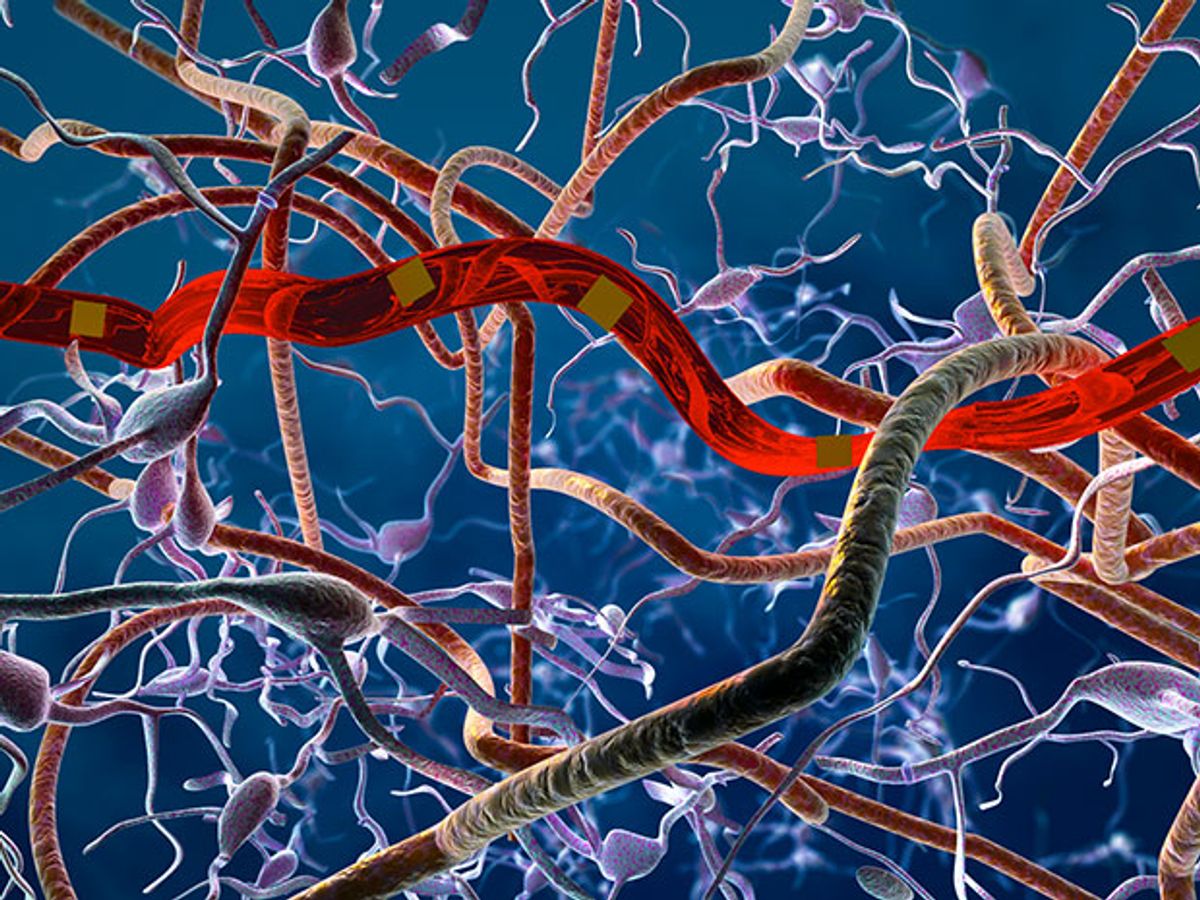
For over two decades, electrodes implanted in the brain have made it possible to electrically measure the activity of individual neurons. While the technology has continued to progress over the years, the implanted probes have continued to suffer from poor recording ability brought on by biocompatibility issues, limiting their efficacy over the long term.
It turns out that size matters: In this case, the smaller the better. Researchers at the University of Texas at Austin have developed neural probes made from a flexible nanoelectronic thread (NET). These probes are so thin and tiny that when they are implanted, they don’t trigger the human body to create scar tissue, which limits their recording efficacy. Without that hindrance, the threadlike probes can work effectively for months, making it possible to follow the long-term progression of such neurovascular and neurodegenerative diagnoses as strokes and Parkinson’s and Alzheimer’s diseases.
In research described in the journal Science Advances, UT researchers fabricated the multilayered nanoprobes out of five to seven nanometer-scale functional layers with a total thickness of around 1 micrometer.
“The thickness of all the functional layers, including recording electrodes, interconnects, two to three layers of insulation are all on the nanoscale,” explained Chong Xie, an assistant professor and one of the paper’s co-authors, in an e-mail interview with IEEE Spectrum. “This ultra-small thickness is crucial for the ultra-flexibility that is necessary to completely suppress tissue reactions to the implanted probes and leads to reliable long-term neural recording.”
Xie adds that the device’s flexibility is not the result of choosing the softest materials, but instead by engineering the dimensions and geometry of the device—in particular the ultra-thin thickness.
The probe functions just like currently used neural electrodes. The recording sites (electrodes) are evenly spaced along the probe, which are individually addressed by micro-fabricated interconnects that connect to bonding pads for external inputs/outputs.
In the demonstration of the device, the electrodes were implanted into a living mouse cortex. The voltage signal is recorded at a high sampling rate (greater than 20 kiloHertz) detected on the electrodes, which represents the electrical activities from nearby neurons, including action potentials and local field potentials.
Xie and his colleagues faced some difficult technical challenges to make the neural probes extremely thin and to keep them implanted in a living brain with continued function for months. The researchers already knew from other work that larger neural probes made from thicker and stronger materials experienced structural damage that reduced their function after being implanted for just a few weeks. The key, according to Xie, was engineering the structure, materials, and fabrication details so that the interlayer adhesion is greatly enhanced. They also were surprised with how well some of the materials behaved.
“It is quite amazing that insulation layers with just hundreds of nanometers in thickness are sufficient to protect all the electrical components including millimeter-long interconnects in a living mouse brain for months,” explained Xie. “From our previous experience we knew that the photoresists we were using make good insulation with low defect rates, which was the reason for our material choice, but we were still surprised about its supreme performance.”
While the initial results of the probe were positive, there remain some pretty significant challenges ahead. The implementation strategy for the probes was tricky business.
These probes have a cross section area greater than 100 times smaller than that of a human hair and cannot free stand in air. As a result, the researchers had to come up with a way to precisely manipulate them to targeted positions and engage with microscale shuttle devices with micrometer accuracy. If this technology is to ever reach beyond the lab, this part of the process will need to be optimized.
“The surgical procedure for implanting these probes in a clinical environment needs to be further optimized,” said Xie. “We need to design new devices and an implantation strategy for clinical application. We have already started collaboration with neurosurgeons to develop the next generation of NETs for human patients.”
The next steps in the research will look to test the neural probes in primates. In the meantime, Xie and his colleagues are looking to improve the underlying technology.
Xie added: “I am continuing to enrich the technologies for novel neural interfaces in rodent models, including high-density recording and chronic recording over years and longer.”
Dexter Johnson is a contributing editor at IEEE Spectrum, with a focus on nanotechnology.