing the pores’ twists and turns. If you can reduce this tortuosity, you can improve the conductivity of the electrode material. This property again becomes of critical concern when you thicken the electrode material to gain greater capacity.
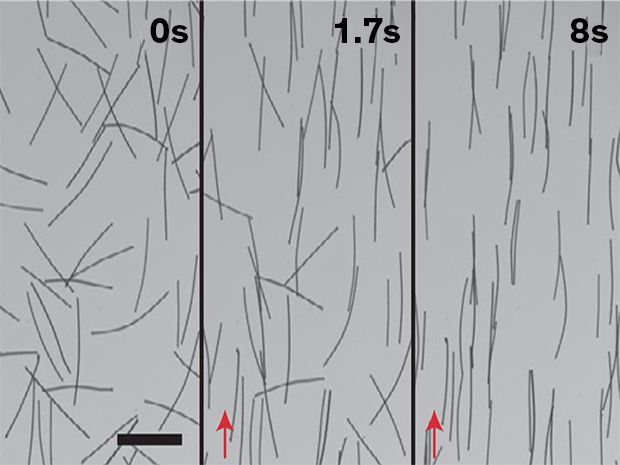
The actual process that the MIT team developed to achieve these properties starts with dispersing a magnetic material in an electrode particle suspension. This magnetic material will be later sacrificed, but before it is eliminated it makes it possible for an external magnetic field to creates an anisotropic ordering of the material.
While two different sacrificial magnetic materials were tested, both achieved the main aim: ordered arrays of pore channels within the electrode material. After the magnetic phase has been oriented, the solvent phases are evaporated to leave a solid electrode. Full descriptions of the two approaches are described in the journal Nature Energy.
In the lithium cobalt oxide (LCoO2) electrodes produced with these two approaches not only did they increase the usable capacity of the electrodes by a factor of three, but they also possess discharge profiles that are more in line with the drive cycles of EVs than the constant current discharge of conventional Li-ion batteries.
The researchers report that the electrodes with aligned pore channels produced deliver areal capacities (a measurement of storage capacity relative to physical dimensions) above 8 mAh/cm2, more than twice that of the highest areal capacity of conventional Li-ion electrodes.
The MIT team believes that the magnetic field alignment is rapid, scalable to large areas, and could serve as the basis for new fabrication processes that enable thick-electrode batteries of higher energy density at a lower cost.
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.