Molecular electronics promise a time when the basic building blocks of electronics are individual molecules. Of course, the transistor is today’s fundamental building block for computing. And along these lines, five years ago, researchers showed that it was possible to make a transistor from a single atom.
That work and much that has followed since is not going to lead to practical devices any time soon. But all of this basic research may someday result in practical devices.
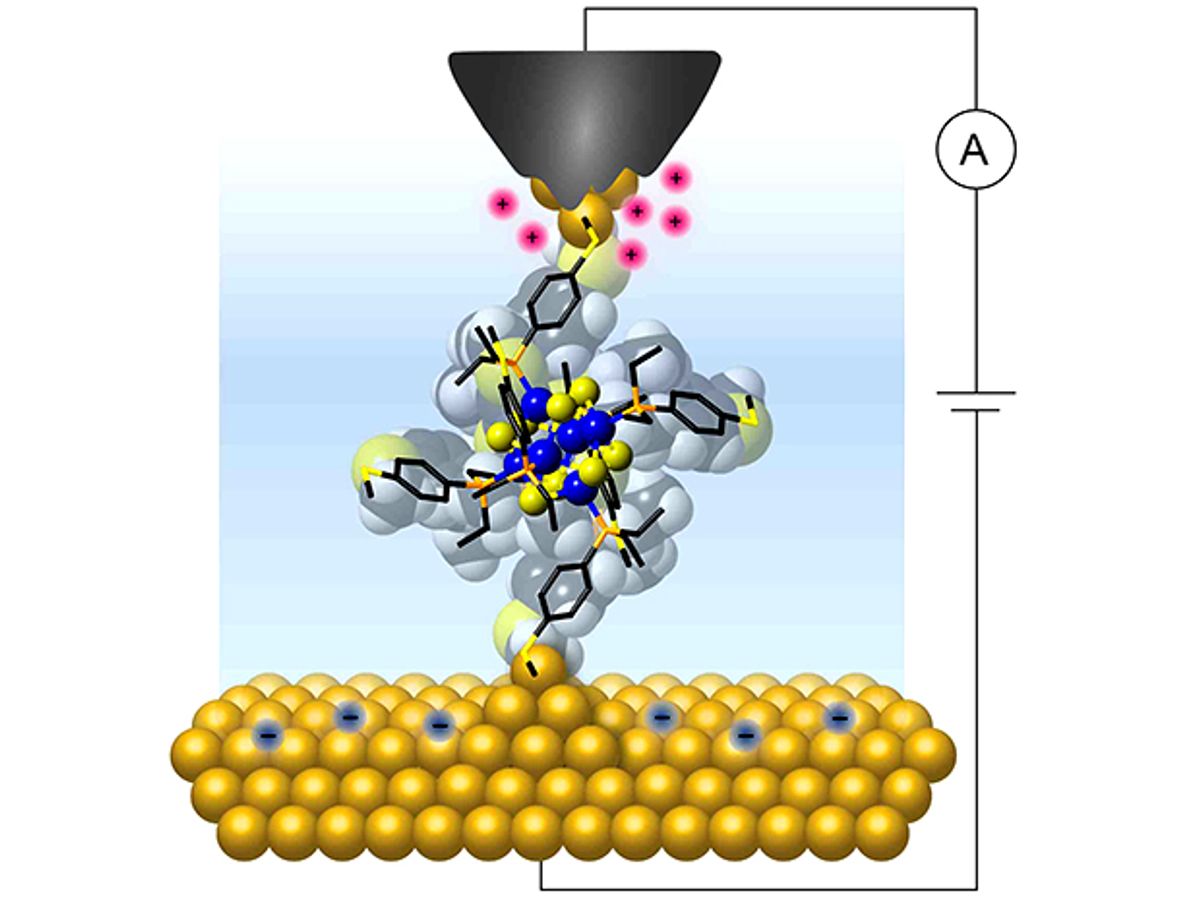
In the latest step in this long journey, researchers at Columbia University have fabricated a small cluster of atoms into a two-terminal transistor capable of switching from insulator to conductor when charge is added or removed, one electron at a time.
In research described in the journal Nature Nanotechnology, the Columbia researchers were able to create a geometrically ordered cluster of atoms with a central core consisting of just 14 atoms. They linked that 14-atom core to gold electrodes that allowed them to monitor its electrical response as they varied the applied bias voltage.
With this arrangement, the researchers created a device that reproducibly demonstrated a current blockade at room temperature. While there have been many other devices that function as a single-electron transistor, most do not do so reproducibly, and very few have demonstrated this capability at room temperature, according to Latha Venkataraman, leader of the research team at Columbia, in an e-mail interview with IEEE Spectrum.
“One of the key innovations in this research was our use of atomically precise inorganic clusters made of just 14 atoms as the functional element in these devices,” explained Venkataraman. “And we also developed a novel method to gate the devices using just two terminals that relies on an electrochemical environment.”
In this electrochemical environment, the researchers were able to change the applied bias voltage to alter the density of ions around the molecular junction, charging the cluster. “By changing the electrostatic environment around the cluster, we allow its charge state to be modified enabling a current to flow across the single-molecule junction,” says Venkataraman.
"We found that these clusters could perform very well as room-temperature nanoscale diodes whose electrical response we can tailor by changing their chemical composition," says Venkataraman in a press release.
Quantum dots—which are nanoscale structures within semiconducting materials that hold tiny reservoirs of electrons, acting like an artificial atom—have been able to do more or less the same thing as this cluster of inorganic atoms. However, quantum dots are not perfectly reproducible, some are smaller and some are larger, leading to their properties not always being consistent.
The Columbia researchers were able to make their inorganic molecular clusters with complete control over their structure, resulting in perfectly identical shapes and structures every time. This makes it possible to fabricate and measure thousands of junctions with reproducible characteristics.
“With these molecular clusters, we have complete control over their structure with atomic precision and can change the elemental composition and structure in a controllable manner to elicit certain electrical response,” says Venkatraman.
In addition to reproducibility and the ability to operate at room temperature, the on/off ratio for this device is extremely high, a quality sorely lacking in other molecular-based transistors.
Along with its high on/off ratio, each electron transited through the junction by first stopping on the cluster momentarily before passing on—a property known as a “sequential” mode of charge flow. This stands in contrast to typical small-molecule junctions in which the electrons go through the junction from one electrode to another continuously so that the number of electrons on the molecule at each instant of time is not well-defined.
"We say the cluster becomes 'charged' since, for a short time interval before the transiting electron jumps off into the other metal electrode, it stores one extra charge," says Bonnie Choi, a graduate student and co-lead author of the work, in a press release. "Such sequential, or discrete, conduction mode is due to the cluster's peculiar electronic structure that confines electrons in strongly localized orbitals.”
Choi added that these orbitals of the electrons also account for the observed “current blockade” regime when a low bias voltage is applied to a cluster junction.
“The current drops to a very small value at low voltage as electrons in the metal contact don't have enough energy to occupy one of the cluster orbitals,” said Choi. “As the voltage is increased, the first cluster orbital that becomes energetically accessible opens up a viable route for electrons that can now jump on and off the cluster, resulting in consecutive 'charging' and 'discharging' events. The blockade is lifted, and current starts flowing across the junction.”
In continuing research, the Columbia team will be looking to increase the on/off ratio of the devices and increase the number of atoms used while maintaining the precision and uniformity of the devices.
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.